Toward Actionable Digital Twins for Radiation-Based Imaging and Therapy: Mathematical Formulation, Modular Workflow, and an OpenKBP-Based Dose-Surrogate Prototype
Digital twins for radiation-based imaging and therapy are most useful when they assimilate patient data, quantify predictive uncertainty, and support clinically constrained decisions. This paper presents a modular framework for actionable digital twi…
Authors: Hsin-Hsiung Huang, Bulent Soykan
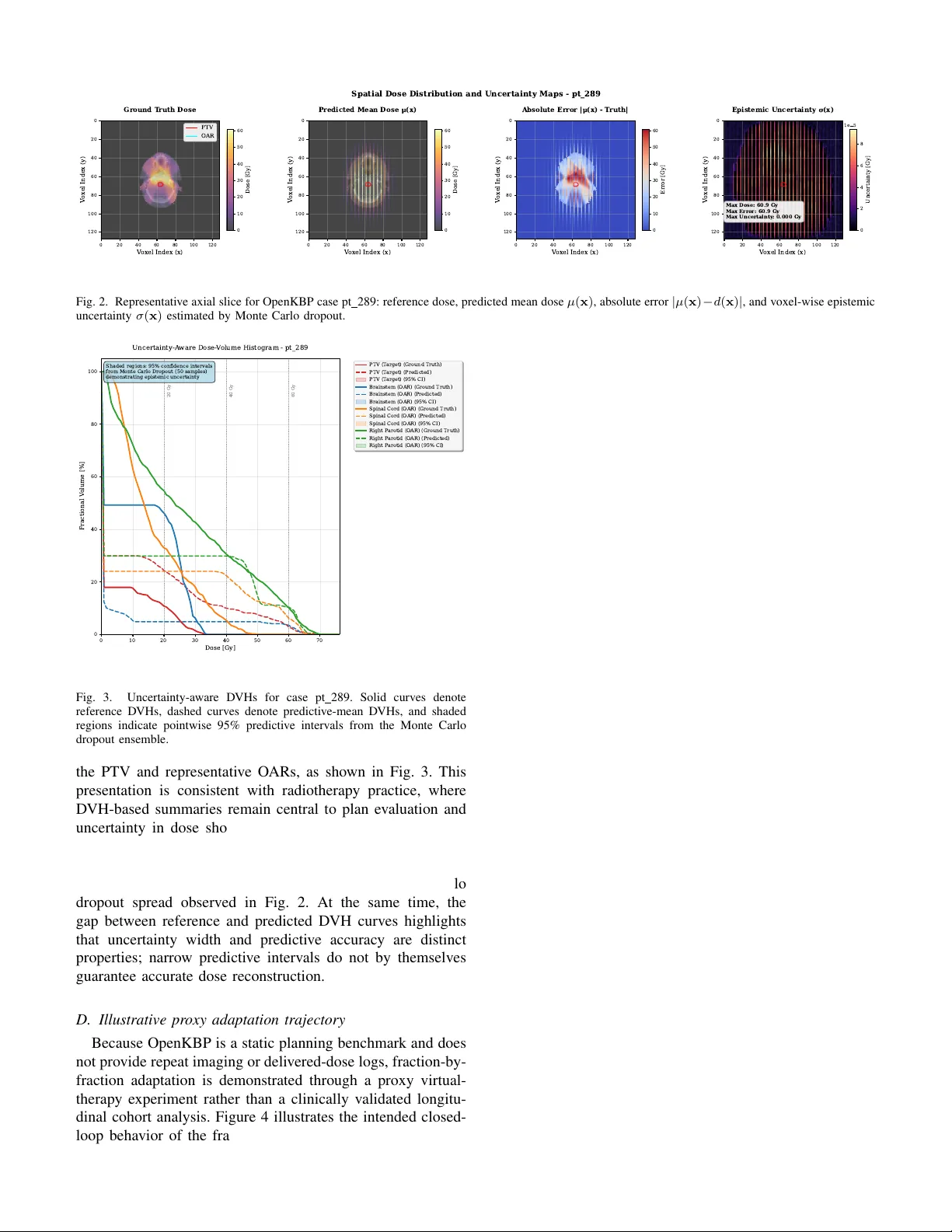
IEEE T ransactions on Radiation and Plasma Medical Sciences , V OL. XX, NO. X, MONTH 2026 1 T o w ard Actionable Digital T wins for Radiation-Based Imaging and Therapy: Mathematical F ormulation, Modular W orkflo w , and an OpenKBP-Based Dose-Surrogate Prototype Hsin-Hsiung Huang, Member , IEEE, and Bulent Soykan, Member , IEEE Abstract —Digital twins for radiation-based imaging and ther- apy are most useful when they assimilate patient data, quantify predicti ve uncertainty , and support clinically constrained deci- sions. This paper presents a modular framework for actionable digital twins in radiation-based imaging and therapy and instanti- ates its reproducible open-data component using the Open-access Knowledge-Based Planning (OpenKBP) benchmark. The frame- work couples PatientData, Model, Solver , Calibration, and Deci- sion modules and formalizes latent-state updating, uncertainty propagation, and chance-constrained action selection. As an initial implementation, we build a GPU-ready PyT orch/MON AI reimplementation of the OpenKBP starter pipeline: an 11- channel, 19.2M-parameter 3D U-Net trained with a masked loss over the feasible region and equipped with Monte Carlo dropout for v oxel-wise epistemic uncertainty . T o emulate the update loop on a static benchmark, we introduce decoder - only proxy recalibration and illustrate uncertainty-aware virtual- therapy evaluation using D VH-based and biological utilities. A complete three-fraction loop including r ecalibration, Monte Carlo inference, and spatial optimization executes in 10.3 s. On the 100- patient test set, the model achieved mean dose and DVH scores of 2.65 and 1.82 Gy , r espectively , with 0.58 s mean inference time per patient. The OpenKBP case study thus serves as a reproducible test bed for dose prediction, uncertainty propagation, and proxy closed-loop adaptation, while future institutional studies will address longitudinal calibration with delivered-dose logs and repeat imaging. Index T erms —Digital twin, adaptive radiation therapy , external-beam radiotherapy , knowledge-based planning, dose prediction I . I N T R O DU C T I O N The idea of a digital twin emerged in aerospace and engi- neered systems as a virtual counterpart that remains linked to its physical system through data, modeling, and predic- tion [1]. In medicine, the concept is especially compelling because patient care already generates a rich digital record of images, treatment plans, deliv ery logs, and clinical outcomes. Radiation-based imaging and therapy are particularly well suited to this paradigm because they combine repeated mea- surement, model-based prediction, and decisions that must be This work did not in volve human subjects or animals in its research. H.-H. Huang was supported in part by the U.S. National Science Foundation under Grants DMS-1924792 and DMS-2318925. Hsin-Hsiung Huang is with the School of Data, Mathematical, and Statisti- cal Sciences, University of Central Florida, Orlando, FL 32816 USA (e-mail: hsin.huang@ucf.edu). Bulent Soykan is with the Institute for Simulation and Training, Uni versity of Central Florida, Orlando, FL 32826 USA (e-mail: Bulent.Soykan@ucf.edu). revised under strict safety constraints. In this setting, a useful digital twin is not merely a static simulator . It must assimilate new patient information over time, represent uncertainty in its predictions, and support actions such as plan ev aluation, adaptation, or treatment selection. Radiation oncology already pro vides two ingredients needed for such systems. First, deep learning has adv anced dose prediction and knowledge-based planning, with the Open- access Knowledge-Based Planning (OpenKBP) AAPM Grand Challenge providing a standardized benchmark for volumetric dose surrogates and D VH-based ev aluation [2], [3]. Second, digital-twin thinking has matured in oncology and medical imaging, including recent patient-specific forecasting frame- works and MRI-based response models [4], [5]. T ogether , these developments suggest a timely opportunity to connect reproducible AI-based dose modeling with the updating and decision logic expected of an actionable digital twin. That connection remains incomplete. Most published dose- prediction models are still one-shot feed-forward predictors. They estimate a reference dose distrib ution from anatomy and structure masks, but they do not explicitly maintain a patient state, incorporate new observations during treatment, or express uncertainty in a way that can guide decisions. A clinically meaningful radiation-therapy digital twin therefore requires more than predictiv e accuracy alone. It requires a formal updating mechanism, uncertainty propagation from vox els to clinically interpretable summaries, and a decision layer that can compare candidate actions under safety and deliv erability constraints. Open datasets are indispensable for reproducibility , yet they rarely contain the longitudinal mea- surements needed for a full bidirectional clinical twin. This pa- per addresses that tension by using OpenKBP for reproducible dose-surrogate de velopment, uncertainty-aware e valuation, and proxy sequential updating, while reserving true longitudinal validation for future institutional studies with deliv ered-dose logs and repeat imaging. W ithin that scope, we dev elop an actionable digital-twin framew ork organized around PatientData, Model, Solver , Cal- ibration, and Decision. The open-data implementation uses an OpenKBP-style 3D U-Net trained with a masked loss over the feasible region, stochastic inference through Monte Carlo dropout for voxel-wise epistemic uncertainty , and a proxy sequential calibration step based on decoder-only transfer learning. These components are coupled to a virtual-therapy IEEE T ransactions on Radiation and Plasma Medical Sciences , VOL. XX, NO. X, MONTH 2026 2 decision module that ev aluates candidate actions using DVH- based summaries and uncertainty-aware biological utilities. The result is both a mathematical formulation of an action- able digital twin and a reproducible prototype sho wing how uncertainty and updating can be embedded in a benchmark dose-prediction workflow . I I . B AC K G RO U N D A N D R E L A T E D W O R K A. Updateable patient models and modular twins Across engineering and biomedical settings, digital twins are virtual counterparts linked to their physical systems through data assimilation, prediction, and decision support [1]. In oncology , recent examples span MRI-based patient-specific digital twins for chemotherapy optimization [4] and modular framew orks such as T umorT win [5]. These lines of work col- lectiv ely motiv ate the present focus on updating, uncertainty- aware prediction, and actionability . B. Radiation-therapy surr ogates and open benchmarks OpenKBP enables fair comparison of dose-prediction meth- ods for knowledge-based planning using standardized voxel- wise and D VH-based metrics [2], [3]. Attention-aw are 3D U-Net variants hav e also been ev aluated on the benchmark and sho w that architectural refinements can improv e emphasis on clinically rele vant anatomy in head-and-neck IMR T dose prediction [6]. Our work builds on this benchmark tradition but mov es beyond static prediction by integrating uncertainty , proxy recalibration, and a decision layer . I I I . M O D E L O F A N A C T I O N A B L E D I G I TA L T W I N A. Latent state, observations, and updating Let x t denote the latent patient state at time t , including anatomy , targets and organs-at-risk, accumulated dose sum- maries, and latent response or toxicity parameters. Let y t denote observations, including CT or CBCT , deliv ered dose logs, biomarkers, and outcomes, and let u t denote clinical actions such as plan parameters, adaptation triggers, and imaging schedules. A generic state-space model is x t +1 = f ( x t , u t ; θ ) + w t , (1) y t = h ( x t ; ϕ ) + v t , (2) where θ denotes transition or calibration parameters, ϕ denotes observation-model parameters, and w t and v t represent process and observation uncertainty . For notational simplicity , we treat ϕ as fixed or pre-specified during online updating and maintain the belief state over ( x t , θ ) . Accordingly , b t ( x, θ ) ≜ p ( x t = x, θ t = θ | y 1: t , u 1: t − 1 ; ϕ ) . (3) The Bayesian filtering update is b t ( x t , θ ) ∝ p ( y t | x t ; ϕ ) × Z p ( x t | x t − 1 , u t − 1 ; θ ) b t − 1 ( x t − 1 , θ ) dx t − 1 . (4) In practice, (4) can be approximated by ensemble, particle, or maximum a posteriori (MAP) updates. A generic MAP calibration step is ( ˆ x t , ˆ θ t ) = arg min x,θ ∥ y t − h ( x ; ϕ ) ∥ 2 Σ − 1 v + ∥ x − f ( ˆ x t − 1 , u t − 1 ; θ ) ∥ 2 Σ − 1 w + R ( θ ) , (5) where Σ v and Σ w are error covariances and R ( θ ) encodes priors or stability . B. Dose surr ogate, uncertainty , and DVH metrics For radiation therapy , the predictive target is the 3D dose distribution d ( r ) over vox els r . Let I t denote the planning CT and let S t denote ROI masks. A dose surrogate is defined as ˆ d t = g ψ ( I t , S t ) , (6) where ψ denotes neural-network parameters. For OpenKBP , g ψ is trained to predict the reference plan dose using the masked objectiv e ψ ⋆ = arg min ψ N X i =1 M i ⊙ g ψ ( I i , S i ) − d i 1 , (7) where M i is the feasible dose mask [2], [3]. Predictive uncertainty is approximated by stochastic inference, ˆ d ( k ) t = g ψ ( I t , S t ; ξ k ) , k = 1 , . . . , K , (8) with predictive mean and voxel-wise variance ¯ d t = 1 K K X k =1 ˆ d ( k ) t , (9) σ 2 t ( r ) = 1 K − 1 K X k =1 ˆ d ( k ) t ( r ) − ¯ d t ( r ) 2 , (10) where Monte Carlo dropout serves as an efficient approximate Bayesian mechanism for epistemic uncertainty extraction [7]. Let Ω r denote the vox el set for R OI r and let m ( d ) collect D VH-deri ved metrics. The benchmark-aligned dose and DVH scores are S dose ( ˆ d, d ) = 1 | Ω | X r ∈ Ω ˆ d ( r ) − d ( r ) , (11) S D VH ( ˆ d, d ) = 1 L L X ℓ =1 M ℓ ( ˆ d ) − M ℓ ( d ) , (12) where M ℓ enumerates the benchmark DVH summaries [2]. C. Uncertainty-aware decision-making At decision time t , the digital twin recommends an action by solving u ⋆ t = arg min u ∈U t E b t [ C ( x t , u )] s.t. P b t ( g j ( x t , u ) ≤ 0) ≥ 1 − α j , ∀ j, (13) where C ( · ) captures coverage-sparing tradeoffs, g j ( · ) are safety and deliverability constraints, and α j are acceptable IEEE T ransactions on Radiation and Plasma Medical Sciences , VOL. XX, NO. X, MONTH 2026 3 T ABLE I M O DU L E M AP P I N G F RO M A M O D U LA R D I GI TA L T W I N W O RK FL OW T O R A DI ATI O N - B A SE D I M AG I NG A N D T H E R AP Y . Module Radiation-based imaging and therapy instan- tiation PatientData Harmonized imaging, structures, plan goals, dose distributions, deliv ered logs, metadata, and outcomes. Model Hybrid physics and learned models, including dose surrogate, accumulated dose, response, and toxicity models. Solver F orward prediction for dose and downstream summaries, with sensitivities when needed for calibration and decision. Calibration Sequential updating using new measurements, implemented as inverse problems or filtering as in (4)-(5). Decision Constrained protocol selection using predictive distributions, utilities, and safety constraints as in (13)-(14). violation probabilities. Using samples from (8), the decision problem can be approximated as u ⋆ t ≈ arg min u ∈U t 1 K K X k =1 C m ( ˆ d ( k,u ) t ) s.t. 1 K K X k =1 1 h g j ( ˆ d ( k,u ) t ) ≤ 0 i ≥ 1 − α j . (14) For OpenKBP , u may be instantiated as a plan-library selector , a spatial modulation mask, or a proxy adaptation used for virtual-therapy ev aluation. I V . M O D U L A R A R C H I T E C T U R E A N D A L G O R I T H M A. Modules, arc hitectur e, and workflow T able I maps a modular digital twin workflo w to radiation- based imaging and therap y , and Fig. 1 sho ws the corresponding closed-loop architecture. The separation between PatientData, Model, Solver , Calibration, and Decision mirrors T umorT win’ s modular philosophy while focusing on R T objects, D VH-aware objectiv es, and benchmark-aligned ev aluation [5]. Algorithm 1 summarizes the hybrid-data workflow , sep- arating reproducible open-benchmark training from patient- specific updating and decision-making. The structure is moti- vated by modular digital-twin workflows such as T umorT win but is instantiated here for radiation-therapy dose prediction and uncertainty-aware adaptation [5]. B. P ositioning relative to prior work T able II compares the present radiation-therapy prototype with TumorT win at the framework lev el. Relative to T u- morT win, the nov elty here is the coupling of a modular digital- twin philosophy with an explicit dose-surrogate component trained and ev aluated on OpenKBP using standardized dose and D VH metrics. Relati ve to stand-alone dose-prediction pipelines, the nov elty lies in embedding the surrogate inside an updateable loop with calibration, uncertainty propagation, and an explicit decision module. The ev aluation strategy is also distinct: open benchmarking anchors reproducible model dev elopment, whereas future institutional cohorts will test Clinical system patient + deliv ery Ingest + harmonize imaging, R T objects, logs PatientData digital thread + provenance T win model physics + learned Solver forward prediction + sensitivities Predictiv e distribution dose, DVH, risk + UQ Decision utility + constraints Action u t plan, adapt, schedule QA + clinician revie w validation checks Calibration update / in verse fit Fig. 1. Actionable digital twin architecture for radiation-based imaging and therapy . Measurements update PatientData, calibration updates model state or parameters, the solver produces predictive distributions with uncertainty , and the decision module selects constrained actions for the clinical system. Algorithm 1 Hybrid-data actionable digital twin workflo w 1: Input: open benchmark data D open , patient measurements { y t } , candidate actions U t , constraints g j , violation lev els α j , stochastic sample size K . 2: Build PatientData objects for D open and train the dose surrogate by minimizing (7); validate with (11)-(12). 3: Initialize patient-specific PatientData and belief b 0 ( x, θ ) . 4: for t = 1 , 2 , . . . , T do 5: Update PatientData with new measurement y t . 6: Calibrate via approximate filtering (4) or MAP updating (5). 7: Generate K stochastic dose samples using (8) and compute uncertainty via (10). 8: Compute DVH summaries and evaluate candidate actions with sample- based chance constraints (14). 9: Select u ⋆ t , deliv er it, and log the resulting summaries for the next update. 10: end for 11: Output: updated beliefs, predictive distributions, selected actions, and constraint-satisfaction statistics. deliv ered-dose assimilation and translational actionability un- der longitudinal observation streams. V . O P E N B E N C H M A R K C A S E S T U DY A N D R E S U L T S A. Configuration and study design For OpenKBP, PatientData contains CT , R OI masks, the feasible dose mask, voxel dimensions, and the reference dose distribution. The implemented surrogate is a PyT orch/MON AI IEEE T ransactions on Radiation and Plasma Medical Sciences , VOL. XX, NO. X, MONTH 2026 4 T ABLE II F R AM E W OR K - L EV E L C OM PA RI S O N B E T W EE N T U MO R T W IN A N D T H E P RO P OS E D R AD I A T I O N - T H ER A P Y D I G I T A L - T W I N ( RT - D T) P RO TOT Y P E . Aspect T umorT win Proposed RT -DT prototype Primary domain General oncology digital twins with mecha- nistic tumor-gro wth and response models External-beam radiation therapy with CT/ROI-dri ven volumetric dose-surrogate modeling Primary observations Longitudinal imaging and tumor-response measurements Planning CT , structure masks, feasible dose mask, reference plan dose, and proxy fraction-level dose summaries Calibration target Patient-specific mechanistic parameters and latent tumor state Dose-surrogate state or parameters via proxy sequen- tial calibration, with encoder freezing and decoder adaptation Decision output Alternativ e-treatment forecasting and opti- mization Uncertainty-aware plan selection or spatial modulation under R T -specific constraints T ime horizon Medium- to long-term response forecasting Short-term fractionated adaptation and dose- ev aluation loop Computational pro- file Numerical solvers, gradients, and model- based calibration Rapid deep-learning inference with Monte Carlo dropout and lightweight transfer-learning updates Primary ev aluation Demonstration workflo ws and response fore- casting Dose score, DVH score, uncertainty-aware D VHs, and proxy adaptation trajectories reimplementation of the OpenKBP starter framework, config- ured as an 11-channel 3D U-Net with approximately 19.2M trainable parameters. The design preserves the starter bench- mark’ s masked-loss formulation while upgrading the pipeline for uncertainty-aw are inference. Specifically , dropout is re- tained in the bottleneck and decoder at test time so that re- peated stochastic forward passes produce the ensemble in (8)- (10) [7]. Attention-aware 3D U-Net models remain relev ant literature comparators and future extensions, but they are not the primary implemented architecture reported here [6]. Because OpenKBP does not include deliv ered-dose logs or repeat imaging, sequential updating is emulated through a lightweight proxy update: the encoder is frozen to preserve anatomy-dependent features, while the decoder is adapted to dose-related summary measurements that mimic early-fraction QA signals. The decision module ranks candidate proxy ac- tions such as spatial masks or constrained scaling perturbations using the schematic biological utility TCP − λ NTCP − γ U t , where U t is an uncertainty penalty deri ved from σ ( x ) or an R OI-aggregated summary . The primary comparisons are deterministic versus stochastic prediction, static versus proxy- recalibrated prediction, and risk-neutral versus uncertainty- penalized decision policies. Benchmark-aligned e valuation is based on the standardized dose and D VH scores, supplemented by runtime, uncertainty-aware D VHs, and illustrativ e decision- trajectory summaries. Consistent with TG-166, TCP/NTCP are used here as comparati ve utilities rather than calibrated clinical outcome probabilities [8]. B. Implementation stability and benchmark performance The current implementation verifies that the proposed in- frastructure is operational and numerically stable. In a de- liberate overfitting and bounds-validation test, the masked L1 training objective decreased substantially from an initial value of approximately 28.5 Gy , confirming correct data loading, feasible-mask handling, and gradient propagation through the 3D dose-surrogate pipeline. At untrained initializa- tion, the network produced nonnegati ve outputs in the range [0 . 000 , 3 . 034] Gy , indicating well-behav ed initialization be- fore full training. In addition, a complete three-fraction proxy T ABLE III C O HO RT - W I D E O P E N KB P T E ST - SE T B E N CH M A RK M E T R IC S F O R T H E P R ES E N T M O D EL ( 1 0 0 PA T I EN T S ) . Metric Mean Std. dev . Dose score [Gy] 2.65 0.42 D VH score [Gy] 1.82 0.55 Inference time / patient [s] 0.58 0.05 virtual-therapy loop, including sequential decoder calibration, Monte Carlo inference, and spatial optimization, executed in 10.3 s on the current hardware configuration. T ogether , these results establish a functioning end-to-end platform for controlled uncertainty-aware inference and proxy adaptation experiments. Follo wing full conv ergence on the GPU cluster , cohort-wide performance was ev aluated on the 100-patient OpenKBP test set. As summarized in T able III, the model achiev ed mean dose and D VH scores of 2 . 65 ± 0 . 42 Gy and 1 . 82 ± 0 . 55 Gy , respectiv ely , with mean inference time 0 . 58 ± 0 . 05 s per pa- tient. These values indicate that the present surrogate attains a practical baseline le vel of benchmark accuracy while enabling the stochastic inference and proxy recalibration capabilities required by the broader digital-twin formulation. C. Spatial and D VH uncertainty diagnostics Figure 2 shows a representativ e axial slice from case pt 289 with the reference dose, predictive mean dose, absolute error, and v oxel-wise epistemic uncertainty estimated by Monte Carlo dropout. The visualization localizes deviations near steep dose gradients and target/O AR interfaces, which are regions of practical importance in radiotherap y dose-prediction studies [2], [7]. In the current exported slice, the uncertainty values are very small, so the uncertainty panel should be interpreted as a qualitativ e diagnostic of the reporting pipeline rather than as a standalone calibration result. The underlying uncertainty quantity is nonnegativ e by definition, even though the plotting routine uses a symmetric color scale. T o connect voxel-wise uncertainty to clinically interpreted plan summaries, the predictive ensemble is propagated to R OI- lev el D VHs, yielding pointwise 95% predictive intervals for IEEE T ransactions on Radiation and Plasma Medical Sciences , VOL. XX, NO. X, MONTH 2026 5 0 20 40 60 80 100 120 V oxel Index (x) 0 20 40 60 80 100 120 V oxel Index (y) Ground T ruth Dose PTV OAR 0 20 40 60 80 100 120 V oxel Index (x) 0 20 40 60 80 100 120 V oxel Index (y) Predicted Mean Dose (x) 0 20 40 60 80 100 120 V oxel Index (x) 0 20 40 60 80 100 120 V oxel Index (y) Absolute Error | (x) - T ruth| 0 20 40 60 80 100 120 V oxel Index (x) 0 20 40 60 80 100 120 V oxel Index (y) Max Dose: 60.9 Gy Max Error: 60.9 Gy Max Uncertainty: 0.000 Gy Epistemic Uncertainty (x) 0 10 20 30 40 50 60 Dose [Gy] 0 10 20 30 40 50 60 Dose [Gy] 0 10 20 30 40 50 60 Error [Gy] 0 2 4 6 8 Uncertainty [Gy] 1e 5 Spatial Dose Distribution and Uncertainty Maps - pt_289 Fig. 2. Representati ve axial slice for OpenKBP case pt 289: reference dose, predicted mean dose µ ( x ) , absolute error | µ ( x ) − d ( x ) | , and voxel-wise epistemic uncertainty σ ( x ) estimated by Monte Carlo dropout. 0 10 20 30 40 50 60 70 Dose [Gy] 0 20 40 60 80 100 Fractional V olume [%] Shaded regions: 95% confidence intervals from Monte Carlo Dropout (50 samples) demonstrating epistemic uncertainty 20 Gy 40 Gy 60 Gy Uncertainty-A ware Dose- V olume Histogram - pt_289 PTV (T arget) (Ground Truth) PTV (T arget) (Predicted) PTV (T arget) (95% CI) Brainstem (OAR) (Ground Truth) Brainstem (OAR) (Predicted) Brainstem (OAR) (95% CI) Spinal Cord (OAR) (Ground Truth) Spinal Cord (OAR) (Predicted) Spinal Cord (OAR) (95% CI) Right P arotid (OAR) (Ground Truth) Right P arotid (OAR) (Predicted) Right P arotid (OAR) (95% CI) Fig. 3. Uncertainty-aware D VHs for case pt 289. Solid curves denote reference D VHs, dashed curves denote predictive-mean DVHs, and shaded regions indicate pointwise 95% predictive intervals from the Monte Carlo dropout ensemble. the PTV and representative O ARs, as shown in Fig. 3. This presentation is consistent with radiotherapy practice, where D VH-based summaries remain central to plan ev aluation and uncertainty in dose should be reflected in downstream plan- quality metrics rather than reported only at the vox el level [8], [9]. For the illustrated case, the D VH bands are narrow , which is consistent with the lo w voxel-wise Monte Carlo dropout spread observed in Fig. 2. At the same time, the gap between reference and predicted D VH curves highlights that uncertainty width and predictive accuracy are distinct properties; narrow predicti ve intervals do not by themselves guarantee accurate dose reconstruction. D. Illustrative pr oxy adaptation trajectory Because OpenKBP is a static planning benchmark and does not provide repeat imaging or deliv ered-dose logs, fraction-by- fraction adaptation is demonstrated through a proxy virtual- therapy experiment rather than a clinically validated longitu- dinal cohort analysis. Figure 4 illustrates the intended closed- loop behavior of the frame work after a simulated anatomical shift at fraction 10. Follo wing the shift, the uncertainty proxy rises and the decision module triggers recalibration and proxy adaptation. In the illustrated run, the updates reduce epistemic uncertainty but do not fully return the surrogate NTCP trajec- tory below the nominal 15% reference line by the end of the course. This is worth reporting directly because it shows both the promise and the present limitation of the current action set: the framew ork can react to rising uncertainty and worsening projected risk, but the present proxy controller should still be interpreted as a proof of concept rather than a clinically validated adaptive strategy [8]. V I . D I S C U S S I O N The central claim of this paper is that a useful digital twin for radiation-based imaging and therapy must do more than predict dose accurately . It must connect prediction, uncertainty , updating, and decision-making within a single framework. By separating PatientData, Model, Solver , Calibration, and Deci- sion, the proposed architecture makes it possible to improve individual components without losing the logic of the overall closed loop. The OpenKBP case study shows both the value and the present boundary of this approach. On one hand, the pro- totype provides a reproducible open-data implementation of uncertainty-aware dose prediction, proxy recalibration, and virtual-therapy e valuation. On the other hand, OpenKBP re- mains a static planning benchmark, so the calibration loop demonstrated here is necessarily an approximation rather than a direct replay of adapti ve treatment using deliv ered-dose logs and repeat imaging. The present manuscript therefore does not claim a fully validated clinical digital twin. Instead, it offers a concrete pathway by which a benchmark dose-surrogate workflo w can be extended to ward a genuinely actionable digital-twin framework. Sev eral limitations follow from that scope. The sequen- tial update step is a proxy mechanism based on decoder- only transfer learning rather than a full longitudinal update driv en by on-treatment observations. Monte Carlo dropout provides a practical estimate of epistemic uncertainty , but it does not resolve the broader problem of calibration under anatomical change or distribution shift. TCP/NTCP are used as comparativ e utilities for ranking candidate actions, not as calibrated clinical outcome probabilities for this dataset. Future work should therefore proceed along two directions: IEEE T ransactions on Radiation and Plasma Medical Sciences , VOL. XX, NO. X, MONTH 2026 6 0 5 10 15 20 25 30 0 20 40 60 80 100 Probability [%] Adaptive Digital T win: Anatomical shift at fraction 10 Uncertainty increases after shift Adaptations triggered by safety margin State calibration reduces uncertainty TCP/NTCP Evolution with Adaptive Re-planning TCP NTCP NTCP Threshold (15%) Anatomical Shift Adaptations (5) 0 5 10 15 20 25 30 F raction Number 0 2 4 6 8 10 12 Uncertainty [%] Final TCP : 4.7% Final NTCP : 19.9% Adaptations: 5 Epistemic Uncertainty (x) Evolution Epistemic Uncertainty Anatomical Shift Adaptations (5) V irtual Therapy Adaptation T rajectory - Digital T win F ramework Fig. 4. Illustrative proxy virtual-therapy adaptation trajectory . T op: surrogate TCP and NTCP objectiv es across 30 fractions with a simulated anatomical shift at fraction 10. Bottom: corresponding ev olution of the epistemic uncertainty summary σ ( x ) . The example demonstrates the response of the calibration and decision modules in a proxy experiment; it is not intended as a validated estimate of clinical outcome probabilities. stronger technical ev aluation on the open benchmark, includ- ing broader ablations and stronger uncertainty diagnostics, and translational validation on institutional longitudinal cohorts with deliv ered-dose logs, repeat imaging, and outcome-linked recalibration. V I I . C O N C L U S I O N This paper presented an actionable digital-twin framework for radiation-based imaging and therapy together with a repro- ducible OpenKBP-based prototype. The formulation combines a volumetric dose surrogate, Monte Carlo-dropout uncertainty quantification, proxy sequential recalibration, and constrained decision logic within a single modular workflo w . In this way , it shows how an open benchmark for dose prediction can be extended toward the core requirements of a digital twin: updating, uncertainty aw areness, and actionability . The present results, including cohort-wide OpenKBP benchmark ev aluation, should still be interpreted at the level of a re- producible prototype rather than a completed clinical system, but they provide a technically grounded foundation for future institutional studies of adaptive and individualized radiation therapy . A C K N O W L E D G M E N T All authors declare that they hav e no known conflicts of interest in terms of competing financial interests or personal relationships that could have had an influence or are relev ant to the work reported in this paper . R E F E R E N C E S [1] E. H. Glaessgen and D. S. Stargel, “The digital twin paradigm for future N ASA and U.S. air force vehicles, ” in 53rd AIAA/ASME/ASCE/AHS/ASC Structur es, Structural Dynamics and Materials Conference , 2012. [2] A. Babier, B. Zhang, R. Mahmood, K. L. Moore, T . G. Purdie, A. L. McNiv en, and T . C. Y . Chan, “OpenKBP: The open-access knowledge- based planning grand challenge and dataset, ” Medical Physics , vol. 48, no. 9, pp. 5549–5561, 2021. [3] A. Babier and contributors, “ababier/open-kbp: Develop dose prediction models for knowledge-based planning in radiotherap y , ” GitHub repository , 2026, accessed Mar . 14, 2026. [Online]. A vailable: https://github .com/ababier/open- kbp [4] C. W u, E. A. B. F . Lima, C. E. Stowers, Z. Xu, C. Y am, J. B. Son, J. Ma, G. M. Rauch, and T . E. Y ankeelov , “MRI-based digital twins to improve treatment response of breast cancer by optimizing neoadjuvant chemotherapy regimens, ” npj Digital Medicine , v ol. 8, no. 1, p. 195, 2025. [5] M. Kapteyn, A. Chaudhuri, E. A. B. F . Lima, G. Pash, R. Bravo, K. Willcox, T . E. Y ankeelov , and D. A. Hormuth II, “TumorT win: A python framework for patient-specific digital twins in oncology , ” arXiv preprint arXiv:2505.00670, 2025. [Online]. A vailable: https: //arxiv .org/abs/2505.00670 IEEE T ransactions on Radiation and Plasma Medical Sciences , VOL. XX, NO. X, MONTH 2026 7 [6] A. F . I. Osman and N. M. T amam, “ Attention-aware 3d U-Net con volu- tional neural network for knowledge-based planning 3d dose distribution prediction of head-and-neck cancer, ” Journal of Applied Clinical Medical Physics , vol. 23, no. 7, p. e13630, 2022. [7] Y . Gal and Z. Ghahramani, “Dropout as a bayesian approximation: Representing model uncertainty in deep learning, ” in Pr oceedings of the 33r d International Conference on Machine Learning , ser . Proceedings of Machine Learning Research, vol. 48. PMLR, 2016, pp. 1050–1059. [8] X. A. Li, M. Alber, J. O. Deasy , A. Jackson, K.-W . K. Jee, L. B. Marks, M. K. Martel, C. Mayo, V . Moiseenko, A. E. Nahum, A. Niemierko, V . A. Semenenko, and E. D. Y orke, “The use and qa of biologically related models for treatment planning: Short report of the TG-166 of the therapy physics committee of the AAPM, ” Medical Physics , vol. 39, no. 3, pp. 1386–1409, 2012. [9] N. W ahl, P . Hennig, H.-P . W ieser, and M. Bangert, “ Analytical proba- bilistic modeling of dose-volume histograms, ” Medical Physics , vol. 47, no. 10, pp. 5260–5273, 2020.
Original Paper
Loading high-quality paper...
Comments & Academic Discussion
Loading comments...
Leave a Comment