GR-Diffusion: 3D Gaussian Representation Meets Diffusion in Whole-Body PET Reconstruction
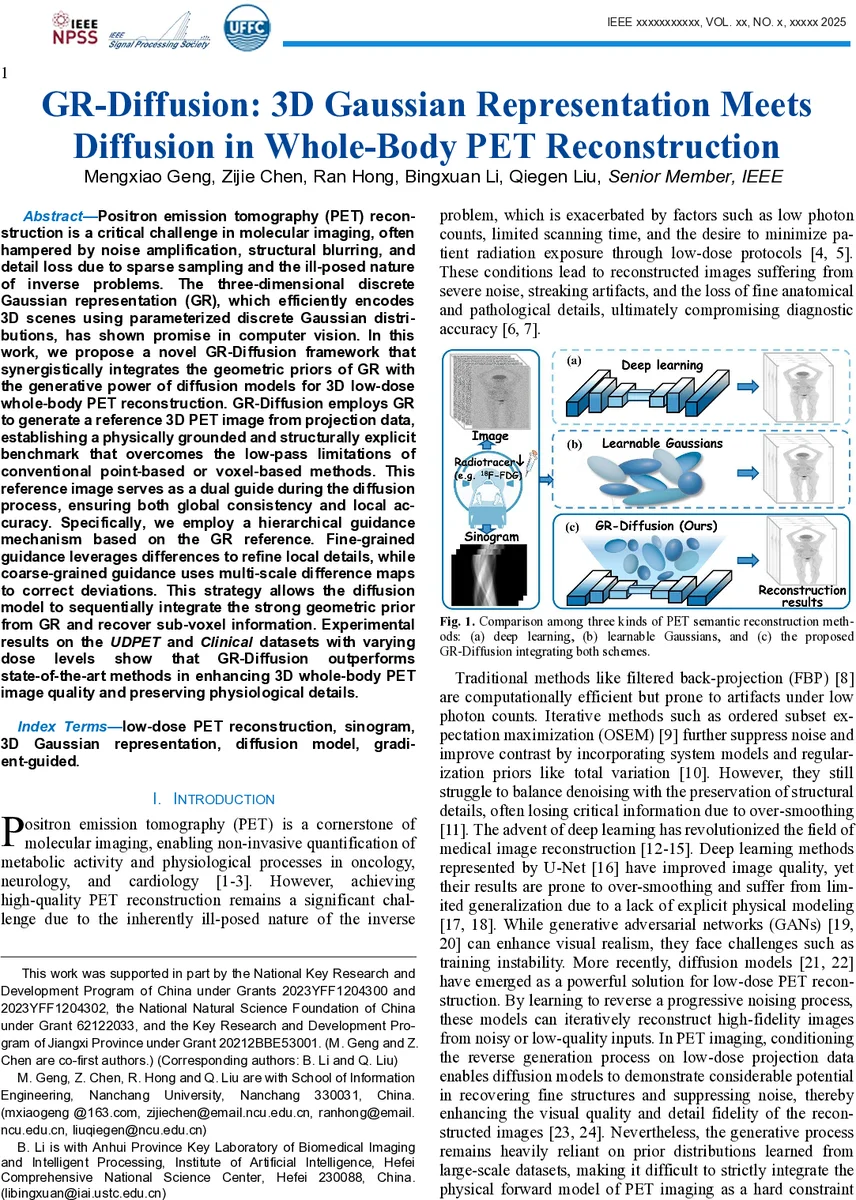
Positron emission tomography (PET) reconstruction is a critical challenge in molecular imaging, often hampered by noise amplification, structural blurring, and detail loss due to sparse sampling and the ill-posed nature of inverse problems. The three-dimensional discrete Gaussian representation (GR), which efficiently encodes 3D scenes using parameterized discrete Gaussian distributions, has shown promise in computer vision. In this work, we pro-pose a novel GR-Diffusion framework that synergistically integrates the geometric priors of GR with the generative power of diffusion models for 3D low-dose whole-body PET reconstruction. GR-Diffusion employs GR to generate a reference 3D PET image from projection data, establishing a physically grounded and structurally explicit benchmark that overcomes the low-pass limitations of conventional point-based or voxel-based methods. This reference image serves as a dual guide during the diffusion process, ensuring both global consistency and local accuracy. Specifically, we employ a hierarchical guidance mechanism based on the GR reference. Fine-grained guidance leverages differences to refine local details, while coarse-grained guidance uses multi-scale difference maps to correct deviations. This strategy allows the diffusion model to sequentially integrate the strong geometric prior from GR and recover sub-voxel information. Experimental results on the UDPET and Clinical datasets with varying dose levels show that GR-Diffusion outperforms state-of-the-art methods in enhancing 3D whole-body PET image quality and preserving physiological details.
💡 Research Summary
The paper introduces GR‑Diffusion, a novel framework that combines a three‑dimensional discrete Gaussian Representation (GR) with a diffusion‑based generative model to reconstruct low‑dose whole‑body PET images. Traditional PET reconstruction methods (FBP, OSEM) suffer from noise amplification and blurring, while deep‑learning approaches (U‑Net, GANs) lack explicit physical modeling and can over‑smooth anatomical details. Recent diffusion models have shown promise but rely heavily on data‑driven priors and do not enforce the PET forward model as a hard constraint.
GR Module
The authors first encode the activity distribution λ as a linear combination of isotropic 3‑D Gaussians (G_i(\mu_i,\sigma_i,I_i)). Each Gaussian is parameterized by its center, standard deviation, and intensity. A “discretize‑and‑align” procedure splits continuous centers into integer voxel coordinates and fractional offsets, enabling differentiable rendering on a voxel grid while limiting each Gaussian’s influence to a local 1×1×1 voxel region. Fast parallel volume reconstruction is achieved by decomposing the Mahalanobis distance into pre‑computed components, dramatically reducing computational overhead. The reconstructed volume is forward‑projected using the system matrix (P) to obtain a synthetic sinogram (\hat y). Optimization minimizes a loss that combines a Poisson‑likelihood term (or KL approximation) with total‑variation regularization, yielding updated Gaussian parameters. An adaptive density control periodically prunes low‑impact Gaussians, clones those in high‑error regions, and splits oversized Gaussians, thereby dynamically adjusting the number of primitives and improving spatial resolution.
Gradient‑Guided Diffusion
The GR output (\hat\lambda) serves as a geometric prior for a pretrained 3‑D diffusion model (DDPM‑style UNet). During reverse diffusion, two hierarchical guidance signals are computed:
- Fine‑grained guidance – pixel‑wise difference (\Delta_{\text{fine}} = x_t - \hat\lambda) that drives local detail recovery.
- Coarse‑grained guidance – multi‑scale difference maps (\Delta_{\text{coarse}}) that enforce global consistency and correct large‑scale deviations.
Both are incorporated into the diffusion sampling step as gradient‑based losses:
\
Comments & Academic Discussion
Loading comments...
Leave a Comment