An Anatomy-specific Guidewire Shaping Robot for Improved Vascular Navigation
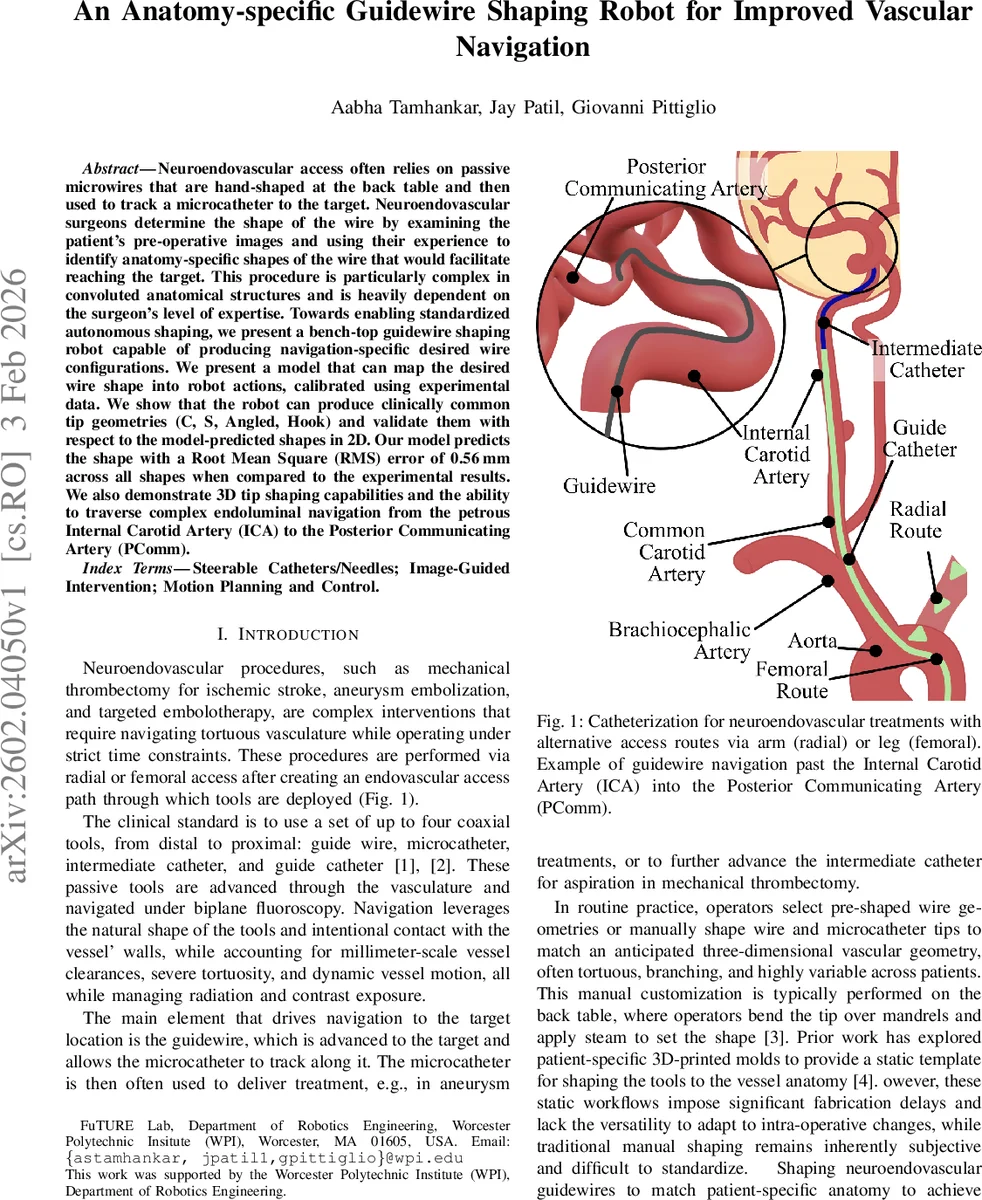
Neuroendovascular access often relies on passive microwires that are hand-shaped at the back table and then used to track a microcatheter to the target. Neuroendovascular surgeons determine the shape of the wire by examining the patient pre-operative images and using their experience to identify anatomy specific shapes of the wire that would facilitate reaching the target. This procedure is particularly complex in convoluted anatomical structures and is heavily dependent on the level of expertise of the surgeon. Towards enabling standardized autonomous shaping, we present a bench-top guidewire shaping robot capable of producing navigation-specific desired wire configurations. We present a model that can map the desired wire shape into robot actions, calibrated using experimental data. We show that the robot can produce clinically common tip geometries (C, S, Angled, Hook) and validate them with respect to the model-predicted shapes in 2D. Our model predicts the shape with a Root Mean Square (RMS) error of 0.56mm across all shapes when compared to the experimental results. We also demonstrate 3D tip shaping capabilities and the ability to traverse complex endoluminal navigation from the petrous Internal Carotid Artery (ICA) to the Posterior Communicating Artery (PComm).
💡 Research Summary
**
The paper addresses a critical bottleneck in neuro‑endovascular procedures: the manual shaping of guidewire tips to match patient‑specific vascular anatomy. Current practice relies on the surgeon’s experience to bend a shapeable microwire on the back table, a process that is time‑consuming, highly variable, and difficult to standardize, especially in tortuous cerebral vessels. To overcome these limitations, the authors present a bench‑top robotic system that autonomously shapes commercially available guidewires into clinically relevant tip geometries.
The robot consists of three serial modules aligned with the wire: (1) a Drive Nozzle that rotates and feeds the wire using a NEMA‑17 stepper motor coupled to a gear train and a micro‑servo clamp, (2) a Stabilizer that grips the shaft to prevent axial slip while allowing free bending of the distal segment, and (3) a Shaping Carriage mounted on a high‑precision linear stage (CBX‑1605) that pinches the wire between two custom jaws and translates a short distance to impose a localized bending moment. By sequencing three primitive actions—axial rotation (ϕk), controlled pinch angle (βk) that determines bend magnitude (θk), and axial advance (δk)—the robot executes a roll‑bend‑advance cycle for each segment, reproducing the surgeon’s “pinch‑and‑pull” technique.
A kinematic model links the robot’s command set {ϕk, βk, δk} to the resulting wire centerline p(s). The wire is discretized into n equal‑length segments, each approximated as a constant‑curvature arc. The model predicts the tip shape by composing rotations about the wire’s longitudinal axis (ϕk) and about the bending axis (θk). Because the exact relationship between pinch force and θk is highly nonlinear and material‑dependent, the authors adopt a data‑driven calibration. They shape a 10‑segment constant‑curvature arc with a fixed pinch force and zero rotation, measure the end‑to‑end chord length, and solve a closed‑form equation to extract a per‑segment bend angle θ*. This single calibrated parameter captures the wire’s response to the robot’s actuation and can be reused for any subsequent complex shape.
Experimental validation used an ARROW™ Marked Spring guidewire (0.64 mm diameter, 20 mm shapeable tip). Calibration across three trials yielded a mean chord length of 18.7 ± 0.21 mm, corresponding to θ* ≈ 6.89°. Using this value, the robot generated four standard 2‑D tip shapes—C, S, angled, and hook—by programming appropriate sequences of {ϕk, βk, δk}. Shape measurements showed an average root‑mean‑square deviation of 0.56 mm between predicted and actual tip contours, well within clinical tolerances.
Beyond 2‑D validation, the system produced multi‑planar 3‑D geometries such as helical tips and demonstrated successful navigation through a realistic neurovascular phantom, specifically from the petrous segment of the internal carotid artery (ICA) to the posterior communicating artery (PComm). This confirms that the robot can create shapes that satisfy both curvature and orientation requirements for complex endoluminal paths.
Key contributions include: (1) a fully autonomous shaping robot that works with off‑the‑shelf guidewires, preserving existing supply chains; (2) a compact kinematic framework that maps low‑dimensional robot commands to high‑dimensional wire shapes; (3) a simple yet effective calibration protocol that reduces the need for extensive training data; and (4) experimental proof that the generated shapes are accurate enough for realistic vascular navigation.
Limitations are acknowledged. All tests were performed on a bench‑top setup and static phantom models; dynamic factors such as blood flow, vessel compliance, and intra‑operative imaging feedback were not addressed. The robot’s precision depends on servo torque and mechanical compliance, which may drift over time, necessitating periodic recalibration. Future work should integrate real‑time fluoroscopic or ultrasound guidance, expand the calibration to a broader range of wire diameters and materials, and develop a seamless user interface for the interventional suite.
In summary, the study presents a practical, low‑cost robotic solution that automates guidewire tip shaping, potentially reducing operator dependence, improving reproducibility, and enhancing safety in neuro‑endovascular interventions.
Comments & Academic Discussion
Loading comments...
Leave a Comment