The microscopic origin of droplet line tension
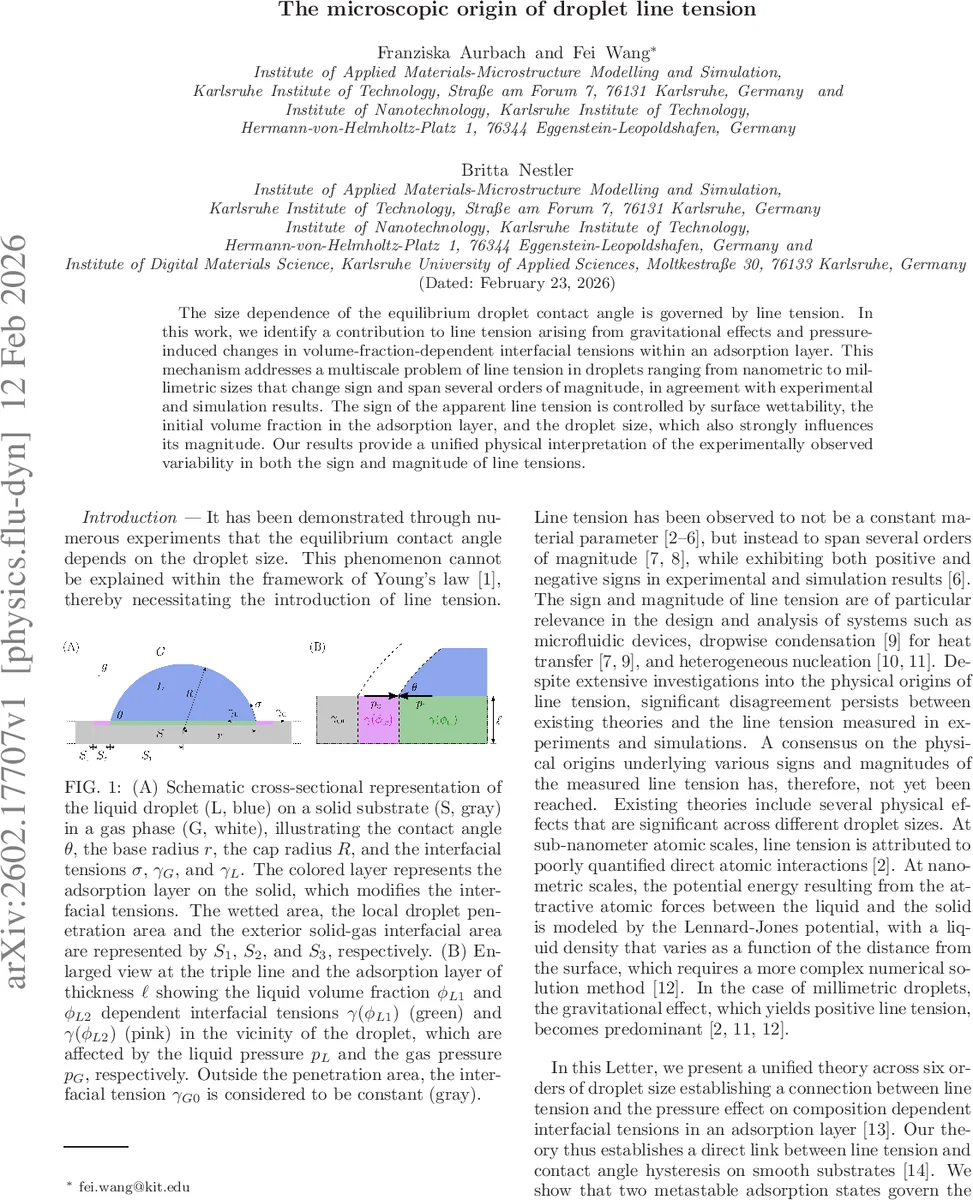
The size dependence of the equilibrium droplet contact angle is governed by line tension. In this work, we identify a contribution to line tension arising from gravitational effects and pressure-induced changes in volume-fraction-dependent interfacial tensions within an adsorption layer. This mechanism addresses a multiscale problem of line tension in droplets ranging from nanometric to millimetric sizes that change sign and span several orders of magnitude, in agreement with experimental and simulation results. The sign of the apparent line tension is controlled by surface wettability, the initial volume fraction in the adsorption layer, and the droplet size, which also strongly influences its magnitude. Our results provide a unified physical interpretation of the experimentally observed variability in both the sign and magnitude of line tensions.
💡 Research Summary
The manuscript “The microscopic origin of droplet line tension” tackles a long‑standing puzzle in wetting physics: why the apparent contact angle of a sessile droplet depends on its size and why measured line tensions span several orders of magnitude, sometimes changing sign. Classical Young’s law predicts a size‑independent contact angle, and traditional line‑tension theories attribute size effects either to atomic‑scale forces, Lennard‑Jones interactions, or macroscopic gravity. However, experimental data across nanometer to millimeter scales show a far richer behavior that cannot be reconciled with any single mechanism.
The authors propose a unified framework that links line tension to two distinct physical contributions: (i) a pressure‑induced modification of interfacial tensions within a thin adsorption layer that coats the solid substrate, and (ii) the familiar gravitational contribution for larger droplets. The adsorption layer, of thickness ℓ≈5 nm, contains a mixture of liquid and gas molecules whose local volume fractions (φ_L, φ_G) are altered by the Laplace pressure difference p_L − p_G = 2σ/R. By modeling the interfacial tension γ(φ_L,φ_G) as the sum of internal energy, pressure energy, and van‑der‑Waals terms, the authors derive an explicit size‑dependent correction:
Δγ = Δγ₀ + 2ℓ Δφ σ/R,
where Δφ = φ_L2 − φ_L1 is the difference in liquid fraction between the wetted area (S₁) and the penetration area (S₂) of the adsorption layer. The first term Δγ₀ is independent of droplet size, while the second term scales with 1/R and embodies the pressure‑induced line‑tension component.
The total free energy includes the cap surface energy, the modified interfacial term, and the gravitational energy E_g = Δρ g V z(θ). Using a spherical‑cap geometry and enforcing the volume constraint, the authors obtain a modified Young equation (Eq. 11) that contains both the pressure‑induced term (∝ ℓΔφ/R) and the gravitational term (∝ Δρ g R⁻²). Minimizing the free energy yields an apparent contact angle θ that deviates from the Young angle θ₀, and the line tension follows from τ = −σ d(cosθ)/d(1/r).
A crucial insight is the existence of two metastable adsorption‑layer configurations:
- Micro‑Cassie‑Baxter (CB) state – the liquid does not penetrate the adsorption layer (φ_L2 = 1, φ_L1 = 0), giving Δφ < 0.
- Micro‑Wenzel (W) state – the gas does not penetrate (φ_L2 = 0, φ_L1 = 1), giving Δφ > 0.
These states produce pressure‑induced line tensions of opposite sign (negative for CB, positive for W). Because gravity always contributes positively, the total line tension can change sign as droplet size varies. For nanometric and micrometric droplets (r < 3 × 10⁻⁵ m) the pressure term dominates, leading to τ ≈ ±10⁻¹⁰ N depending on the metastable state. For larger droplets (r > 10⁻⁴ m) the gravitational term grows as r² and overwhelms the pressure contribution, yielding a universally positive τ.
The theory is validated against a broad set of experimental and simulation data (e.g., Zhao 2019, Mugele 2002, Zhang 2008, Werder 2003). Figure 2 shows that the predicted τ(r) curves for both CB and W states align closely with measured values spanning from piconewtons to micronewtons. Figure 3 separates the pressure and gravity contributions, illustrating the crossover regime where the sign reversal occurs for the W state. Figure 4 demonstrates that, for droplets in the 16–116 nm radius range, the apparent line tension varies linearly with cosθ versus 1/r, and the sign change occurs near a contact angle of ≈ 66°, in agreement with experimental observations (Zhao et al., 2016).
By incorporating the pressure‑dependent interfacial tension of the adsorption layer, the authors resolve the multiscale discrepancy that plagued earlier models. The framework explains why line tension is not a material constant but a function of wettability (θ₀), initial adsorption‑layer composition, and droplet size. This unified picture has immediate implications for microfluidic design, dropwise condensation heat transfer, and heterogeneous nucleation, where precise control of contact angles and line tensions is essential.
In summary, the paper delivers a comprehensive, physically transparent model that unifies nanoscale pressure effects and macroscopic gravity into a single expression for line tension, successfully reproducing the observed magnitude, sign, and size dependence across six orders of magnitude in droplet size.
Comments & Academic Discussion
Loading comments...
Leave a Comment