Physiological Closed-Loop Control (PCLC) Systems: Review of a Modern Frontier in Automation
Over the past decade, there has been an unprecedented international focus on improved quality and availability of medical care, which has reignited interest in clinical automation and drawn researchers toward novel solutions in the field of physiolog…
Authors: Mohammad Javad Khodaei, Nicholas C, elino
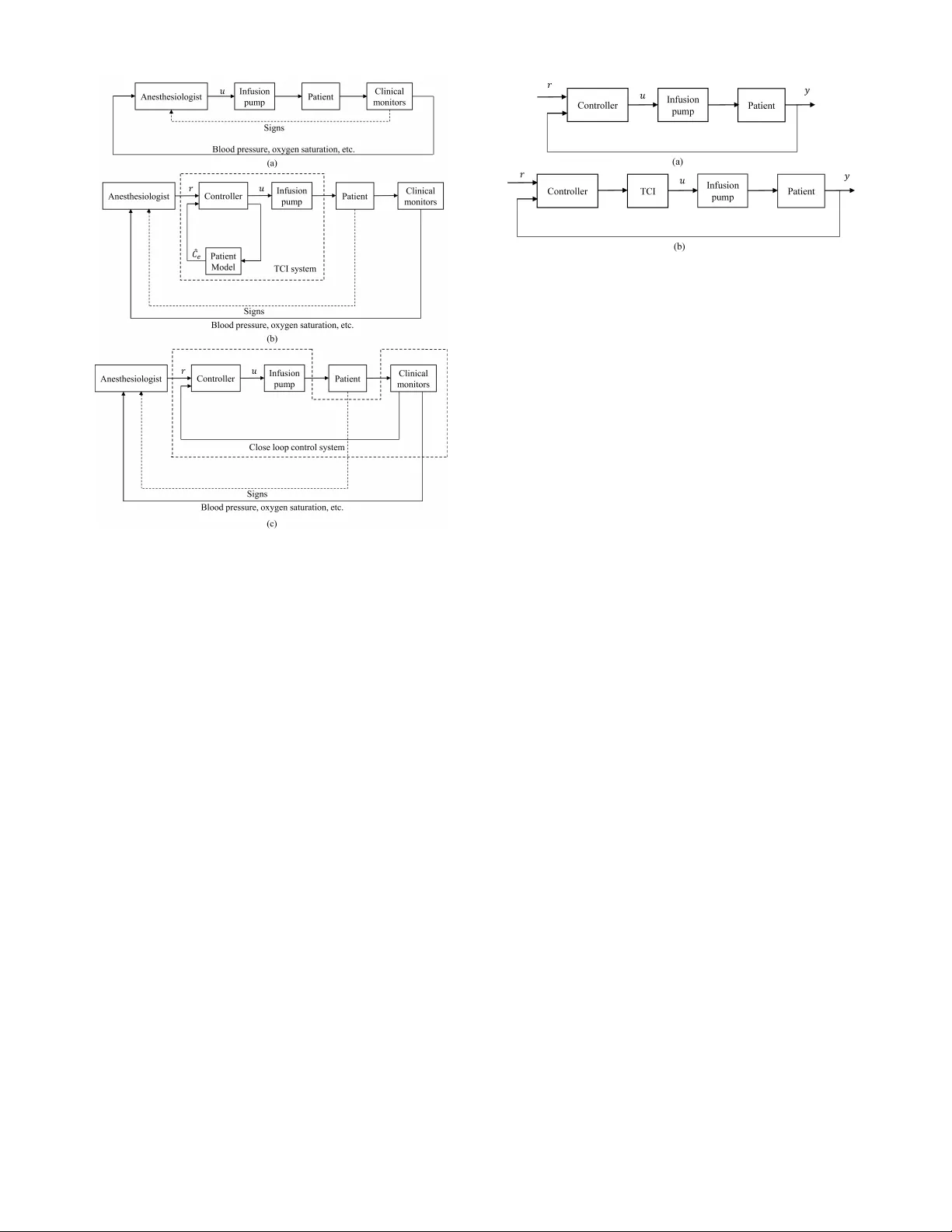
Physiological Closed-Loop Control (PCLC) Systems: Re vie w of a Modern Frontier in Automation Mohammad Jav ad Khodaei 1 , ∗ , Nicholas Candelino 1 , ∗ , Amin Mehrv arz 1 , and Nader Jalili 1 , 2 1 Department of Mechanical and Industrial Engineering, Northeastern Uni versity , Boston, MA, USA 2 Professor and Head, Department of Mechanical Engineering, Uni versity of Alabama, T uscaloosa, AL, USA ∗ Co-first author Abstract —Over the past decade, there has been an unprece- dented international focus on impr oved quality and availability of medical care, which has r eignited interest in clinical automation and drawn resear chers toward novel solutions in the field of physiological closed-loop contr ol systems (PCLCs). T oday , multidisciplinary groups of expert scientists, engineers, clini- cians, mathematicians, and policy-makers are combining their knowledge and experience to develop both the next generation of PCLC-based medical equipment and a collaborative commer - cial/academic infrastructure to support this rapidly expanding frontier . In the following article, we pro vide a robust introduction to the various aspects of this growing field motivated by the recent and ongoing work supporting two leading technologies: the artificial pancreas (AP) and automated anesthesia. Following a brief high-level overview of the main concepts in automated therapy and some rele vant tools from systems and control theory , we explore – separately – the developments, challenges, state-of- the-art, and probable directions f or AP and automated anesthesia systems. W e then close the review with a consideration of the common lessons gleaned from these ventures and the implications they present for future in vestigations and adjacent research. Index T erms —Adaptive contr ol, artificial intelligence, anes- thesia, artificial pancreas, automated anesthesia, automation, Bergman minimal model, BMM, blood glucose , closed-loop, control, cyberphysical systems, decision-making, diabetes, drug dosage, fuzzy-logic, fuzzy logic, F-L, general predictiv e con- trol, GPC, IDDM, in-silico, insulin, medical cyberphysical sys- tems, model predicti ve control, MPC, PCLC, pharmacody- namics, pharmacokinetics, ph ysiological systems, physiological closed-loop control, physiology , PK-PD, prediction, Propof ol, proportional-integral-deri vative, PID, r emifentanil, systems phar - macology , systems physiology , therapy , T ype 1 diabetes, T ype 2 diabetes, T1DM, T2DM I . I N T RO D U C T I O N The modern adv ance of automated and intelligent sys- tems tow ard more-and-more visible roles in society (e.g., commercially av ailable automated vehicles, humanoid and biomimetic robots, household v acuums and thermostats, etc.) has accelerated the dev elopment of many new technologies and restored interest in sev eral challenging fields. Principal among these recent works, are the broad ongoing ef forts to deploy collaborative human-machine systems, such as those designed to provide advanced support in medical diagnostics and therapy . In this re view , we focus on a special class of these systems that apply concepts from feedback control to attain automatic regulation of physiological variables – such as blood pressure, depth of anesthesia, blood glucose concentrations, and others (see examples in T able I ) – known as physiological closed-loop control systems (PCLCs). While the pioneering works toward many kinds of PCLCs were performed over a half-century ago, we are now only nascent in an era of suitable computational/technological av ailability and physiological/pharmaceutical scientific understanding to begin realizing these devices on a broad commercial scale. From the outset, it is important to understand that these devices do not and are not intended to replace e xpert and supporting clinical staff. In fact, automated devices rely on the knowledge of experts and simply put, promise to do a much better job at follo wing clinical instruction than present technol- ogy . By introducing sensors capable of continuous monitoring (i.e., generating measurements every few seconds or minutes depending on the treatment) these devices stand to recognize and eliminate unintentional variations in therapies, including missed doses and faults like sensor detachment or blocked fluid pathways. Furthermore, post analysis of the measure- ments obtained by these de vices and the treatment profiles the y generate can provide important insights into the effecti veness of treatment strategies (e.g., the clinical setpoint is maintained to no effect), possible heterogeneity in disease pathologies, and goals for future pharmaceuticals (e.g., short or longer acting doses). The de velopment and adoption of these new systems is currently providing research directions in control engineering, artificial intelligence, human-machine interactions, social sci- ence, medical practice, therapeutic technology , pharmaceutical dev elopment, and biological/physiological systems modeling. Further , since poor controller performance in physiological systems can lead to immediate adv erse, potentially irre versible, and even fatal physiological responses, PCLCs are a rapidly dev eloping field of safety critical control systems and addi- tional research on safe PCLC operation will be of particular value in the transition to common medical practice (esp. as networked and ambulatory systems are realized). As a primer for researchers in these and other disciplines, we ha ve sought in this revie w to introduce key information from various aspects of the ongoing research to provide both a background and up-to-date perception of the field for unfa- 𝐂 𝐥𝐢 𝐧𝐢 𝐜 𝐢 𝐚 𝐧 𝐚 𝐛 𝐏 𝐂 𝐋 𝐂 𝐃𝐞𝐯𝐢 𝐜𝐞 𝐏 𝐚 𝐭𝐢 𝐞 𝐧𝐭 T h e r a p y A ctu a to r ( s ) Re l e v a nt M e a s u r a b l e Sta te ( s ) T h e r a p y A d j u s tm e nt 𝐂 𝐥𝐢 𝐧𝐢 𝐜 𝐢 𝐚 𝐧 T r e a tm e nt T a r g e t ( s ) Cl i n i cal G u id e l i ne s & O b s e r v a ti o ns C l in ica l V a r ia b l e s ( e . g . , BI S ) T r e a tm e nt ( e . g . , d o s a g e ) T h e r a py Ac t u a t o r ( s ) T h e r a p y A d j u s tm e nt T r e a tm e nt ( e . g . , d o s a g e ) S e ns o r ( s ) Re l e v a nt M e a s u r a b l e S ta te ( s ) E l e ct r i cal S ig na l s C o nt r o l A l g o r ith m & S a fe ty P r o to co l S u p e r v is o r y O b s e r v a tio ns T r e a tm e nt T a r g e t ( s ) S e ns o r De v ice ( s ) S i g n a l P r o ce s s in g E l e ctr ica l S ig na l s C l in ica l V a r ia b l e s ( e . g . , B I S ) 𝐏 𝐚 𝐭𝐢 𝐞 𝐧𝐭 S ig na l P r o ce s s ing Fig. 1. Comparison of (a) manual therapy applied by a clinician according to observations and set guidelines for making therapy adjustments and (b) automated therapy applied by a physiological closed-loop control (PCLC) device according to a control algorithm with the possibility of a clinician acting in a supervisory capacity (e.g. monitoring Bispectral Index (BIS) for additional safety in anesthesia). miliar readers. This includes a brief introduction to modeling and control, within the scope of those methods common to physiological systems, in Section II ; the use of case studies in Sections III-IV to frame and discuss important topics in current research; and a minimalist presentation of the detailed mathematical models, which are all v ery well documented in the cited references. Following the case studies, in Section V we will discuss several aspects from these in vestigations that are of great importance in the broader scope of physiological control as well as possible directions for the future. I I . P R E L I M I NA R I E S Regardless of the controlled v ariables and outputs, the general process implemented in PCLCs can be represented conceptually by a diagram resembling Fig. I . Here, a control algorithm – normally implemented on a computer/processor – provides an interface between (i) tar get v alues for phys- iological variables that ha ve been specified by clinicians, (ii) feedback signals containing measurements and estimates of system variables, and (iii) the infusion pumps or other actuators that apply treatment to the patient. Notice, from a high-lev el perspective, there is little dif ference between the ”automated” approach and the classical manual approach aside from the simplification of tasks required from the clin- ician/patient, who no longer has to interact with the infusion pump (or other actuator) directly and instead can focus on specifying treatment goals and assessing therapy performance. Recognizing the generality of this concept, many agencies and or ganizations are in the process of dev eloping more rigorous guidelines for commercial PCLCs that will help to establish the scope of this up-and-coming class of devices. As noted in a recent paper from researchers at the Center for Devices and Radiological Health (CDRH) at the US Food and Drug Administration (FD A), the CDRH maintains a working definition of PCLC medical devices (or PCLC devices) as, ”a medical de vice that incorporates physiological sensor(s) for automatic manipulation of a physiological v ariable through actuation of therap y that is con ventionally made by a clinician” [9] . This definition – demonstrated in Fig. Ib – recognizes the role of automation in PCLC while allo wing a great deal of flexibility in the disorder being treated, the types of sensors and therapies used (many are certainly yet to be discov ered), and the extent to which the PCLC operates independent of clinician intervention – kno wn as the lev el of automation (LO A) [9] . T o help guide the development of commercial PCLC devices, the United States [10] , Canada [11] , and the European Union [12] have formally recognized v ersions of the recently de veloped IEC 60601-1-10 consensus standard [13] , which is the first standard to provide, ”requirements for the dev elopment (analysis, design, verification and validation) of a physiologic closed-loop controller (PCLC) as part of a physiologic closed-loop control system (PCLCS) in medical electrical equipment and medical electrical systems to control a physiologic v ariable” [13] . Beyond regulatory concerns, such guidance may help the broader research and dev elopment communities establish a common language (possibly even one that is shared among medical and engineering professionals); a set of safe design practices, testing procedures, and quality management objectives; and a platform to qualify and address issues raised by designers, clinicians, and patients. T reating this feedback system similar to those in more tradi- tional engineering disciplines, the design and implementation of PCLCs requires accurate models of the patient, sensor, and actuator dynamics, including any fundamental limitations and safety constraints. While it may be obvious that the sensor and actuator dynamics can be described using traditional dif- ferential equations, similar mathematical modeling techniques can also be applied to develop equations representing patient physiology and pharmaceutical action. In fact, the human body is generally modeled using compartmental techniques (see Section II-A ) from mathematical systems physiology , which can describe the behavior of healthy and perturbed or gan/tissue interactions as well as the distribution/transportation of phar - maceuticals in the body (pharmacokinetics) and the action of these pharmaceuticals on certain organ systems (pharmacody- namics) in terms of nonlinear differential equations. A. Compartmental Models When working with physiological or biological systems, the dynamic interactions of system states/variables are often depicted in the form of compartmental models. This represen- tation is slightly different from the standard block diagram models of systems theory b ut con veys similar information. Under this modeling paradigm, a system is comprised of one or more compartments, each of which has a (normally) fixed volume V i and contains a variable state quantity q i ( t ) . The representation of this quantity q i ( t ) as a fraction of the compartment volume giv es the concentration c i ( t ) = q i ( t ) /V i T ABLE I E X AM P L E S O F R E SE A R CH I N P H Y SI O L O GI C A L C L OS E D - LO O P S Y S TE M S ( P C L C S ) . T reatment T arget T reatment Actuator T reatment Sensor Control Method Blood Glucose (e.g. [1] ) Insulin infusion/bolus Blood Glucose Concentration PID Anesthesia (e.g., [2] ) Propofol/Remifentanil Electroencephalogram /Bispectral Index Fuzzy-Logic Blood Pressure (e.g., [3] ) V asoactive drugs such as phenylephrine Mean Arterial Pressure Linear Parameter V arying Control Anemia in End-stage- Renal-Disease (e.g., [4] ) Erythropoiesis- stimulating agents Hemoglobin Level Model Predictive Control T umor Growth (e.g., [5] ) Abiraterone T umor Composition Evolutionary Game Theory Heart Failure (e.g., [6], [7] ) Implantable Rotary Blood Pump Left V entricle Pressure /Flow Pulsatility Sliding Mode Control Fluid Resuscitation (e.g., [8] ) Intrav enous fluid administration (IV injection of replacement fluids/blood) Stroke V olume; Heart Rate; Mean Arterial Pressure; Pulse-Pressure V ariation Predictiv e Rule- Based Control Fig. 2. General compartmental model for 2 compartment systems . When the change in these quantities or concentrations takes the form of ordinary differential equations, a compartmental modeling diagram can be drawn to depict the contributions of each state quantity to the dynamics (similar to state-variable representation). In the case of a linear time-in variant system, ev ery compartment is associated with a linear first order differential equation described by the signals entering and leaving the respectiv e compartment. Considering the general two-compartment model shown in Fig. 2 , the change in q 1 ( t ) is giv en by ˙ q 1 ( t ) = u 1 ( t ) − k 21 q 1 ( t ) + k 12 q 2 ( t ) − k 01 q 1 ( t ) (1) where u 1 ( t ) is the input to compartment 1; k 01 q 1 ( t ) is the leakage from this compartment to the en vironment; k 21 q 1 ( t ) is the exchange from compartment 1 to compartment 2; and k 12 q 2 ( t ) is the exchange from compartment 2 to compartment 1. Similarly , based on the arrows pointing tow ard and away from compartment 2, the change in q 2 ( t ) can be written as ˙ q 2 ( t ) = u 2 ( t ) + k 21 q 1 ( t ) − k 12 q 2 ( t ) − k 02 q 2 ( t ) (2) where u 2 ( t ) is the input to compartment 2; k 02 q 2 ( t ) is the leakage/loss from compartment 2 to the en vironment; and all other terms are the same as described previously . T ogether , these equations describe the exchange between compartments and the en vironment. In general, there may be n compartments, each of which may ha ve a constant or changing volume V i and can be linked to any or all other compartments by linear or nonlinear interactions. For more detailed information on compartmental modeling, readers are referred to the extensiv e coverage in [14] . B. Control Methodologies In this section, we briefly revie w the fundamentals of the 3 most common control methods currently used in physiological systems: (i) proportional-integral-deri vati ve (PID) control, (ii) model predictiv e control (MPC), and (iii) fuzzy-logic (F-L) control. The application of these control schemes to specific physiological systems, including the advantages, variations, and recent de velopments to each strategy , are described in later sections. Of course, many other control methods are also being pursued in the literature and will be discussed as needed. 1) Proportional-Inte gral-Derivative (PID) Contr ol: PID control is one of the most frequently used control approaches in industrial systems due to its simple mathematics, well- known gain tuning methods, and broad application to both linear and nonlinear systems. The Laplace domain transfer function of the ideal PID controller can be gi ven by U ( s ) E ( s ) = k p 1 + 1 sT i + T d s (3) where s is the complex-valued Laplace variable; E ( s ) and U ( s ) are, respectively , the Laplace domain input and output of the PID controller; k p is the proportional gain; T i is the integral time-constant; and T d is the deriv ativ e time-constant. In most cases, the coefficients k p , T i , and T d are free- parameters that take constant values and are designed to obtain desirable system properties. The values of these parameters may be obtained by either theory-based design strategies or numerical/experimental tuning methods. 2) Model Pr edictive Contr ol (MPC): Model predictiv e con- trol is a robust and optimal control technique used to handle systems with constraints while optimizing the controller action and predicting system outputs for robustness against noise and Fig. 3. Fuzzy logic sets disturbances. The basic formulation of this controller can be giv en by U ∗ t ( x ( t )) := arg min N − 1 X k =0 q ( x t + k , u t + k ) (4) subject to x t = x ( t ) (Measurement or estimation) x ( t + k +1) = Ax ( t + k ) + B u ( t + k ) (System model) x ( t + k ) ∈ χ (State constraints) u ( t + k ) ∈ u (Input constraints) U ∗ t ( x ( t )) = { u ∗ t , u ∗ ( t +1) , , u ∗ ( t + N − 1) } (Optimization variables) where U ∗ t ( x ( t )) is the optimal input sequence for the entire planning windo w N ; q is the cost function; and χ and u are, respectiv ely , sets of state and input constraints. At any giv en time t , this controller finds the set of optimal control actions U ∗ t based on the predicted dynamics ov er the entire window N and implements only the first control action u ∗ t in this sequence. 3) Fuzzy-Logic (F-L) Contr ol: Fuzzy systems are knowledge-based or rule-based systems consisting of fuzzy ”if-then” rules that represent heuristic verbal conditions as continuous membership functions Fig. 3 [15] . This method can be used to model and control complex dynamics, nonlinear systems, and systems that are difficult to express mathematically . In general, fuzzy-logic systems can be classified as Mamdani and Sugeno systems [16]–[18] . Fuzzy- logic systems have been successfully implemented in many industries. C. Case Studies In order to demonstrate and contextualize many important principles in PCLCs, we will proceed over Sections III and IV with detailed case studies of the two most acti ve areas in physiological control: (i) automated anesthesia and (ii) automated insulin therapy for diabetes. For each system, a detailed background on therapeutic goals, physiology , and mathematical representations is given prior to discussions on current trends in the control methodologies and specific challenges encountered in each area. I I I . D I A B E T E S A N D T H E A RT I FI C I A L P A N C R E A S T o understand the problem of blood glucose regulation, let us begin with a brief revie w of the essential physiology of a properly functioning glucoregulatory system and build tow ards a fundamental understanding of the diabetic disorders (pathophysiology) as well as their underlying causes (etiology) and associated health risks (complications and comorbidities). By way of this introduction, we hope to provide clarity for the coming discussions on system models and insights when broaching the subjects of treatment and closed-loop therapy . A. Physiology of the Healthy Blood Glucose Regulatory Sys- tem The natural re gulation of blood glucose in a healthy in- dividual is the result of a variety of interacting physiological processes inv olving hormones, neurotransmitters, and our own consumption of food. Historically , the accepted metric for judging the quality of this regulation is the amount of glucose present in the blood stream, measured as a molar (mmol/L) or mass (mg/dL) concentration. This blood glucose concentration is commonly referred to as an individual’ s blood glucose lev el and is generally well-maintained between about 4-6 mmol/L (72-108 mg/dL) during healthy glucose homeostasis. Ho we ver , ev en in healthy subjects, there are occasional excursions above this range (e.g., after recent meals) or slightly belo w it (e.g., during or after periods of high activity). The interactions that gov ern glucose concentrations are highly div erse and most, if not all, of these interactions are reciprocal – both the glucose concentration and the acting processes are mutually affected by the regulatory procedure. The systems in volv ed in this regulation include the brain, li ver , kidney , gastrointestinal tract, adipose and muscle tissues, and pancreas. Of these systems, the pancreas is arguably the most critical for maintaining proper blood glucose and will be our focus in the sequel. The pancreas, located behind the stomach, is a glandular organ primarily responsible for secreting digestiv e enzymes through ducts to the small intestines and releasing pancreatic hormones into the blood stream. While both enzymes and hor- mones play important roles in proper metabolic homeostasis, it is the latter (i.e., hormones) that are principally responsible for regulating blood glucose lev els [19] . These hormones, known as the pancreatic hormones, are produced at small clusters of endocrine cells (i.e., cells that secrete directly into the blood stream), known as islets, that are distributed sparsely throughout the pancreatic tissue (com- prising < 2% ). These pancreatic islets (or islets of Langerhans) are constructed from a combination of 5 kinds of endocrine cells called α -cells, β -cells, γ -cells, δ -cells, and -cells. Each of these cell types secrete a specific set of hormones that are known to be inv olved in at least some aspects of normal glu- coregulatory function. Of these cells, the α and β types make- up > 80% of all pancreatic islet cells and are considered the 2 most essential endocrine cells for blood glucose regulation [19] . The first of these, α -cells, produce a single hormone called glucagon, which driv es the production of endogenous glucose by catabolizing (breaking-down) stored glycogen in the liver (kno wn as hepatic glycogenolysis) and to a lesser extent by promoting the generation of glucose from non- carbohydrate compounds in the liver and kidneys through pro- cesses termed, respectiv ely , hepatic and renal gluconeogenesis. The latter type of cells, β -cells, produce 3 hormones called C -peptide, amylin, and insulin, all of which hav e important roles in glucose regulation and are believed to correspond with various symptoms of blood glucose pathophysiology . Of these 3 hormones, insulin is the most well-known, and is responsible for lowering blood glucose concentrations through promotion of glucose utilization by adipose and muscle tissues, as well as by prompting the creation of glycogen and fatty acid energy stores from glucose through glycogenesis and lipogenesis, respectiv ely . Thus, in contrast to glucagon, which raises blood glucose le vels, insulin provides the primary means to reduce this concentration [19] . As one might expect, based on their roles in the blood glucose regulatory function, glucagon is primarily secreted during periods of low blood glucose such as sleep or fasting, while insulin secretion is stimulated by elev ated blood glucose concentrations and the incretion effect promoted by ingestion of glucose, fructose, amino acids, and long-chain free fatty acids [19] . Of course, insulin secretion is highest follo wing meals (i.e., when blood glucose lev els are at their highest). T ogether , glucagon and insulin provide the basis for many simple and intuitiv e bi-hormonal models of the pancreatic endocrine system. While many of the physio-chemical mech- anisms for these secretions and uptakes are quite well known, it is important to understand that these hormones are released directly into blood stream and interact with many different organ systems. Thus, the specific mechanisms and outcomes of these interactions remain highly active research topics. Of course, the 2 other β -cell hormones and those secreted from the γ , δ , and -cells are also intrinsic to proper phys- iology . Unfortunately , the functions of these hormones are not yet as well understood as those of insulin and glucagon and are often (perhaps necessarily) disregarded in the control- oriented literature. Howe ver , there is reason to belie ve study of these pancreatic hormones, and their roles in both physiology and pathophysiology , will likely lead to better multi-hormonal models and treatment approaches in the coming years [19], [20] . For example, amylin – produced by β -cells along with insulin – is believ ed to perform complementary functions to the insulin-based removal of glucose, instead inhibiting the appearance of absorbed (exogenous) and glycogenolytic glucose by , respectiv ely , reducing the flow of food from the stomach to the small intestines (gastric emptying) and blocking the release of postprandial glucagon (i.e., inhibiting unnecessary endogenous glucose secretion following meals) [20] . While an FD A-approv ed amylin analog (i.e., pramlintide) is av ailable, the cost of treatment with such complimentary hormone analogs may be prohibitively expensi ve and perhaps ev en detrimental if not properly (e.g., physiologically or pseudo-physiologically) controlled. 1) P athophysiology and Etiology: T ype 1 Diabetes Mellitus (T1DM): The term type 1 diabetes mellitus describes blood glucose regulatory disorders resulting from the complete (or near complete) destruction of all pancreatic β -cells, leading to hyperglycemia and life-threatening complications. T1DM is generally caused by an auto-immune response, but does occur infrequently without any known or identified cause (idiopathic T1DM) [21], [22] . The characteristic β -cell destruction of T1DM leads to a dependence on external sources of insulin (i.e., insulin therapy) for surviv al; hence, this form (esp. the auto-immune form) was previously identified by the term insulin-dependent diabetes mellitus (IDDM) [21], [23] . T ype 1 diabetes accounts for 5-10% of all cases [21] . Considering the preceding discussions on healthy glucoregulatory function, it is clear that destruction of β -cells in T1DM suppresses the production of the insulin, amylin, and C -peptide hormones. W ithout these hormones (particularly insulin), cells throughout the body cannot remove glucose from the blood stream for use in metabolic processes (e.g., storage as glycogen). As a result, the body turns to ward the metabolism of fats, which leads to increased levels of free fatty acids and ultimately ketones in the blood stream [23] . Overproduction of the latter leads to life-threatening diabetic ketoacidosis. Furthermore, complications due to elev ated blood glucose lev els (hyperglycemia) appear across numerous organ systems and include microv ascular diseases such as neuropathy , retinopathy , and nephropathy [23], [24] . In T1DM, the absence of interactions between pancreatic α -cells and β -cells remov es an important pathway for natural glucagon regulation, leading to continued glucagon production during periods of elev ated blood glucose, resulting in more sev ere hyperglycemia and ev en greater risk of complications [23] . Unfortunately , while many of the risks associated with T1DM can be reduced by tight regulation of blood glucose levels [23] , the destruction of β -cells affects multiple hormones and man y systems, some of which are not fully restored by external blood glucose regulation. For instance, increased risk of cardiovascular disease – the leading cause of reduced life-expectanc y in type 1 diabetics – remains prev alent in type 1 diabetics with good blood glucose control [23] . T ype 2 Diabetes Mellitus (T2DM): The term type 2 dia- betes mellitus describes a family of blood glucose regulatory disorders that are marked by relativ e insulin deficiency , due to a combination of poor insulin production and poor insulin utilization, leading to episodes of hyperglycemia [21], [22] . Previously identified as non-insulin-dependent diabetes melli- tus (NIDDM), current opinion is trending to wards a more grad- uated classification system that may improve differentiation of T2DM by sev erity and pathophysiology of impaired insulin- secretion (production) and insulin-sensiti vity (utilization) [21], [25], [26] . T ype 2 diabetes accounts for about 85% of all cases [21], [23], [26] . Unlike T1DM, T2DM is not an autoimmune disorder and has neither a single underlying cause (etiology) nor a unique physical origin. Instead, the term T2DM applies to various non-specific disorders (i.e., not specifically meeting the criteria for definition as any distinct disorder such as monogenic, gestational, or type 1 diabetes mellitus) that result in chronic e xcess blood glucose [21], [22] . While in some cases, T2DM is marked by a reduced pancreatic insulin secretion that results in a relati ve insulin deficiency , in others, insulin production is unaffected, but cells exhibit an inability to utilize available insulin (i.e., insulin resistance). Howe ver , most of the type 2 diabetic population exhibit both insulin deficiency and insulin resistance [21], [22], [25], [26] . Thus, it is becoming more common to characterize each instance of T2DM along gradients of insulin-sensitivity and insulin secretion (among other v ariables including coexisting conditions), to better identify root-causes and optimal treatment regimens [25], [26] . While T2DM is not normally life-threatening by itself, it is associated with reduced life-expectancy due to macrov ascular complications, as well as numerous microv ascular complications that are believ ed to be linked with episodes of hyperglycemia. Like T2DM itself, the accompanying complications are di verse and, some research suggests, potentially well-correlated with pathophysiologically distinct forms of the disorder [25]–[27] . Gestational Diabetes Mellitus (GDM): Glucose intolerance that dev elops in women during pregnanc y , indicated by the emergence of persistent hyperglycemia, is known as gestational diabetes mellitus [21], [22], [28] . Like T2DM, GDM is comprised of a highly heterogeneous set of disorders, which may present predominantly insulin resistant or insulin deficient characteristics [28] . While standards for diagnosing GDM vary , the critical feature of GDM is that it is developed during pregnancy and was not present prior [21] . The pathophysiology of GDM is not yet well-understood [28] , but ongoing research may benefit from more accurate and detailed sensing technologies. GDM is a disorder of international interest and can result in complications for both mother and child and can progress into a form of T2DM after pregnancy [21] . Other Specific T ypes of Diabetes: In addition to T1DM, T2DM, and GDM, there are numerous, less common dia- betic disorders (together comprising ¡5% of cases) with more specific association with distinct etiological factors such as genetics, drug or chemical exposure, and pancreatic diseases [21], [22] . Dif ferentiation of these specific forms from T1DM and T2DM is critical, as the treatment requirements for many of these disorders differ from general guidelines and mischaracterization can lead to poor or detrimental outcomes and unnecessary expenses [21], [25] . B. T raditional Monitoring and T reatment Considering the v ariety of disorders that may exist, or coexist, in diabetic individuals, it is not surprising that there are a plethora of diabetic diagnostic tools and interventions, some of which hav e been shown to be most effecti ve in mon- itoring and treating specific disorders, while simultaneously potentially detrimental for treating others. Like wise, treatment guidelines are often adjusted for improv ed effecti veness in specific demographics often based on age, gender , and overall health, with the final recommendation for individuals tailored by their physician [29], [30] . 1) Laboratory Scr eening: For the past half century , blood glucose regulatory function and concentration targets for di- abetics and non-diabetics have primarily been specified ac- cording to 2 standard measurements: (i) static blood glucose concentrations and (ii) percent glycated hemoglobin (HbA1c, or less formally A1C). While a static measurement of blood glucose provides insight into the current glycemic state (i.e., the current mmol/L concentration of glucose in the blood stream), it does not provide any insight into recent concen- trations or an indication of whether this v alue is increasing or decreasing [31] . Con versely , A1C provides an approximate indication of the av erage blood glucose concentration over a period of about the preceding 2-3 months [29], [31] . While A1C is less sensitive to daily blood glucose variations and a primary predictor of diabetes related complications, acceptable A1C values have been shown to vary significantly across pop- ulations and with v arious en vironmental factors, and further do not provide any specific information regarding occurrences of hypoglycemia, hyperglycemia, or their severity [21], [29] . In clinical settings, diabetic screening is often performed using plasma blood glucose concentrations that are measured by accredited laboratories using standardized tests [32] to de- termine lev els under the moderately controlled circumstances either following a period of fasting to obtain the so-called fasting plasma glucose (FPG) le vel or 1-2 hours into an oral glucose tolerance test (OGTT) administered after a period of fasting. If the FPG or OGTT results fall belo w certain thresholds (see characteristic thresholds from the AD A [21] in T able II ), these tests results, respectively , indicate impaired fasting glucose (IFG) or impaired glucose tolerance (IGT) associated with metabolic disfunction. The accuracy of these tests depends heavily on patient adherence to the f asting guidelines and clinical sample handling. Thus, for clinical A1C (also performed in laboratories [32] ), FPG, and OGTT testing, confirmation of an initial diagnosis of diabetes often requires that 2 of these values fall outside the acceptable thresholds [21] . 2) Self-Monitoring of Blood Glucose (i.e., ”F inger-Stic k” Monitoring): Once diagnosed, most diabetics (especially those with T1DM or T2DM requiring insulin therapy) will re- quire portable diagnostic equipment so that blood glucose concentrations can be monitored according to the severity of their disorders or in the ev ent of symptoms that indicate excursions from normal glycemic control. For many years, the predominant method for this daily testing was self-monitoring of blood glucose (SMBG) by so-called ”finger-stick” devices [31], [33] , which require drawing a small sample of blood during every test. Initially accepted for their marked improv e- ment ov er urine testing equipment, use of these finger -stick devices causes pain, and measurements can be uncomfortable or unsafe (due to blood spill) to perform in public [31] . While significantly less accurate than accredited laboratory testing [32] , these portable diagnostic tools have enabled diabetics to maintain tighter glycemic control by regular assessment of their blood glucose levels, especially prior to meals, sleep, and insulin dosing. Howe ver , like static laboratory measurements, T ABLE II F P G, O G TT, A N D A 1 C T H R ES H O L DS F O R I N IT I A L D I AG NO S I S O F D I A BE T E S . T est Prediabetes Thresholds Diabetes Threshold FPG 100-125 mg/dL (5.6-6.9 mmol/L) ≥ 126 mg/dL (7.0 mmol/L) 75-g OGTT (2 hour) 140-199 mg/dL (7.8-11.0 mmol/L) ≥ 200 mg/dL (11.1 mmol/L) A1C 5.7-6.4% (39-47 mmol/mol) ≥ 6.5% (48 mmol/mol) SMBG does not provide any historical or predictive informa- tion by itself and must be combined with knowledge of recent past/future carbohydrate intake, ex ercise, and medication dos- ing to estimate appropriate correcti ve actions. Coupled with the incon venience and limitations of re gular blood sampling, the static nature of SMBG has prompted the modern quest for safe, accurate, and continuous blood glucose monitors (CGMs). 3) Continuous and Flash Glucose Monitors: Starting in the late 1990s, diagnostic tools for continuous home monitoring of blood glucose began to emerge as a potential remedy to the shortcomings of traditional SMBG [31] . Originally designed to record, but not display , blood glucose measurements over sev eral days – to be analyzed retrospectiv ely by a physician [32]–[34] – more recent real-time CGM (rtCGM) de vices that log data and provide immediate access to measurements and analytical trends are now av ailable on a broad commercial scale ( T able III ) [31] . Unlike SMBG, which requires taking blood samples at each measurement time, most rtCGM de vices are minimally in vasi ve and measure glucose concentrations subcutaneously through the interstitial fluid rather than whole blood or plasma [32], [34] . These sensors can be worn continuously for up to a week, and provide updated measure- ments every 5 minutes, requiring finger-stick measurements for calibration (up to twice per day) [32] . While only one such device is FD A approved for non-adjunctiv e use in insulin therapy (i.e., for use in determining insulin dosage without the need to confirm blood glucose levels by traditional SMBG), this is a recent dev elopment and more de vices may be e xpected in the future [29] . This consistent av ailability of current data has led to sev eral important features that are impossible with traditional SMBG. First, man y rtCGM systems allow users to set alarms that indicate or e ven predict the onset of hypoglycemia, allo wing diabetics to increase their carbohydrate/glucose intake and av oid hypoglycemia, which is especially useful for those suffering from hypoglycemia unawareness [31] . Second, many rtCGM systems allow users to automatically share data/trends with caregi vers and physicians, having important implications for young children and their parents [31] , as well as elderly and disabled individuals who are living alone. Finally , rtCGM systems not only provide improved predictive measures for users through real-time measurements, measurement histories, and predicted glycemic trajectories, many of these systems can be interfaced with portable insulin infusion systems to auto- matically suspend insulin dosing when near or approaching hypoglycemia [31], [34] . Combinations of rtCGM and insulin infusion systems, known as sensor augmented pump (SAP) therapy , are further enabling improved overnight glycemic control, and represent a step toward wide-spread acceptance of closed-loop insulin therapy systems. More recently , a new class of glucose monitor kno wn as the ”flash” or ”intermittently sensed” glucose monitor (isCGM) have become av ailable that provide immediate, on demand glucose measurements (lagging true blood glucose concentrations by about 5 minutes) in a minimally inv asiv e package worn continuously for 14 days without calibration (the sensing element is replaced after 14 days without re- quiring any SMBG calibrations) [33], [34] . While this device may not be suitable for those with hypoglycemic unaw areness (dev eloped by repeated hypoglycemic excursions) as it does not currently provide an autonomous alert, various studies have demonstrated impro ved time-in-range and greatly reduced time in hypoglycemia in both type 1 and type 2 hypoglycemic aware populations previously using finger-stick SMBG. Small observational studies indicate that these results may follow from the rapid and pain-free nature of blood glucose self- monitoring with these devices, which allows users to follow ev en stringent (¿10 times per day) testing guidelines without experiencing discomfort [33] . This research has also reported behavioral changes in patients, including better adherence to monitoring timelines, which may produce additional societal benefits, as unchecked lo w blood sugar is associated with otherwise av oidable injuries and a increased risk of car ac- cidents [42] . Due to time lag and measurement inaccuracy – possibly exceeding 15% mean absolute relati ve difference (MARD) from matched reference measurements – users of these devices must still perform traditional SMBG tests when hypoglycemia is expected, measurements vary rapidly or do not corroborate symptoms, and when required by law [33] . While these flash monitors are available and gaining significant interest, there remain significant concerns in areas including sensor placement, failure modes, user insulin stacking due to time lags, and measurement variations over the duration of the 14-day sensor life-span. Studies on CGM devices have consistently indicated that, compared to control groups using SMBG, insulin treated diabetics using CGM systems sho w improved HbA1c as well as significantly decreased frequency and se verity of hypoglycemia and hyperglycemia, with rtCGM demonstrating superior outcomes to isCGM [29], [31], [33], [34] . Despite their benefits, modern CGM sensors suffer from delays due to glucose transport from plasma to the interstitial fluid, may be subject to sensor interference from mechanical [43] and phar- maceutical (e.g., acetaminophen [44] ) sources, and are kno wn to demonstrate reduced accuracy during the first day of use T ABLE III E X AM P L E S O F C OM M E R CI A L L Y A V A I LA B L E C G M C O M P ON E N T S A N D D E V I CE [ 3 1] , [ 35 ] – [ 41 ] . Manufacturer Device Accuracy (MARD %) Duration Required Calibration Medtronic Enlite Sensor* - 6 days - Enlite with Gaurdian 2 link transmitter 11 6 days 2/day Enlite with MiniLink transmitter 13.6 6 days 2/day Enlite with Guardian connect transmitter 11 6 days 2/day Guardian Sensor 3* - 7 days - Guardian Sensor 3 with Guardian link 3 transmitter 10.6 7 days 2/day Guardian connect system 10.6 (abdomen) 9.0 (arm) 7 days 2/day Dexcom G4 13 7 days 2/day G5 9 7 days 2/day G6 9.8 10 days none Abbot FreeStyle Libre 11.4 14 days none *Sensing elements may limit the duration CGM systems and affect their accuracy but do not alone give the mean absolute relative difference (MARD) or calibration schedule. and at low glucose concentrations (i.e., near hypoglycemia) [31], [43] . W ith further research, these issues may be resolved in the near future. 4) Non-Invasive , W earable, and Implantable Monitoring: During the past decade, there has been increased academic and commercial interest in the dev elopment of non-in vasi ve and continuous monitoring of blood glucose levels through wearable technologies. Unlike traditional monitors, many of these sensors do not measure blood glucose directly , but at- tempt to leverage alternati ve fluids such as urine, sweat, sali va, breath, ocular fluid, and interstitial fluid [45] . While some of these devices are promising, with man y designed to be discrete and comfortable, measurements taken from these alternativ e physiological fluids tend to be less reliable than traditional methods, and any such technology will surely require rigorous testing and clinical studies before any statements can be made regarding safety and accurac y [46] . 5) Non-Insulin Therapy: While not generally suitable for T1DM, non-insulin oral antihyperglycemic agents are com- mon in treating T2DM. In fact, the first-line pharmaceuti- cal therapy for T2DM is a non-insulin antihyperglycemic called metformin, which reduces hepatic glucose production (i.e., in the li ver) and is often highly ef fectiv e at reducing HbA1c and associated cardiov ascular risks, without raising significant concerns of hypoglycemia [30] . According to AD A guidelines, metformin is frequently prescribed as part of a dual or triple pharmaceutical regimen (combination therapy) when metformin alone does not provide sufficient reductions in HbA1c – used in this case as a primary indicator of glycemic control. These additional treatments may be oral agents such as sulfonylureas (increases insulin secretion), DPP-4 inhibitors (increases glucose dependent insulin secre- tion and decreases glucose dependent glucagon secretion), and Thiazolidinediones (increases insulin sensitivity), but may also include subcutaneous injections of insulin or GLP-1 receptor agonists, which act on multiple levels [30] . Considering the underlying lifestyle factors (e.g., sedentary behavior , obesity , and diets that are high in fats and sugars [47] ) that are known to promote the development of T2DM, prev ention and treatment through lifestyle modification is a significant area of study [47]–[49] . Intensiv e lifestyle modifi- cations hav e been shown to improv e HbA1c, and are believ ed to be associated with reduced microcardiovascular and macro- cardiov ascular complications as well as lower rates of diabetes related mortality in T2DM. Similarly , there is e vidence to support the benefits of nutritional balance in T1DM; howe ver , the direct evidence for reduction of diabetes related ailments (beyond general health and fitness) associated with physical activity and obesity are much better supported for T2DM. In fact, due to the increased glycemic variability of individuals with T1DM, the AD A recommends physicians take care in prescribing specific exercises and durations for individuals when using physical activity as a means of glycemic control [49] . While these non-insulin therapies are often effecti ve at slowing the progression of T2DM, in some cases leading to remission of the disorder, most individuals with T2DM will often require insulin therapy at later stages of treatment, ev en with careful adherence to therapy guidelines and healthy lifestyle choices [30] . 6) Multiple Daily Insulin Injections: Recalling that severe insulin deficiency is the hallmark of T1DM related β -cell destruction, it is not surprising that primary treatment for T1DM is insulin therapy [30] . The most common method of insulin therapy consists of the regular periodic injection of basal (baseline) insulin multiple times a day – known as multiple daily insulin injections (MDI)– supported by additional insulin doses (boluses) and oral glucose or glucagon as needed to maintain normoglycemic conditions (e.g., at meal times). While several dif ferent practical guidelines exist for calculating the required basal and bolus insulin doses, dosage is always described in terms of units of insulin or units/day (based on weight as units/kg/day). Recent pharmaceutical advances hav e produced a variety of injectable insulins that may be categorized as either synthetic human insulin or modified insulin analogs. Referring to T able IV [30], [50] , v ariations in these insulin formulations ha ve led to markedly different properties, and so it is common to classify insulins according to their concentration in units/mL (e.g., an insulin specified as U-# contains # units/mL), the time it takes for the insulin to begin reducing blood glucose lev els (onset time), the time it takes for the insulin action to reach its peak effecti veness (peak time), and the length of time that the formulation will remain effecti ve after injection (duration) [50] . When prescribing or taking insulin, each of these factors comes into play and (in an MDI scheme) make some forms better suited as basal insulin and others as mealtime boluses [30] . Thus, e ven within insulin therapy , there is now an array of options that allow for personalization and situational ev aluation of treatments. Note, the use of insulin is not limited to T1DM, and there are, of course, many factors affecting the selection, efficacy , and safety of different insulin formulations and treatment regimens. In addition to single insulin formulations, some injections are de veloped as a mix of rapid acting (i.e., quick onset and peak times with short duration) and long acting (i.e., delayed onset time, low or no peak, and long duration) insulins to provide both basal and bolus action from a single injection, thereby reducing the number of injections required per day [30], [50] . In some cases, an analog of the pancreatic β -cell hormone amylin may be used to augment insulin therapy [30] . Howe ver , addition of such agents may increase treatment costs signifi- cantly . Further , in certain cases, it may be beneficial to perform pancreas or islet transplantation, in place of insulin therap y . 7) Continuous Subcutaneous Insulin Infusion: Initially de- veloped over 40 years ago [51] , continuous subcutaneous insulin infusion (CSII) systems are portable pump therapy devices that are generally constructed as a combination of an onboard insulin reserv oir, an electromechanical infusion pump, and an infusion apparatus (tubing and cannula) [51], [52] . According to v arious studies, these systems can be operated using synthetic human insulin or rapid-acting insulin analogs (RAIA), with RAIA providing superior performance to synthetic human insulin [52] . In most cases, CSII uses the same basal dosage as MDI, with the basal insulin dosage applied more consistently over the day in CSII [52] . While pragmatic evidence supporting substantial improve- ments to patient outcomes achieved by switch from MDI to CSII is minimal or frequently contradicted [30], [52] , there is evidence that switching to CSII is beneficial for diabetics with poor glycemic control under MDI [52] , and CSII is beginning to see adoption as a recommended first-line therapy for some type 1 populations (e.g., adolescents) [30], [52] . Howe ver , when CSII is used in conjunction with rtCGM for sensor augmented pump therapy , the benefits are more pronounced. Connected systems of monitoring and treatment de vices, like SAP , provide therapeutic opportunities that cannot be obtained using MDI, such as ov ernight glycemic regulation. As an extension of this concept, the ability to continuously monitor and control insulin concentrations (whether by CSII or other nov el future technology) is paramount to the development of closed-loop insulin therapies. Like SAP , closed-loop therapy systems are designed to utilize the features of continuous infusion that cannot be matched by MDI, with or without CGM, to achiev e tighter glycemic control than is possible by any current treatment. As with all therapy de vices, CSII must be appropriately used and maintained, as de vice performance depends on proper operation (e.g., timely replacement of consumables) to av oid failure modes such as clogged or impeded infusion pathways, which can lead to insulin deficiency and hyperglycemia [52] . C. Modeling for Analysis and Control of Diabetes Building on nearly 2 centuries of research and modeling studies focused on the pancreatic endocrine system, the re- cent worldwide interest in diabetes has instigated a surge in the dev elopment of glucoregulatory models and simulation platforms, primarily aimed at T1DM [53]–[57] and, to a lesser extent, T2DM [54], [56], [57] . In fact, according to sev eral re views [53], [57] , there are well ov er 100 dif ferent mathematical models describing, reproducing, and/or predict- ing various aspects of the blood glucose regulatory dynamics from cellular mechanisms and neurological signaling to long- term outcomes, disease progression, and risks of complications [57] . The mathematical descriptions used in these dynamic models are diverse and include ordinary , partial, and stochastic differential equations, which often include time-delays, and are frequently treated with a degree of modularity – as new models may be established as composites of subsystems from multiple predecessors [57] . Among these models, some, such as the mechanistic models that describe the C a 2+ and K + ion channel mediated release of insulin from pancreatic β -cells, ha ve solid footing in the first principles of electrochemistry and physics. Howe ver , the inclusion of f such models tends to add unwarranted complexity as the pathophysiological and etiological rele vance of these states are not well-understood, while producing negligible variations in the observable input-output behaviors that are presently more relev ant to healthcare outcomes. Thus, in the dev elopment and analysis of systems for the closed-loop treat- ment of diabetes, models are generally categorized according to their lev el of detail and may be considered as either (i) reduced complexity control-oriented models – for controller synthesis methods [53] , or (ii) high-fidelity models for analysis and validation [55], [56] . In either case, these models attempt to replicate the glucoregulatory dynamics of diabetic patients. Howe ver , while the former is concerned with representing accurate input-output relationships, in the latter cate gory it is also important that models are structured such that vari- ables and parameters maintain physiological significance and produce physiologically viable results at internal states. Thus, T ABLE IV C H AR AC T E R IS T I C S O F V A R IO U S C O M ME R C I AL LY AV A I L AB L E I N S UL I N F O R M UL A T I O N S . Classification F ormulation Onset Time Peak T ime Duration Rapid acting Aspart < 15 min 30-90 min 3-5 hours Lispro < 15 min 30-90 min 3-5 hours Glulisine < 15 min 30-90 min 3-5 hours Short acting Regular Human Insulin < 1 hour 2-4 hours 4-8 hours Intermediate acting Human NPH 1-2 hours 4-10 hours 10-18 hours Long acting (basal) Glargine 1-2 hours - 24 hours Detemir 1-2 hours - 24 hours Ultra-long acting (basal) Degludec 30-90 min - 42 hours models of this second kind, can provide additional insights into substance and organ interactions, and have become known as virtual patient models – as they are generally used as simulated analogs of real patients. While early models of virtual patients utilized nominal or av erage parameter values, obtained from population studies, their more recent use in dev eloping closed-loop therapy sys- tems has led to a need for individualized model behaviors that closely match the sev erity and mode of dysfunction in specific patients. Accordingly , while some parameters may still be estimated by nominal values from population data [58] , others ha ve been identified from measurements on specific patients. For sev eral models, this method has been used to generate arrays of virtual patients – called cohorts – that can be used to represent the interpatient-variability that may exist within a particular pathophysiology . When used for internal model type control schemes, the ability or inability to identify these indi vidualized parameters from readily av ailable data frequently dictates whether a simulation model is useful for real-time control. The av ailability of such data may also dictate the en vironments in which it is possible to implement certain control schemes. This is of particular concern when adaptation/tuning or online identification is required, as some diagnostic tools may only be accessible, or practical, in a clinical setting. While many physiological models have been studied, sev eral, such as the Bergman minimal model [59], [60] , UV a/Pado va simulator [61] , and Cambridge model [62] rep- resent the overwhelming majority of simulation studies using virtual patients. 1) Minimal Glucor e gulatory Models: While any glucoreg- ulatory or subsystem model that depicts the essential dynamics while using the fewest possible states may be considered minimal [56], [63] , the Bergman minimal model (BMM) is by-far the most well-known and frequently adapted. Despite simplifications and neglected dynamics, the parameters and variables in minimal models must both con vey clear physio- logical significance and be readily identified from measurable clinical data [63] . Notice, this requisite simplicity and iden- tifiability are often shared with the kinds of models that are sought in controller synthesis. Hence, in addition to its primary analytical purpose, the BMM has also been adapted for use in the implementation of closed-loop blood glucose control strategies. T o understand the importance of this model, it is interesting to consider its origin. Initially dev eloped as a means to quan- tify the insulin sensiti vity of individual patients [59] , the BMM was the result of early experiments to identify the structure of blood glucose-insulin interactions by obtaining frequent measurements of both plasma glucose and plasma insulin concentrations through the duration of a 1 hour intrav enous glucose tolerance test (IVGTT) applied following a single bo- lus injection of glucose. Blood samples were taken first before injection, to obtain basal le vels, and following injection at specific intervals (i.e., rapidly at first but reducing in frequency ov er the hour). The results of these blood tests were fitted by nonlinear least squares to various pre-established nonlinear ODE model candidates, which were e valuated according to the number and physiological meaning of model parameters, as well as the accuracy of the parameter estimates and each model’ s o verall goodness-of-fit. T wo of 7 possible models (one linear , the other nonlinear) passed this initial testing, both of which included insulin-dependent glucose utilization - 3 of the models were insulin-independent and were rejected for their poor prediction of the glucose kinetics. Ultimately , the nonlinear model was found to reproduce known physiological responses with higher fidelity . Thus, the BMM was selected as the 3-compartment nonlinear system of ODEs with 5 parameters shown schematically in Fig. 4 [59] . Notice, this model has 1 glucose compartment and 2 insulin compartments (one plasma compartment and one remote compartment). Since the time of this original model, many adaptations hav e been made to account for additional dynamics, such as the connection of a second glucose compartment, the addition of glucagon feedback, and the replacement of IVGTT by oral glucose tolerance testing (OGTT) to obtain a more physiological measure of insulin sensiti vity [64] . Howe ver , ev en with these augmentations, the BMM must be further adjusted to be useful for closed-loop control. For example, one prev alent control-oriented model intended for T1DM control that utilizes the BMM is detailed in ref. [65] . This model augments the original BMM with insulin-dependent glucose dynamics, renal glucose clearance, meal absorption, external insulin infusion and subcutaneous transport, circadian variations, and an adjusted hepatic balance. While many of the equations in this model are linear first- order ODEs (e.g., all of the insulin and the meal absorption dynamics), there are sev eral nonlinearities including circa- 𝐼 𝑡 𝑘 6 𝑘 1 𝑘 5 𝑘 4 𝑘 3 𝑘 2 𝐼′ 𝑡 𝐺 𝑡 Liv e r P e r i p her y ሶ 𝑋 = − 𝑘 3 𝑋 + 𝑘 2 𝑘 4 + 𝑘 6 𝐼 ሶ 𝐺 = − 𝑘 1 + 𝑘 5 + 𝑋 𝐺 + 𝐵 0 Pla s ma In s ulin Re mo te In s uli n P l a s ma G l uc o s e 𝑋 = 𝑘 4 + 𝑘 6 𝐼′ 𝐵 0 : H e p a ti c G l uc o s e P r o d uc ti o n a t 0 P l a s ma G l uc o s e C o n c e n tr a ti o n Fig. 4. Original Bergman minimal model of blood glucose/insulin dynamics. dian variations in insulin sensiti vity in the interstitial glucose compartment and hard nonlinearities in the endogenous blood glucose production dynamics. Further , any effects caused by glucagon or physical activity have been excluded. Note, the original model also includes intrav enous routes for insulin and glucose, which have been omitted abo ve. Due to its questionable accuracy and remaining complexity , the BMM is not alw ays the first choice for model-based closed-loop control. Howe ver , it is frequently adapted for this purpose, and has provided an important clinical tool for establishing an individual’ s lev el of β -cell function through the insulin sensitivity index [60], [64], [66] . Like the BMM, other models used in controller design for closed-loop insulin therapy generally trade physiological complexity for model structure, identifiable parameters, and appropriate input-output behaviors – typically by linearization. This trade-off with tradi- tional physiological models is – at least partially – responsible for the increasing role of data-driven models for personalized control of diabetes [67] . 2) UV a/P adova Models and Simulations: Since the time of the BMM, se veral detailed models ha ve been dev eloped, or modified, to analyze the glucoregulatory behavior of diabetic patients. Ho wever , to date, there is only one family of models that has been approved by the United States Food and Drug Administration (FD A) as a replacement for pre-clinical trials in the v alidation of a closed-loop control design. These are the so- called UV a/Padov a models [68] de veloped primarily between collaborators at the Univ ersity of V irginia in the USA and the Univ ersity of Padov a in Italy . In contrast to the simplicity of minimal models, the UV a/Pado va model is a complex and hierarchically organized model that integrates high and low-le vel details into submod- els of organ systems and metabolic pathways, to obtain a comprehensiv e, physiological simulation of human patients [69] . This model, sometimes referred to as a maximal model due to its maximal level of detail [56] , contains over 30 parameters [58] that must be identified to match a specific patient and is generally too complex to produce desirable controllers without simplification. Conv ersely , this complex model can accurately reproduce many aspects of the glu- coregulatory dynamics of healthy and diabetic individuals, which has led to the dev elopment of indi vidualized patient simulation models kno wn as in silico subjects. Using high quality clinical data, researchers hav e dev eloped a large collec- tion of patient models representing numerous individuals from each of v arious demographics, enabling studies on the entire population as well as particular cohorts of interest [61], [68], [69] . Such detailed high-fidelity modeling is the foundation for an emerging symbiotic relationship, wherein simulations are not only useful for analyzing the ef fects of treatments on known (patho)physiology , but also for testing hypotheses regarding the (patho)physiology itself and models thereof [56] . Presently , a distributed version of the UV a/Padov a type 1 diabetes metabolic simulator (T1DMS) for use in MA T - LAB/Simulink can be obtained by academic and commercial entities through The Epsilon Group [70] . In addition to its early capabilities, the current version of the UV a/Padov a simulator includes dynamic models of the bi-hormonal (i.e., insulin and glucagon) glucoregulatory dynamics, an improved patient cohort, and updated depiction of regulatory feedback during hypoglycemia [61], [70] . As of 2018, researchers from UV a/Pado va ha ve announced further updates to their simula- tion platform that introduce, among various other extensions and improvements, a set of time-varying patient models to account for diurnal variations in patient dynamics associated with o vernight rise in blood glucose (i.e., the da wn phenom- ena) and patterns of varying insulin sensiti vity at morning, afternoon, and ev ening meals [71] . While the previous updates [61] are FDA approved, approv al for the more recent time- varying simulator [71] is still pending at the time of writing this article. The dev elopment of the UV a/Padov a model is well doc- umented, and interested readers are referred to the relev ant literature for extensi ve details on the underlying model, in silico population, recent updates, and application to the val- idation of control algorithms. Note, despite its comple xity , sev eral closed-loop control algorithms have been designed using reduced order approximations of these models [55], [72], [73] . 3) Cambridge (Hovorka) Models: Similar to the UV a/Pado va model, the Cambridge model [62], [74], [75] has frequently been used for in silico validation studies of closed- loop insulin systems and has its own MA TLAB/Simulink based simulation en vironment and cohort of 18 clinically validated virtual subjects [55], [62] . Ho wev er, unlike the UV a/Pado va simulator, results from this softw are are not FD A appro ved substitutes for pre-clinical trials. While this may lead to sev eral additional steps prior to clinical studies, the Cambridge model has significantly fe wer mathematical states and parameters than the UV a/Pado va model [58] , while maintaining physiological significance and modeling diurnal variations, which has allo wed some researchers to modify and adapt it more readily for their specific needs [76] . Despite its complexity , the Cambridge model is frequently used in internal model or model predictiv e control schemes [53], [74] and has been the basis for several clinically tested in vestigatory closed-loop de vices [75] . 4) Alternative and Data-Driven Models: In addition to the more analytical and physiologically guided virtual patient models, numerous simplified control-oriented models have been proposed that attempt to leverage various modeling paradigms. Of particular interest, the use of linear parameter varying (LPV) type models [77] has been shown to provide a simplified means to accurately reproduce the complex time- varying and nonlinear dynamics of maximal diabetes models. Using appropriate techniques, reduction of maximal models to LPV models may provide a superior model-based and control- oriented approach to blood glucose controller synthesis. Noting that even the best physiological blood-glucose mod- els are, at present, phenomenological and difficult to in- dividualize for optimal real-time control, many researchers hav e instead adopted data-driven modeling paradigms such as autoregressi ve exogenous (ARX) and autoregressi ve mov- ing average exogenous (ARMAX) models. Provided with a predefined model structure, these techniques utilize online system identification to obtain and, potentially , update models to achieve a best approximation of the input-output behaviors of the glucose-insulin system based on a giv en performance index [67] . Like LPV models, it is unlikely that data-driven models will maintain any detailed physiological information about a giv en patient, but instead provide a useful foundation for implementing established controller synthesis approaches. 5) Models of Ther apeutic Agents and CGM Sensor s: Follo wing previous discussions on sensors and treatment ap- proaches, it is natural that these components will contrib ute dynamics to the ov erall system. Models for CGM sensor accuracy [78] and insulin formulations and kinetics [79], [80] hav e been developed by various in vestigators. These models enable simulations that provide a complete picture of blood glucose control under specific therapy strategies and allow the in vestigation of closed-loop systems while taking sensor and actuator performance limitations into account. D. Automated Therapy for Diabetes Attempts to de velop fully closed-loop insulin deliv ery sys- tems date back more than 50 years [81] , and many companies and academic researchers hav e contributed significantly in these de velopments [82] . While early work focused on sugar and insulin infusion by intrav enous methods, the success of these systems along with the dev elopment of both CSII pumps and CGM sensors has shifted the modern focus to ward minimally in v asive therapies applied via subcutaneous routes [83] . Within the last 5 years, there has been – and continues to be – rapid progress tow ard the commercialization of fully auto- mated insulin therapy systems [84], [85] , frequently referred to as artificial β -cell, artificial pancreas (AP), or automated insulin deli very (AID) systems. The primary goal of these automated therapy devices is to alleviate the burden and risks associated with multiple daily injections from T1DM patients and caregi vers by reducing diabetes treatment to a combination of proper lifestyle modifications (perhaps even less stringent ones once on AP therapy) and device maintenance (to ensure proper AP function and patient safety). Although most current work is focused on T1DM, there are parallel efforts to in vesti- gate the use of these systems for specific cases such as insulin dependent T2DM [86], [87] , T1DM during pregnanc y [88], [89] (but not yet GDM as far as we are aw are), and blood glucose regulation for patients in intensi ve care units (ICUs) [90] . 1) Challenges to Automatic Contr ol: While the virtual patient models discussed previously can reproduce many as- pects of normal and diabetic blood glucose regulation, there remain several challenges that prev ent these models from fully predicting the dynamics of real patients in real-time and limit the accuracy of designs for real-world control performance. The most notable among these challenges are the blood glucose disturbances caused by meals and exercise and the intrapersonal variations – particularly in insulin sensitivity and glucose production – that may occur within a single patient ov er their course of treatment. While man y meal absorption and gastric emptying models hav e been proposed to describe how meal contents – generally carbohydrates – act as sources of e xogenous glucose [91] , the timing, duration, and nutritional content of meals and snacks are generally unknown for patients in an uncontrolled, or free li ving, en vironment. As a substitute for such detailed information, many current algorithms resort to manual meal announcement strategies, through which patients or caregiv ers can provide nutrition and schedule information to the con- troller regarding an upcoming meal. Howe ver , even with meal announcement and approximate nutritional content, there may still be unpredicted digestive variations. Thus, some schemes – with or without meal announcement – implement meal detection algorithms that identify when a meal has occurred and may also estimate carbohydrate content [91]–[95] . At the current time, very few controllers can provide tight glycemic control when subjected to unannounced meals as it generally takes around 70 min after administration for peak insulin action to occur [95] . This delay in insulin action can provide a window for hyperglycemia to occur following ev en a well identified meal and may result in either hyperglycemia or hypoglycemia if meal properties are estimated incorrectly . Like meals, periods of physical activity are known to disturb the nominal behavior of the blood glucose re gulatory system. Howe ver , while meals primarily act by regulating exogenous glucose, physical activity can simultaneously affect numerous aspects of a closed-loop therapy system including physiological requirements [96]–[98] , internal bio-mechanical pressure differentials [98], and CGM accuracy [98] . In fact, it is well known that different types, intensities, and durations of physical activity may provok e contrasting physiological responses (e.g., light aerobic ex ercise can cause hypoglycemia and brief stints of intense combined aerobic-anaerobic e xercise can cause prolonged hyperglycemia [85], [96] ). Thus, it is not only important to detect physical activity , but also to obtain enough data to classify – and perhaps continuously reclassify , track, or predict – the kinds of acti vity or exercises a patient performs. Further complicating this problem, certain kinds of physical activity can increase the rate of insulin absorption from the subcutaneous compartment to the blood stream [96], [98] and the corresponding increase in insulin action can cause hypoglycemia without any infusion of additional insulin. As noted previously , insulin does not act immediately and will not reach peak effecti veness for about an hour; thus, even if insulin has been regulated correctly for the current lev el of physical activity , v ariations in physical acti vity can change the internal utilization of the existing insulin dosage and affect hypoglycemic excursions that cannot be corrected by a single-hormone insulin therapy system [96] . T o counteract these effects, many researchers are considering more adv anced control systems that may utilize dual-hormone (i.e., glucagon and insulin) infusion sets [98], [99] or multiple sensors and algorithms to detect and characterize physical activity [92], [96], [100] . Despite these promising developments, there are currently no accurate models for simulations of ex ercise in diabetics [92], [97] , and physical activity remains a difficult challenge for fully automated free-li ving diabetes therapy . Additionally , the effects of many other en vironmental fac- tors and lifestyle behaviors on localized and body-wide dy- namics including glucose production, insulin secretion, and insulin sensitivity are still poorly understood. For example, recent studies have suggested that insulin sensitivity decreases significantly in both healthy [101] and diabetic individuals [102], [103] with reduced duration and quality of sleep. Further , the ef fects of circadian phasing, misalignment, and behaviors (i.e., timing of meals as hours after waking up) were recently shown to produce 15-20% changes in insulin sensitivity and β -cell function in healthy adults [104] . T o the authors’ knowledge, similar results have yet to be reported in diabetics with T1DM or T2DM. Combined with other influ- ential factors (e.g., sensor and physiological interference from outside substances, user error in attachment and maintenance, and electromechanical faults [105] ), any model or controller that has been indi vidualized for a specific patient will almost certainly be subject to significant time-varying uncertainty , depending on patient behaviors and operating environment. While these challenges apply to most diabetics, the e xten- sion of AP systems to T2DM may benefit from updated models that include compartments and inputs reflecting the action of noninsulin medications such as metformin, sulfonylureas, and GLP-1 agonists or additional hormones such as amylin (e.g., by the amylin analog pramlintide), which could also benefit T1DM patients and researchers [106] . Considering the appar- ent importance of glucagon for avoida nce of hypoglycemia during exercise and the potential benefits of amylin for meal- time blood glucose regulation, it is not surprising that there has been recent interest in the de velopment of closed-loop insulin-glucagon-amylin therapy [107], [108] . Howe ver , to the authors’ kno wledge, such complex hormone and combination therapy models are not yet common in closed-loop diabetes therapy de vices b ut may provide significant avenues for system augmentation and a more physiological replication of β -cell function as the relev ant technologies mature. 2) Proportional-Inte gral-Derivative Contr ol Methods: Proportional-Integral-Deri vati ve (PID) control is one of the most v ersatile and commonly applied methods of control for both linear and nonlinear systems and has been likened to the physiological behavior of the pancreatic β -cells [109], [110] . Numerous variations on PID control hav e been studied in simulations of T1DM [111], [112] and clinical trials. Most of the recent clinical studies using PID for closed-loop insulin deliv ery are related to the de velopment of the PID-based ex- ternal physiological insulin delivery (ePID) strategy [1], [110] including the introduction [1], [113] and refinement [114] of insulin-feedback (IFB) to a form near the one currently implemented in the commercially av ailable Medtronic 670G hybrid closed-loop (HCL) therapy system [115] . In addition to the PID-based closed-loop control of basal insulin, these systems provide feedforward control action by meal-time bo- luses in response to manually entered announced carbohydrate loads [115] . Systems that implement this kind of manual, feedforward control are considered hybrid rather than fully closed-loop. Another recent algorithm, kno wn as the safety auxiliary feedback element (SAFE) has been de veloped that implements a two-step approach to augment therapy controllers with safety constraints in order to decouple the problems of achieving nominal control performance and abiding system limitations [116] . This auxiliary algorithm runs in a rapidly sampled (i.e., much faster than the 1 sample/min in the current core control algorithms) outer -loop that uses sliding mode reference conditioning to shape the desired insulin infusion profile to av oid violating safety constraints without af fecting the nominal performance of the main control scheme. The SAFE algorithm can be used with a variety of controllers and constraints of known, constant relati ve degree, and has been used to enhance the meal-time performance of standard PID [116] and modified ePID algorithms [117] . In addition to these single-hormone systems, PID has also been implemented in sev eral dual-hormone (i.e., glucagon and insulin) systems where it has been used as the control algorithm for glucagon micro-bolusing (in conjunction with a separate insulin control algorithm) as well as the control of both insulin and glucagon [118] . 3) Model Pr edictive Contr ol Methods: For many , model predicti ve control (MPC) has become the standard formulation for insulin delivery algorithms because of its intuitiv e treatment of constraints and ease of modification. Like the many implementations of PID v ariants, numerous MPC algorithms hav e been reported that attempt to leverage new or existing MPC techniques to improve the compatibility of these algorithms with the requirements and constraints of diabetes treatment [82] . Among the more recent de velopments in artificial pancreas systems are the extended [119] , multiple model [82], [94], [120] , run-to-run tuning [82], [121] , zone [122], [123] and periodic-zone [124] , e vent-triggered [125] , and velocity-weighted & velocity-penalized [126] MPC algorithms. Each method of MPC attempts to overcome one or more of the challenges discussed pre viously including practical/safety restrictions [127] , interpatient variability , intrapatient variability , and unannounced disturbances such as meals or e xercise. Some of these methods have already become quite successful and provide important guides for researchers and engineers seeking to refine diabetes control strategies. Safety Considerations: Among the first challenges overcome by AP algorithms were the restrictions imposed by limitations on insulin infusion. In principle there are limitations on both (i) the rate of insulin infusion, for instance due to saturations in pump displacement and the inability to remove insulin from the bloodstream once injected, and (ii) the amount of insulin in a patient’ s bloodstream at any gi ven time. Notice, howe ver , that while these problems are analogous to issues present in other industries, and general tools like anti-windup are readily av ailable, the management of insulin is safety critical and MPC provides a means to design control systems that not only acknowledge these limitations but anticipate and av oid them while providing optimal control action. In fact, even predictiv e infusion shut-off systems [128] , which temporarily halt basal insulin therapy when continued treatment is predicted to cause hypoglycemia, without any further feedback capabilities hav e been shown to drastically improve system safety by decreasing the risk of ov ernight hypoglycemia. Ho wever , the effecti veness of these simple predictive shut-off algorithms can be diminished if the patient already has significant insulin in their subcutaneous or plasma compartments. This point is of particular concern when insulin is infused according to feedback control la ws, which may frequently exceed basal rates. Thus, many recent systems use insulin action curv es to implement estimates of the patient’ s ”insulin-on-board” (IOB) [129] to account for delayed insulin ef fects as part of their predictiv e algorithm and apply infusion rate limits to avoid dangerously aggressiv e profiles when glucose concentrations are low . Similar to IOB estimates, another recent and promising MPC algorithm uses a more accurate estimate of the patient’ s plasma insulin concentration (PIC) to perform online adapta- tion of dosage constraints for individual patients. This method has performed well in tests on the UV a/Pado va simulator without using any meal announcement but has not yet gone to clinical trials [130] . While IOB and PIC estimates can be useful for minimizing unnecessary control actions, the zone MPC approach attempts to resolve this issue by replacing the usual desired blood glucose set-point with a larger region, or zone, of acceptable concentrations [123] . Thus, the objectiv e of the controller is better aligned with the normal physiological regulation of blood glucose to the euglycemic range, rather than a single specific concentration, and control action is only applied inside this range if the predictive algorithm anticipates an excursion. When no excursion is predicted, the basal infusion rate is maintained, greatly reducing unwanted blood glucose v aria- tions and the risk of insulin induced hypoglycemia. V ariations of the zone MPC approach include periodic zone-MPC control [124] , which accounts for diurnal variations in acti vity and reduced hyperglycemic risks ov ernight, and zone MPC with adaptiv e cost functions [126], [131] . Considering the non-negati ve nature of blood glucose/insulin concentrations and infusion rates, the performance limitations of the diabetes control problem, without glucagon, has been likened to recent results regarding fundamental trade-offs limiting the achiev able response in linear positive systems [132] . Despite nonlinearities in the UV a/Padov a simulator, this approach may provide a means to obtain (in the ideal numerical case) the control signal that would gi ve a near ideal combination of safety and performance, which has been suggested as a potential benchmarking tool for simulation studies on AP systems. One recent MPC variant [126] seeks to achie ve this ideal response by applying (i) a ”velocity-weighted” optimization wherein the cost of blood glucose de viations is a function of the rate of change of the measured blood glucose and (ii) a ”velocity-penalization” term in the cost function that penalizes actions that would allow blood glucose to increase (i.e., ha ve a positiv e rate of change) when blood glucose is tending tow ard hyperglycemia. In combination, the velocity-weighting and velocity-penalty MPC scheme acts aggressively against disturbances by switching on the velocity-penalty at the first signs of impending hyperglycemia, as e videnced by elev ated blood glucose levels (above some threshold) with a positi ve rate of change, while av oiding hypoglycemia due to ov er corrections by effecti vely tapering the control action as the blood glucose rate of change decreases. Notice, this combined action is reminiscent of a nonlinear PD control with predictiv e capabilities and appears to provide some benefits of both MPC and PD control strate gies. Model Individualization: Recalling that ev ery patient affected by type 1 diabetes may respond to treatment differently , there is no single model or set of model parameters that can capture ev ery patient’ s physiology with acceptable accuracy . One method to eliminate the uncertainty (and conservatism) associated with the presence of such interpatient v ariability is to generate patient specific models prior to system deployment or during an initial training period. T o this end, research into individualization of models [133]–[136] has been an activ e subject for many years, and remains an open problem in diabetes control. Despite this, individualized models alone may not be suf ficient to account for the intrapatient variations discussed pre viously , and adaptiv e control schemes may provide a more robust answer to both kinds of variability . Adaptiv e Control: Unlike the model individualization tech- niques discussed previously , adaptiv e online identification techniques can be used to overcome both interpatient and intrapatient variability . Clinical uses of online model identi- fication with MPC date back to the transformati ve Adv anced Insulin Infusion using a Control Loop (ADICOL) trials of 2004 [74] , where model parameters were updated at ev ery control interval using Bayesian methods over a predetermined learning window . Other implementations of adaptive control in MPC include the run-to-run tuning methods in [121] , the cost function updating schemes in [126], [131] , and several of the adaptiv e schemes discussed in [137] . Furthermore, generalized predictive control (GPC) algo- rithms [122], [138] hav e been used successfully in both clinical and simulation studies. Like MPC, GPC strategies utilize an internal model to predict future states and determine the next immediate optimal control signal. Ho wev er, in GPC, a simplified model is used for prediction, and this model is updated (i.e., adapted) at e very control interval to minimize the difference between the predicted and actual responses. Recent implementations of GPC in diabetes are reported in [137], [139] using recursi ve time-series models and in [130] using recursiv e predictor-based subspace identification. 4) The Contr ol to Range Method: Similar to the two-step approach taken by the SAFE algorithm [116] , the control-to- range (CTR) method was proposed as a modular two-element strategy consisting of (i) a range correction module (RCM) that operates at a longer (e.g., 1-15 minutes) sampling interval to maintain blood glucose within a specified target range and (ii) a safety supervisor module (SSM) that operates at a much higher sampling rate to predict and prev ent dangerous hypoglycemic excursions [140], [141] . Although this concept may be applied without regard for the control method used, the CTR strategy has de veloped around the use of MPC-based RCM algorithms [141] . 5) Fuzzy-Logic Contr ol Methods: While PID and MPC hav e historically dominated the field of diabetes control, more recently , fuzzy logic controllers [138], [142]–[144] hav e been quite successful in generating AP systems that closely mimic expert clinical decision making in the face of complex en vironmental factors and incomplete information. Among the most prominent fuzzy AP systems used in clinical trials is the MD-Logic system [142], [145] , which includes information on the patient’ s clinical physiology and therapy guidelines obtained from a set of training data to provide fully automated individualized care (without meal announcement) using a combination of control to range and control to target (standard set-point control) methodologies. T reatment decisions are made using past, present, and pro- jected future data (based on trends), forced to abide safety constraints similar to those discussed previously , and applied according to a variable basal infusion plus bolus strate gy . Alternativ ely , another clinically tested fully closed-loop fuzzy-logic controller (without meal announcement) known as the dose safety controller (DSC) [143] eliminates the basal insulin infusion entirely and instead applies a micro-bolus strategy based on quantized sets of blood glucose level, rate (i.e., first time deriv ative of blood glucose), and acceleration (i.e., second time deri vati ve of blood glucose). T o individualize treatment, each patient is giv en an initial ”personalization factor” (PF), which scales the dosage associated with each lev el-rate-accelerate combination, and the PF is adjusted to provide more or less aggressive treatment depending on the occurrence of hypoglycemic e vents. More information on recent updates to the DSC is a vailable online at [146] . 6) Run-to-Run Contr ol: Apart from the more well-known PID, MPC, and fuzzy-logic controllers, a some what unique class of industrial process control techniques known as run- to-run algorithms [147] ha ve been used for a variety of tasks in diabetes care. Run-to-run methods utilize a measurement- based algorithm to improve the effecti veness of a controller between distinct iterations of a repeated process. For diabetes, this process may be the 24-hour/3-meal c ycle [147]–[149] , in which case the control law is updated before the start of each day to account for some intrapatient variability and improv e the rejection of blood glucose disturbances due to meals. Alternativ ely , similar run-to-run methods hav e been used to adjust basal insulin infusion rates in simulated trials [150] and to improv e the accuracy of CGM measurements based on week-to-week recalibration [151] . 7) Alternative Contr ol Methods: While clinical studies ha ve been dominated by PID, MPC, and Fuzzy-Logic controllers (with several using CTR), a v ariety of additional paradigms hav e been studied in academic simulations. These methods include artificial intelligence methods [152] , H ∞ control [55] , linear parameter varying (LPV) control [77] , and sliding mode control [153] . Howe ver , recent clinical studies implementing these techniques are sparse, if available at all. E. Outlook for Automated Diabetes T r eatment The insulin-based treatment of type 1 diabetes is currently in the midst of an international transformation from manual to automated care. While more complete evidence of the benefits and burdens of closed-loop therapy will become a vailable as these systems are adopted, the momentum behind this transi- tion is bolstered by the significant well-known improvements in safety and accuracy of closed-loop control over sensor augmented pump (SAP) therapy and manual multiple daily insulin injection (MDI) approaches during overnight blood glucose regulation. Furthermore, both hybrid closed-loop (e.g., requiring meal announcement) and fully closed-loop treat- ments have demonstrated promising additional impro vements in more general simulated and clinical scenarios including ambulatory and free-living conditions. Since the initiation of modern closed-loop insulin therapy clinical trials with the 2004 ADICOL studies [74] , there hav e been over 100 publications concerning new clinical trials. A collection of av ailable data regarding recent (2004-2016) clin- ical trials obtained from sev eral studies [85], [105] is shown in Fig. 5 . This data was cross-referenced against the AP clinical trial publication database [154] and shows a trend toward the use of MPC in clinical studies. Among the attractive features of MPC is its predictive adherence to complicated constraints, which cannot be offered by PID control or controllers that 0 5 10 15 20 25 PID M P C F u z z y O th e r Fig. 5. Clinical AP trials from 2004-2016 grouped according to controller type. implement reference conditioning such as the SAFE algorithm. Despite interest in MPC, the first and only commercially av ailable hybrid closed-loop system to-date (MiniMed 670G by Medtronic), released in 2016, uses a PID controller with insulin feedback (IFB) [155], [156] . While recent studies regarding the effecti veness and patient reception of this de- vice are promising [155], [157] , many patients expected the device to have a higher degree of autonomy and found the system difficult to wear . Ho we ver , as a larger variety of commercial systems and fully automated AP systems become av ailable over the next few years [158] , patients will be able to select between systems to find one that best meets their needs and expectations. With the widespread adoption of these technologies will come ne w safety concerns and hazards [159] associated with the unfav orable conditions that may be encountered during continued, long-term, free-living use. Early action to preemptiv ely address these obstacles may provide substantial benefits to patient safety , ergonomics, and perception of closed-loop artificial pancreas systems. Such actions might include adoption and dev elopment of standards relev ant to commercial artificial pancreas systems, such as IEC 60601-1-10 [13] , and familiarization with relev ant FDA guidance (e.g., [160], [161] ) and regulatory considerations arising from public discussions (e.g. [9] ). In addition to commercial systems, opensource solutions to the treatment of type 1 diabetes, such as the open artificial pan- creas system (OpenAPS) [162] , have been gaining popularity in recent years. These systems are not regulated by the FDA and must be designed and constructed by diabetic patients themselves. While there is a gro wing repository of source code and community feedback for interested patients, and the results of the higher-end patient-driven studies [163], [164] are impressiv e, the safety and performance of opensource medical systems raise serious concerns. Implemented across a broad and inconsistent scale, these systems are vulnerable to algorithmic, communication, and electromechanical errors, which may arise when devices are combined and used in a manner inconsistent with their design. While some projects, such as OpenAPS provide a thorough reference guide and code that incorporates safety limitations, the essence of opensource implies that there is ine vitably little prev enting the end- user from tampering with these constraints and accidentally creating dangerous de vices. This becomes an issue of further concern when considering the potential for proliferation of improper designs and the estimated 1/3 of OpenAPS users who are children operating systems designed by their parents [165] (ho wever this may not be representativ e of the larger opensource community). The av ailability of simple and reli- able AP systems may help to significantly reduce the number of patients who choose to dev elop their o wn AP systems, and in fact the desire for such systems to come-to-market appears to underlie the goals of OpenAPS. Even with the av ailability of name-brand, FD A certified HCL and fully closed-loop systems, curiosity surrounding their operation – as well as the desire to replicate and improve it – will undoubtedly drive continued interest in opensource solutions. Finally , it is important to keep in mind that poor glycemic control is a consequence of type 1 diabetes and n ot the under- lying cause. As medical practice and technology progress, the artificial pancreas may be able to grow beyond insulin infusion and implement more comprehensi ve and physiologically ap- propriate therapies. Steps in this direction include the ongoing work to facilitate bi- and tri-hormonal systems augmented with glucagon and amylin. Furthermore, there may be benefits to the in vestigation of closed-loop therapies that are capable of af fecting both glucose/insulin concentrations and insulin action (i.e., insulin sensitivity) – perhaps through combination therapy with non-insulin treatments – to provide personalized treatment options for the large and div erse population of diabetics with forms of diabetes other than T1DM. Of course, economic viability , education for clinicians and new users of AP de vices, and research into a cure for diabetes may ultimately affect the impact and longevity of automated AP technology . I V . A U T O M A T E D A N E S T H E S I A The word anesthesia means the loss of sensation and can be defined as the lack of response to and recall of noxious stimuli. The first application of anesthesia was performed by Crawford W illiamson Lang in 1842 using inhaled ether [166] . In clinical settings, anesthesia is induced by the administration of anesthetic drugs into a patient’ s body to achieve localized or general anesthetic effects. In the latter case, the term ”general anesthesia” refers to a lack of movement across the whole body while also maintaining an unconscious state. In general anesthesia, patients may be gi ven two types of anesthetic drugs: (i) inhaled anesthetics (gases or vapor drugs) and (ii) intravenous anesthetics (injection drugs). A concise comparison between these two methods is described well by Nascu: ”Intrav enous medications are given directly into a vein and are commonly used to induce anesthesia, as induction is usu- ally smoother and more rapid than that associated with most of the inhalational agents. Intra venous anesthetics administered as repeated bolus doses or by continuous infusion may also Fig. 6. The three functional components of clinical anesthesia: hypnosis, analgesia and immobility . be used for maintenance of anesthesia, either alone or in combination with inhalational agents. An advantage of inhaled anesthetics is that measuring the difference between inhaled and exhaled concentrations allows an accurate estimation of plasma or brain drug uptake · · · ” [167] . Generally , intra venous anesthesia drugs can be cate gorized into three different classes based on their physiological effects: (i) hypnotic drugs, (ii) analgesic drugs, and (iii) neuromuscular blocking (NMB) drugs. The first kind of anesthetic, called hypnotics, are used to numb the brain, keeping the patient unconscious during surgery . Propofol is currently the most common type of intravenous hypnotic drug due to its fast redistribution and metabolism inside the body , its lack of accumulation in tissues [167] , and its lower chance of negati ve side effects compared to other hypnotic drugs [168] . The second class of anesthetics, called analgesics, attenuate the sensation of pain. Currently , opioid analgesics (e.g. remifen- tanil) are the most common type of analgesic drug. Finally , the third class of anesthetics, known as NMB drugs, interrupt the transmission of nerve impulses in the neuromuscular junction and cause paralysis of the affected skeletal muscles [166] . NMB can be used to facilitate the processes of endotracheal intubation and mechanical ventilation [169], [170] . As shown in Fig. 6 , each of the 3 classes of drugs used in anesthesia contribute, respectively , to achie ving the 3 main tar gets of general anesthesia: (i) hypnosis, or loss of consciousness (due to hypnotic drugs), (ii) analgesia, or loss of sensitivity to pain (due to analgesic drugs), and (iii) immobility (due to NMB drugs). General anesthesia is performed in 3 phases: (i) induction, (ii) maintenance, and (iii) emergence. Induction is most critical phase in anesthesia because all the three types of drugs – hypnotic, analgesic, and NMB – have to be administered at high enough doses to induce all 3 main aspects of anesthesia and allow the anesthesiologist to insert an endotracheal tube for mechanical ventilation to help the anesthetized patient breathe (i.e., endotracheal intubation). Maintenance follo ws the induction phase. In this phase, the anesthesiologist balances anesthesia with the use of anesthetics agents, manages the infusion of intra venous fluids, and regu- lates the administration of dif ferent drugs to maintain hemo- dynamic stability (i.e., stable blood flow), normal function of body organs, and adequate depth of hypnosis [171] . Emergence is the last stage of anesthesia and occurs at the end of surgery when the patient is removed from anesthesia and brought back to normal conditions as drug administration is tapered off. The goal of emergence is the full reversal of neuromuscular blockade and the smooth return of spontaneous ventilation and refle xes while maintaining the stability of hemodynamics and other physiological functions. Thus, the return of consciousness can occur in a safe and comfortable en vironment [171] . A. Monitoring T o achiev e satisfactory anesthesia, the amount and timing of drug infusions must be estimated in each phase during surgery . Sev eral clinical indicators and physiological parameters ef fect the anesthesiologist’ s estimation. Clinical indicators such as heart rate, blood pressure, lacrimation, sweating, and papillary dilatation are monitored during surgery and provide anesthe- siologists valuable information about the anesthetic adequacy [172] . Howe ver , physiological monitors (i.e., electromyogra- phy (EMG), electrocardiography (ECG), blood pressure (BP), electroencephalography (EEG), and oxyhemoglobin saturation (SpO2)), and quantified indexes such as bispectral inde x (BIS), entropy , auditory ev oked potential (AEP) and sur gical stress index (SSI) can provide anesthesiologists more accurate information about a patient’ s status [173] . The monitors and quantified index es corresponding to sev eral important clinical signs and physiological parameters are shown in T able V for reference. Analyzing the electroencephalogram (EEG) is one of the most common approaches for precise ev aluation of anesthetic deliv ery . EEG signals depict the electrical activity in the cerebral cortex and the characteristics of the EEG wav eforms (i.e., frequency and amplitude) v ary by the type of drug that has been used and the amount of its dosage [174] . There are a number of signal monitoring devices such as the Bispectral Index (BIS) monitor, Narcotrend monitor , Cerebral state monitor (CSM), and AEP monitor [175] that measure and quantify the EEG in order to deri ve a surrogate measurement of anesthetic adequacy [176], [177] . Bispectral index (BIS) is the most notable index and is closely related to the le vel of consciousness [178] . Measure- ments of the BIS provide anesthesiologists important infor- mation during anesthesia, and clinical trials hav e shown its potential to increase the patient safety . [179], [180] . The BIS index is scaled between 0 and 100, where a value of 100 T ABLE V M O NI T O RS A N D I N D I CE S F OR C L I NI C A L S I GN S A N D P H Y S IO L O G IC A L PA RA M E T ER S ( A DA P T ED F RO M [ 1 7 3 ] ) . Clinical signs of inadequate general anesthesia Monitors Signal processing Evaluation indices Somatic responses Motor Movement W ithdrawal EMG Observation Power spectral analysis, BIS, Entropy BIS Entropy AEP Consciousness Nociception A wareness Pain EEG Subjectiv e experience Autonomic responses Breathing Breathing pattern change Observation Respiration rate V olume Coherence Cross-power Spectrum CPC(cardiopulmonary coupling) combined with ECG Hemodynamic T achycardia ECG HR V (heart rate variability) RRI (R peak interval) FFT (fast Fourier transform) LFP (low frequency power) HFP (high frequency power) LHR (low/high frequency ratio) DF A (detrended fluctuation algorithm, α index) MSE (multi scale entropy) CI (complex index) SSI (surgical stress index) PVI (pleth variability index) V olume SPO2 PPGA (plethysmograph amplitude) Hypertension BP BPV (blood pressure variability) Sudomotor Sweating Skin conductivity Papillary dilatation Pupillometry Lacrimation Observ ation Hormonal Catecholamines Corticosteroids Blood drawing and lab analysis Fig. 7. Patient states corresponding to BIS indices (adapted from [181], [182] . represents the fully conscious state and value of zero depicts an isoelectric EEG. W ith increasing concentrations of anesthetic drugs, the BIS index decreases [181] . Fig. 7 shows the patient state for ranges of BIS indices along with the corresponding effects on memory of sur gical e vents. Acceptable BIS scores for general anesthesia range from 40-60 [178] . Despite its wide-spread use, recent experimental results have shown that the BIS index is not reliable when patients have been infused with NMB drugs. In one piv otal study [183] , fully awake subjects were sho wn to achiev e BIS scores below 50 (i.e., corresponding with BIS during general anesthesia) when dosed with NMB drugs alone. Furthermore, additional research has suggested that BIS v alues should be interpreted with caution when patients are children [184] , elderly [185] , hav e known neurological disorders, or are taking psychoacti ve medications [186] . Beyond BIS, it has been noted that, ”the use of DoA monitors remains a major controversy in anesthesiol- ogy . Indeed, many factors, such as age, race, gender , low core body temperature, acid-base imbalances, low blood glucose, drugs administered to the patient (e.g. neuromuscular blocking agents), and brain ischaemia, have a significant effect on raw EEG data. Additionally , DoA monitors are limited by their calibration range and inter patient variability in dose response curves... ” [186] . These limitations and interactions can degrade the performance of manual infusion systems and will no- doubt impose challenges to high-quality closed-loop control of anesthesia. Recently , patient state index (PSI) algorithm-based devices have been considered as alternati ves to BIS monitors for their high sensitivity to variations in sedation/hypnosis and uniform response to v arious anesthetic agents [186], [187] . Although the PSI devices hav e some advantages to classical EEG monitors, including reduced sensitivity to noise, the PSI index is more sensitiv e to interpatient v ariability than BIS [188] and may be affected by EMG signals [186], [189] . In general, there remains a need for studies in vestigating the effects of drug combinations and EMG signals on sedation indices. For more detailed information on current challenges and limitations of monitoring during anesthesia, readers are referred to the extensi ve cov erage in the literature (e.g., [186] ). B. Modeling An accurate infusion of anesthesia drugs and continuous monitoring of brain states during each phase can help de- crease the frequency and severity of dosage errors, improve the efficienc y of anesthesia, and reduce surgical costs. As mentioned in Section IV -A , the BIS index is the most com- mon monitoring metric av ailable for displaying a patient’ s anesthesia level. Howe ver , to fully utilize the BIS and other measures for optimal drug dosing, a mathematical model is needed that represents all aspects of anesthesia including the effecti ve physiological parameters and various dynamics (i.e., corresponding to the hypnotic, analgesic, and paralytic states). V arious such models exist and can be categorized as single- input single-output (SISO), multi-input single-output (MISO), or multi-input multi-output (MIMO) models. In what follows, anesthesia models from each of these three classes will be briefly discussed. 1) SISO Model: The pharmacokinetic/pharmacodynamic (PK/PD) model is a compartmental model that illustrates both the drug distribution in the human body and its effect on certain physiological systems. For anesthesia, the pharma- cokinetics are represented by a linear compartmental model that defines the concentration of the administered drug in different parts of human body , while the pharmacodynamics are represented by a static nonlinear function (i.e., Hill curve) relating the resulting drug concentration to the measured effect (e.g., BIS index) [174], [190] . The current PK/PD model is a SISO model with either hyp- notic drugs (e.g., propofol) or analgesic drugs (e.g., remifen- tanil) as the input and either patient consciousness or degree of pain as the output. Such SISO models are used mainly in anesthesia modeling by control engineers and clinicians. As shown in Fig. 8 , the PK/PD model of anesthesia is a compart- mental model with three main compartments and one effect- site compartment where V 1 describes the central compartment including arterial blood, brain, and li ver volumes; V 2 denotes the fast peripheral compartment, which has fast dynamics and contains the well perfused body tissues such as muscles and viscera; V 3 represents the slow peripheral dynamics, which include the dynamics of poorly perfused body tissues like fat and bone; and the effect-site compartment is used to account for the time lag between drug concentration in blood plasma and its effect [191] . Pharmacokinetic Model: A pharmacokinetic model of a drug is a mathematical expression relating the concentration C p ( t ) (mg/ml) of the drug in the blood plasma to the infusion rate of the drug into the central compartment I(t) (mg/min) [178] . Pharmacokinetic compartmental models are deriv ed by establishing balance equations for the amount of drug ( x i in mg) distributed in each compartment. Accordingly , the gov erning equations for the PK model in Fig. 8 are gi ven by ˙ x 1 = I ( t ) V 1 − k 12 x 1 − k 13 x 1 − k 10 x 1 + k 21 x 2 + k 31 x 3 (5) Fig. 8. Patient states corresponding to BIS indices (adapted from [181], [182] . ˙ x 1 = k 12 x 1 − k 21 x 2 (6) ˙ x 1 = k 13 x 1 − k 31 x 3 (7) where the constants k ij ( min − 1 ) indicate the transfer rate of the drug from the i -th compartment to the j -th compartment; the constant k 10 is the rate at which the drug is metabolized; and V 1 is the volume of the first compartment. These equations can be written in state space as ˙ x 1 ˙ x 2 ˙ x 3 = − ( k 12 + k 13 + k 10 ) k 21 k 31 k 12 − k 21 0 k 13 0 − k 31 × x 1 x 2 x 3 + 1 / V 1 0 0 I ( t ) (8) C p = 1 0 0 x 1 x 2 x 3 (9) Then, the transfer function for the PK model from infusion rate I ( t ) to concentration C p ( t ) = x 1 ( t ) is P K ( s ) = C p ( s ) I ( s ) = 1 V 1 ( s + k 21 ) ( s + k 31 ) ( s + p 1 ) ( s + p 2 ) ( s + p 3 ) (10) where ev ery p i is a function of the constants k ij as giv en by the conditions p 1 + p 2 + p 3 = k 10 + k 12 + k 13 + k 21 + k 31 p 1 p 2 + p 1 p 3 + p 2 p 3 = ( k 10 + k 12 ) ( k 21 k 31 ) + k 13 k 21 p 1 p 2 p 3 = k 10 k 21 k 31 (11) The parameters k ij are different for each person and can be calculated as [191] k 10 k 12 k 21 k 31 k 13 = 1 V 1 0 0 0 1 V 1 0 0 0 1 V 1 0 1 V 2 0 0 0 1 V 3 C l 1 C l 2 C l 3 (12) where V i and C l i denote, respectiv ely , the volume and clear - ance rate (i.e., the rate at which the drug is cleared from the body or removed from the central compartment) of the i -th compartment. For propofol, the clearance rate and volume parameters can be obtained as C l 1 C l 2 C l 3 = 0 . 0456 0 0 0 . 0264 0 0 − 0 . 0681 0 0 0 − 0 . 024 0 × w eight heig ht lbm ag e + − 2 . 271 0 . 018 0 . 836 (13) V 1 V 2 V 3 = 0 − 0 . 391 0 [ ag e ] + 4 . 27 39 . 623 238 (14) It should be noted that although the abov e expressions are the most commonly used relations for calculating the values of V i and C l i , there are alternativ e, generally more complex, relations used to calculate these parameters for the infusion of propofol, such as those de veloped by [192] . For the infusion of remifentanil, the PK parameters can be calculated as [193], [194] C l 1 C l 2 C l 3 = − 0 . 0162 0 . 0301 − 0 . 00113 0 . 0191 0 0 ag e lbm + 2 . 197 3 . 254 0 . 1212 (15) V 1 V 2 V 3 = − 0 . 0201 − 0 . 0811 0 0 . 072 0 . 108 0 ag e lbm + 2 . 043 7 . 124 5 . 42 (16) Note, in (13)-(16), the patient’ s height, weight, and age are giv en in units of cm, kg, and years, respectiv ely . Furthermore, the lean body mass (lbm) is calculated differently for male and female patients and can be obtained as [195] lbm m lbm f = m 0 0 f 1 . 1 − 128 1 . 07 − 148 " w eight weig ht heig ht 2 # (17) where m and f are set equal to one and zero, respectiv ely , for male patients and vice versa for female patients. Pharmacodynamic Model: Pharmacodynamic models are used to describe the relationship between the concentration of a drug and its ef fect on the body . As sho wn in Fig. 8 , the general PD model has two parts. The first part is a compartment called the effect-site, introduced by [196] , that represents the lag between drug concentration and its effect. In the case of anesthesia, since the effect site compartment is small in comparison with the central compartment, its effect is assumed to be negligible [197] . In steady-state, the concentration of the effect-site can be related to the plasma concentration by ˙ C e = ˙ x e = k 1 e x 1 − k e 0 x e (18) where k e 0 and k 1 e are constants and x e is the amount of the drug in the ef fect compartment. The rate at which drugs enter and exit this compartment is assumed to be constant and equal to k e 0 (e.g., k 1 e = k e 0 = 0 . 456 for propofol) [191] . For the infusion of remifentanil, k e 0 can be calculated as k e 0 = 0 . 595 − 0 . 007 ( ag e − 40) (19) Consequently , (19) is often used as ˙ C e = k e 0 ( C p − C e ) (20) where C e is the concentration within the ef fect-site compart- ment. Thus, for propofol, the effect site works like a lo w-pass filter that relates the plasma propofol concentration (i.e., in the blood) to the effect-site propofol concentration. The transfer function describing the relation from the plasma concentration to the effect-site concentration is given by C e ( s ) C p ( s ) = k e 0 s + k e 0 (21) The second part of the PD model, shown as the ”nonlinear drug/effect relation” in Fig. 8 , is often comprised of a sigmoid Hill equation E = E 0 − E max C γ e C γ e + E C γ 50 (22) where E 0 indicates the baseline value (obtained from the awak e state without propofol), which is typically set to 100; E max is the maximum effect achiev able by the drug infusion; E C 50 denotes the drug concentration at half maximal effect, which represents the patient’ s sensitivity to the drug and should be measured experimentally; and γ determines slope of the sigmoid curve (i.e., the recepti veness of the patient to the drug) [198] . In (22), the value of the effect E is obtained in terms of the BIS scale. In T able VI , the values of these parameters are shown for a cohort of 12 representativ e patients. These v alues are among the most frequently used for designing and simulating anesthesia control systems. Additional data sets are av ailable in [166] . Although the aforementioned models are used quite fre- quently , they are in need of some refinements. Among other issues, the most critical weaknesses of the models presented thus f ar include a lack of standard parameters for other kinds of hypnotic drugs, important neglected factors such as cardiac output and mode of drug administration (e.g., bolus or infusion), and unmodeled drug interactions [174] . It is also worth noting that the commonly used values in T able II are not representativ e for a variety of important demographics. In fact, in the case of infants and adolescents, different PK/PD models are required, which have been described completely in [199]–[202] . It should be noted that the SISO models discussed above incorporate the infusion of hypnotic and analgesic drugs but do not consider neuromuscular blocking drugs because neu- romuscular blockade has no effecti ve interactions with anes- thetic drugs and opioids. In some studies, separate (dif ferent) SISO models are used to represent the dynamics of NMB [203], [204] . Furthermore, a different PK/PD model structure, comprised of one central compartment (representing both T ABLE VI H I LL E Q UA T I ON C H A RA CT E R I ST I C V A R I AB L E S F O R P RO P OF O L ( A DA P T ED F RO M [ 1 9 8] ) . Patient # Age Height (cm) W eight (kg) Gender E C 50 E 0 E max γ 1 40 163 54 F 6.33 98.80 94.10 2.24 2 36 163 50 F 6.76 98.60 86.00 4.29 3 28 164 52 F 8.44 91.20 80.70 4.1 4 50 163 83 F 6.44 95.90 102.00 2.18 5 28 164 60 M 4.93 94.70 85.30 2.46 6 43 163 59 F 12.10 90.20 147.00 2.42 7 37 187 75 M 8.02 92.00 104.00 2.10 8 38 174 80 F 6.56 95.50 76.40 4.12 9 41 170 70 F 6.15 89.20 63.80 6.89 10 37 167 58 F 13.70 83.10 151.00 1.65 11 42 179 78 M 4.82 91.80 77.90 1.85 12 34 172 58 F 4.95 96.20 90.80 1.84 Mean 38 169 65 F 7.42 93.1 96.6 3 blood and org ans heavily supplied with blood), one peripheral compartment (representing the parts of the body with a lower blood flow), and the effect compartment (representing the PD model) is presented in the literature [205], [206] . Alternativ ely , a second order linear dynamic model is presented for NMB in [207] . 2) MISO Model: While the SISO model in Section IV -B1 can be used separately for both hypnotic and analgesic drugs, it is not capable of fully representing the combined ad- ministration and effects of these drugs. As a work around, some studies have used parallel SISO models of each drug to obtain a simple MISO model. In this arrangement, the W A VCNS index used by the NeuroSense NS-701 (NeuroW ave Systems, Cle veland Heights, USA) monitor appears to be a more suitable measurement index than BIS for measuring the anesthesia le vel because it is obtained through a linear time- in variant relation that does not cause an additional delay in the closed-loop system [208], [209] . The interaction of propofol and remifentanil is described in [210] as E ( v p , v r ) = ( v p + v r + αv p v r ) γ ( v p + v r + αv p v r ) γ + 1 (23) where ν p = ( c p e ) / ( E C p 50 ) , ν r = ( c r e ) / ( E C r 50 ) , and the parameter α (identified from clinical data by [210] ) determines the degree of interaction between propofol and remifentanil. This interaction is clasified as synergistic if α > 0 ; additi ve if α = 0 ; and antagonistic if α < 0 . The parameters E C p 50 and E C r 50 are the propofol and remifentanil concentrations at half maximal effect. As an alternati ve to using the PK/PD models of propofol and remifentanil in parallel, a new MISO W einer model called the parameter parsimonious model (PPM) has recently been intro- duced by [211] . In this model, the effect-site concentrations of propofol and remifentanil (i.e., c p e and c r e , respectively) due to their infusion rates (i.e., u p and u r , respectively) are given in the Lapalce domain as C p e ( s ) = k 1 k 2 k 3 α 3 ( k 1 α + s ) ( k 2 α + s ) ( k 3 α + s ) u p ( s ) (24) C r e ( s ) = l 1 l 2 l 3 η 3 ( l 1 η + s ) ( l 2 η + s ) ( l 3 η + s ) u r ( s ) (25) where α and η are dependent on patient parameters and do not hav e any physiological meaning. The parameters k i and l j are dimensionless constants that were obtained from clinical data by [212] as k 1 = 10 , k 2 = 9 , k 3 = 1 , l 1 = 3 , l 2 = 2 , and l 3 = 1 . The corresponding BIS value z ( t ) is approximated by [211] as z ( t ) = 97 . 7 1 + U ( t ) γ (26) where U ( t ) = µ C p e E C p 50 + C r e E C r 50 (27) In these equations, the parameters µ and γ do not have any physiological meaning and are dependent on each patient. This model can be expressed in state space as [213] ˙ x ( t ) = Ax ( t ) + B u ( t ) (28) C p e ( t ) C r e ( t ) = 0 0 0 0 1 0 0 0 0 0 0 1 x ( t ) (29) U ( t ) = C x ( t ) (30) where A = A p 0 0 A r , B = B p 0 0 B r (31) A p = − k 1 α 0 0 k 2 α − k 2 α 0 0 k 3 α − k 3 α , A r = − l 1 η 0 0 l 2 η − l 2 η 0 0 l 3 η − l 3 η (32) B p = k 1 α 0 0 , B r = l 1 η 0 0 (33) C = h 0 0 µ E C p 50 0 0 1 E C r 50 i (34) The common values for α , γ , µ and η are identified by [214] and are given in T able VII . Although the PPM model is not entirely physiological, it is better suited to model-based control than the general PK/PD model because it has fewer parameters that need to be T ABLE VII C O MM O N P P M PA RA M E T ER V A L UE S ( A DA PT E D F RO M [ 2 1 3 ] ) . Patient # Gender Age Height (cm) W eight (kg) α η γ µ 1 F 56 160 88 0.0667 0.3989 1.7695 2.1502 2 F 48 158 52 0.0874 0.0670 0.9365 4.7014 3 F 51 165 55 0.0693 0.0482 2.8186 1.1700 4 F 56 160 65 0.0590 0.0425 2.7594 1.4077 5 F 64 146 60 0.0489 0.1269 1.5627 1.4171 6 F 59 159 110 0.0677 0.3373 4.1247 1.1444 7 F 29 163 59 0.0737 0.2793 0.7812 0.8986 8 F 45 155 58 0.0860 0.0212 0.9780 1.4203 9 F 51 163 55 0.0701 0.2837 1.0956 1.2164 10 F 32 172 56 0.1041 0.1038 1.2165 1.9085 11 F 68 160 64 0.0343 3.5768 1.7097 2.5451 12 F 50 161 68 0.0467 0.1254 2.4877 1.4884 13 F 68 158 113 0.0687 4.5413 1.0859 2.3951 14 F 70 161 78 0.0774 0.0397 1.4038 1.5460 15 F 73 160 75 0.0995 0.0377 1.3706 2.0485 16 F 34 162 57 0.0929 0.1205 4.5194 1.5565 17 F 43 155 62 0.0811 0.1033 2.1978 2.0338 18 F 66 155 74 0.1336 0.2307 1.0849 1.2061 identified [213] . Ho wev er, both the PK/PD and PPM models hav e been used in many controller design studies. Some im- portant factors for choosing between these models include the av ailability of measurement devices, the type of desired control structure, the types of drugs being administered, and the costs associated with the ov erall design and implementation. In the following sections, some recent studies that utilize these models will be discussed. 3) MIMO Model: While the previous models hav e a single measurable output (e.g., BIS or W A VCNS), there are sev eral other v ariables that can be considered as additional system outputs. T wo MIMO models hav e been reported prev alently in the literature, both of which take the dosage of hypnotic and analgesic drugs as inputs but differ in the quantities used as outputs. When considering MIMO systems with m inputs and n outputs, we can define m × n transfer functions that describe the effects of each input on each output. The first of these MIMO models [215] , which is used less frequently in the literature, includes some clinical metrics like heart rate and blood pressure as system outputs. By assuming the analgesic drugs hav e negligible effects on the BIS value [216] and considering depth of hypnosis (DoH) and blood pressure as the system outputs, this model is gi ven by x B = e − τ B s K B T B s + 1 U B ( s ) (35) x P = e − τ P s K P T P s + 1 U P ( s ) (36) y B = 100 − f B ( x B ( t )) + d B (37) y P = 100 − f P ( x P ( t )) + d P (38) where indexes B and P denote hypnotic and analgesic drugs, respectiv ely; τ B and τ P are initial time delays; K B and K P are defined as drug sensitivities; T B and T P are time-constants representing the response speed of the patient; f B and f P are defined in [217] as nonlinear sensitivity functions; d B and d P are external disturbances; U B ( s ) and U P ( s ) are the Laplace Fig. 9. MIMO Wiener model for the depth of anesthesia (adapted from [218] ). transforms of drug infusion rates; x B and x P are the ef fect- site concentrations of the anesthetic drugs; and y B and y P are the DoH and mean blood pressure, respectively . According to (27), this representation consists of four (i.e., 2 × 2 ) different transfer functions, which are reported in [215] . The second common MIMO model, depicted in Fig. 9 , defines the infusion rates of propofol (i.e., the hypnotic drug) and remifentanil (i.e., the analgesic drug) as the inputs, and the patient’ s depth of hypnosis and degree of pain are used as the system outputs. Thus, the depth of hypnosis and degree of pain are interpreted from BIS and state entropy (SE) measurements, which consequently may be considered alternativ e model outputs. The state-space of this model is similar to the MISO model of (24)-(33); the main difference between these models is the output matrix, which is described as [218] y = y 1 y 2 = y 01 1+ C r e E C r 50 + ρ 1 C p e E C p 50 γ 1 y 02 1+ C r e E C r 50 + ρ 2 C p e E C p 50 γ 2 (39) where y 1 denotes the BIS value; y 2 denotes the SE value; y 01 and y 02 are equal to 97.7 and 91, respectively; and ρ 1 , ρ 2 , γ 1 and γ 2 are parameters that are estimated by clinicians. T ABLE VIII I N CI D E N CE O F AW A R EN E S S D U RI N G S U R GE RY ( AD A PT E D F RO M [ 2 2 1 ] ) . Y ear Sample size A wareness (percent of patients) 1960 656 1.2 1971 120 1.6 1973 200 1.5 1975 490 0.8 1990 1000 0.2 1997-1998 11785 0.15 1993-2000 10811 0.11 2001-2002 19575 0.13 2003 7826 0.18 2004 1238 0.9 2005 150000 0.07 C. Automation in Anesthesia In 2010, 48.3 million surgical operations were performed in the U.S. [219] . Many of these surgeries require gen- eral anesthesia, yet ensuring optimal and on-time infusion of anesthetic drugs remains an important challenge that is complicated by the unique physiology of each patient and numerous known and unknown environmental factors. Further complicating this problem, patients’ anesthetic requirements are known to change during surgery in both predictable and unpredictable ways, and once administered, anesthetics cannot be remo ved. Poor re gulation of anesthetics, such as too little hypnotic or too much analgesic, can result in complications including anesthesia awareness (i.e., unintended consciousness during surgery), pain during surgery , prolonged emergence period, and increased risks of other side effects [220] . As an example of the regularity of these complications, T able VIII shows data collected on the incidence of anesthesia awareness ov er the last half-century . Although the statistics abov e sho w a near steady improv e- ment, there are still many faults occurring during anesthesia administration which may be av oidable by the dev elopment of an automated anesthesia system that improves the admin- istration of anesthetics and mitigates the hazards of system faults. In fact, besides a reduced risk of surgical complications, the use of an automatic control system may provide signifi- cant additional benefits such as a reduction in the cognitive workload of the anesthesiologist (who must be present to supervise any automated system), a decreased volume of drugs administered (which implies a faster and better recovery time of the patient in the post-operative phase), and a more robust performance with fewer ov er- and under-dosing episodes and better rejection of sur gical disturbances. Of course, the main goal is increased safety and comfort for the patient [222] . The advantages of automatic anesthesia systems ov er manual anesthesia ha ve recently been validated by experimental results [223] . Any automated anesthesia system will likely include two main phases: first, the desired depth of anesthesia (DoA) (i.e. desired BIS index) must be reached, and second the DoA needs to be maintained by the application of v arious anesthetic drugs. During the first phase, the system should calculate the sufficient drug dose and regulate its infusion to move from the initial BIS value E 0 of the patient during the induction phase to a final desired BIS value. Note, the determination of a desired DoA means setting final values for all the three types of anesthesia (hypnotic, analgesic, and neuromuscular blocking drugs). Howe ver , to date, most closed-loop studies hav e been focused only on achieving the depth of hypnosis (DoH), which is the most important type of anesthesia, while the analgesic and neuromuscular blocking drugs must still be infused manually by clinicians. During the second phase, the system should infuse the drugs continuously while monitoring the aforementioned phys- iological parameters and clinical signs and also rejecting disturbances that occur during the maintenance phase. The main goal during this phase is to maintain the patients at the final BIS v alue throughout the surgery until anesthesia is stopped during the emergence phase. Control applications in general anesthesia began receiving attention sev eral decades ago [224], [225] from both control engineers and clinicians. Throughout this period, a v ariety of methods hav e been proposed with v arying de grees of automation. As illustrated in Fig. 10 , most systems can be categorized as manual, open-loop feed-forward, or closed-loop controllers based on the approach used to calculate the drug infusion rates in general anesthesia. Manual systems (i.e., without automation), shown in Fig. 10a , are the current standard practice. In these systems, an anesthesiologist first sets the final v alue of the DoA or DoH and then monitors the anesthetic state of the patient and regulates the drug dosages accordingly [226] . The success of this method is highly dependent on the expertise of the individual anesthesiologist and is not reliable in some critical situations (e.g., irregular ev ents during sur gery). In 2003, a large step toward achieving fully automated anesthesia was taken with the introduction of target controlled infusion (TCI) systems [227] . The Diprifusor system was the earliest commercial TCI pump for propofol [228] and since its introduction several other TCI systems have become av ailable from various manufacturers. In fact, dev elopments and modifications to TCI systems has made them a standard among infusion systems for the administration of opioids, propofol, and other anesthetics in clinical practice [228] . Considering our categorization, TCI systems operate as open- loop feed-forward controllers, sho wn in Fig. 10b , which rely on both an anesthesiologist to assess the patient’ s state and the output of a PK/PD model to estimate an exact drug dosage and infusion time. In practice, the anesthesiologist sets a target drug concentration, and the TCI system determines an infusion rate and sends this value to the infusion pump. Although these systems are used in many countries and have some advantages over manual anesthesia systems, the success of TCI still depends heavily on the expertise of the anesthesiologist and does not have its own means of identifying the patient’ s current needs when tuning the infusion rate. Since TCI lacks real-time feedback, its performance depends on the accuracy of the patient model and is susceptible to disturbances caused by surgical stimulation and hypnotic-opioid synergy (i.e., the Fig. 10. Schematic overvie w of drug dosage calculation and infusion used in clinical anesthesia with (a) manual control, (b) open-loop feedforward control, also known as TCI, and (c) closed-loop control system (adapted from [226] ) combined effects of these drugs) [226] . Closed-loop controllers are the most advanced of the 3 categories and consider the individual patient’ s physiological parameters in addition to the clinical metrics (used by anes- thesiologists in manual and TCI approaches) in the process of calculating infusion rates. As mentioned in Section IV -A , such physiological parameters may already be a vailable as the measured output of adv anced monitors that record and display v alues including the patient’ s EMG, EEG, ECG, and BIS. In this case, feedback control is used instead of an anesthesiologist to frequently adjust the infusion profile or target concentration in closed-loop controllers; ho wever , anes- thesiologists are still needed to set the desired DoA or DoH and supervise the patient’ s state via clinical metrics. The result is a reduction in the anesthesiologist’ s workload, potentially helping to avoid problems associated with distractions and fatigue, as well as increased safety for the patient due to continuous monitoring, the possibility of lower administered drug dosages with a faster postoperative recovery , and a reduction in the occurrence of drug-induced side effects [229] . For these reasons, fully automated systems may hav e the potential to one-day outperform manual infusion dosing [230], [231] . T wo dif ferent approaches for implementing closed-loop controllers in anesthesia are shown in Fig. 11 . The first, in Fig. 11a , is based on a scheme that directly identifies the anesthetic Fig. 11. T wo commonly used closed-loop control architectures in anesthesia drug deliv ery with (a) direct control of the infusion rate, and (b) cascaded control of TCI system (adapted from [226] ) infusion rates and sends control signals directly to the infusion pump (e.g. [232] ). The second, in Fig. 11b , continuously adjusts the target v alue for a downstream TCI system, which in turn continuously sets the rate of the infusion pump (e.g. [233], [234] ). The second method can be considered a special case of the first, and so the first (direct) approach is less restrictive and may be preferable for control design as the TCI dynamics can be replaced with an y ef fectiv e algorithm. Generally , automation in anesthesia consists of three main components: a patient model, a measurement system (i.e., sensors or monitors), and a controller . Each of these com- ponents can ha ve significant ef fects on the performance of the other components and the ov erall system. For example, as mathematical models become more accurate and include more detail on variables and ef fective parameters, controllers can be designed to provide higher quality drug infusion. Furthermore, as measurement devices become more accu- rate, individualized physiological model parameters can be identified more accurately and controllers will ha ve access to a higher quality and quantity of relev ant information via feedback and observation - ultimately resulting in improv ed drug infusion. Note, the quality of monitoring directly effects the controller performance in implementation. While Sections IV -A and IV -B hav e focused on model- ing and monitoring, controller design is among the largest remaining challenges to automated anesthesia, and we will now revie w se veral prominent strategies. Many scientists and control engineers ha ve explored various control approaches such as PID, MPC, fuzzy-logic, adaptive, and neural networks as well as hybridizations of these controllers to design and produce automatic anesthesia infusion systems for clinical use following the general structure in Fig. 12 . Some comprehen- siv e revie ws on controller designs can be found in [174], [235]–[238] . While the majority of recent publications in the physio- logical closed-loop control of anesthesia are related to the use of intravenous anesthesia, sev eral closed-loop studies have specifically considered the application of inhalational anesthe- sia [239]–[241] . One benefit of the inhalational approach to anesthesia is that it may be considered safer than intravenous anesthesia because end tidal gas measurements are av ailable Fig. 12. Closed-loop control for drug administration in anesthesia (taken from [178] ) with permission pending. to monitor the drug concentration within the brain and the vessel rich group (VRG) at steady state. Howev er , these sensors provide a poor approximation of the VRG concen- tration during common dynamic situations such as initial uptake and emergence [242] . Thu s , some researchers hav e dev eloped mathematical models to represent these dynamics (e.g., for purposes of concentration estimation) with the aim of in vestigating, by simulation and clinical studies, the closed- loop control of inhalational anesthesia using model predictiv e control [243]–[245] . For detailed information on modeling and closed-loop control of inhalational anesthesia, interested readers are referred to the extensiv e cov erage in the literature (e.g., [246] ). 1) Challenges to Automated Anesthesia: During the design and simulation phases of the controller dev elopment, there are sev eral important constraints that must be taken into account. First, each drug has a maximum ef fective dosage, which means higher doses will not have any greater effect on the patient. Second, because the control signal defines the infusion rate, it cannot have a negati ve value (i.e., the drug cannot be extracted once infused) and must further be within the operating range of av ailable infusion pumps. Third, excessi ve doses infused ov er a short time can shock the human body and may harm organ systems [247] ; this can be av oided by maintaining lower infusion rates. The selection of proper drug infusion rates is determined as a trade-off between the side-effects of rapid infusion and the desire to reach the required BIS v alue within a certain time-frame. In current surgical operations, the optimal time-frame has been reported as around 15 minutes [167] , while shorter periods are frequently obtained in simulation studies found in the literature (e.g., 4-8 minutes in [222] ). Lastly , the designed controller must work in the presence of both interpatient and intrapatient v ariability . In addition to these systematic constraints, different sources of disturbances and noise – such as poor signal quality and surgical stimulations – should be considered in the system modeling and controller design activities. Of these distur- bances, surgical stimulations are among the most challenging to manage and are a subject of current in vestigations by both clinicians and control engineers. As an example of how these stimulations may arise, Fig. 13 sho ws a typical timeline of the variations in BIS index, caused by surgical stimulations, during the time-course of an archetypal surgical procedure. In this figure, stimulus A shows the arousal due to laryn- goscopy/intubation; B represents surgical incision follo wed by Fig. 13. A standard surgical stimulation profile (adapted from [179] ). a period of no surgical stimulation (e.g., waiting for laboratory results); C represents an abrupt stimulus after a period of low lev el stimulation; D represents the onset of continuous normal surgical stimulation; E, F , and G simulate high amplitude short-term stimulations within the sur gical period; and H simulates the withdrawal of stimulation during the closing period [179] . As discussed in Section IV -A , there is currently a need for more accurate, reliable, and robust sensing techniques that provide real-time monitoring of a patient’ s depth of anesthesia (DoA) without reliance on demographic characteristics or undesirable interference from EMG signals, NMB drugs, or interactions with other substances (e.g., opioids affect AEP measurements [248] ). The present limitations to these sensing technologies may represent a fundamental barrier to accurate closed-loop control. Consider , for instance, the recent results regarding NMB drugs and BIS [183] , which ha ve a direct consequence on closed-loop control. According to this study , an anesthetic PCLC de vice can – under the right circumstances – achiev e the desired BIS value without actually reaching the desired DoA (e.g., potentially resulting in anesthesia awareness). Even in the case where DoA is generally well represented by the sensing paradigm, automated anesthesia remains challenged by sensor noise, measurement error , and sensing delays. In the next sections, the most common controllers used in closed-loop control of anesthesia will be discussed, and the recent dev elopments and remaining challenges to each approach are presented. 2) Proportional-Inte gral-Derivative Contr ol: Proportional- integral-deri vati ve (PID) control is one of the most used control approaches in industrial applications due to its simple mathematics, well-known gain tuning methods, and broad capabilities. Sev eral successful clinical and simulation studies of automated anesthesia have been performed using PID based controllers ov er the past two decades such as [227], [249]– [254] . The simulated and experimental results of these studies hav e generally indicated that PID control may hav e some utility in the closed-loop control of anesthesia. One of the most common PID controllers used anesthesia is giv en by the Laplace domain transfer function [222] U ( s ) E ( s ) = k p 1 + 1 sT i + T d s 1 ( T f s + 1) 2 (40) where U ( s ) and E ( s ) are, respectively , the Laplace domain control and error signals; k p is the proportional gain; T i is the integral time-constant; T d is the deri vati ve time-constant; and T f is the time-constant of a second-order filter used to attenuate measurement noise. In the relev ant works, the proposed PID controllers hav e been shown to track desired anesthesia levels (e.g., desired BIS values); howe ver , anesthetists and control engineers agree that this simple PID algorithm suffers from major deficiencies because it is not a robust and adapti ve controller and does not perform well in the presence of noise and physiological variability . As a result, simple PID gives poor disturbance rejection, unw anted oscillations in the BIS response, and can suffer from windup of the integral control term during the induction phase. Therefore, recent studies are trying to address these problems to achie ve comparable performance to other more advanced controllers. For instance, in [255] it is shown that the lo w-pass filter, included in (40), reduces the sensitivity of the deri vati ve term to random changes. Furthermore, introducing zero reference weight for the propor- tional and deriv ativ e control terms (i.e. no reference input for proportional and deriv ativ e parts) results in better disturbance rejection and avoids unwanted oscillation in the BIS response [256] . Other in vestigations hav e shown benefits from applying a tracking time-constant as the geometric mean of the integral and deriv ativ e time-constants of the PID controller [257] and introducing reference shaping (i.e. the desired BIS index is changed from a step input to a specialized profile) [170] and integrator anti-windup [258] to prev ent integrator windup during the induction phase. Some alternative approaches that hav e also been shown to improve the PID controller perfor- mance include using an e vent-based control scheme, which decreases the variations of controller signal and optimally cancels the noise and disturbances, and using an inv ersion- based methodology , which increases patient safety by reducing BIS overshoot and producing a smoother drug infusion rate [259] . From the clinical point of view , event-based control is similar to manual infusion and is more transparent to anesthesiologists because the y can easily monitor the drug infusion profile [229] . Additional challenges to controller design stem from the presence of nonlinearities (i.e., a Hill equation) in the anes- thesia dynamics. One common approach to handle this nonlin- earity is to linearize (22) around the nominal operating point (e.g., a BIS v alue of 50) in the maintenance phase [260], [261] . This linearization conv erts (22) into a constant coefficient k m giv en by k m = − B I S 0 γ 4 E C 50 (41) where B I S 0 is the nominal operating point in the maintenance phase. Howe ver , this approach is not feasible in the induction phase because the lack of a well-defined nominal operating point causes a large ov ershoot in the response due to the integral term of the controller . Recently , a ne w approach has been proposed, which is applicable to both induction and maintenance phases. This approach uses a linearizing filter in series with the in verse of the nonlinear part of the Hill function [256], [262] . Since the structure of PID controllers is generally well- defined, the selection or tuning of controller gains is v ery important. This task is challenging in closed-loop control of anesthesia, since the gains should be tuned according to the physiological parameters of each patient [263], [264] . While trial and error is common in tuning PID controllers [261] , optimization methods, such as genetic algorithms [257], [265], [266] aimed at minimizing the integrated error, can increase the performance significantly [258], [267] . Nev ertheless, the physiological parameters of patients v ary based on age, weight, disease, and type of sur gery being performed, and presently the av ailable patient data is limited and does not adequately depict the physiological parameters of all patients. Therefore, online identification of patient parameters can be useful for improving the controller performance [268] . Additional studies hav e sho wn that the use of gain scheduling techniques may also be beneficial. In this case, the gains should be tuned different for different phases of surgery . During the induction phase, the gains are tuned to follow the set-point with a reasonable setting-time and low ov ershoot. Then, during the maintenance phase, the gains should be tuned for disturbance rejection. Therefore, when DoH or BIS levels reach the target value and have stabilized around the set-point for an adequate time interval (i.e., signaling the end of induction phase) the controller parameters should be switched to those tuned for the maintenance phase [222] . Another important point is that PID controllers are fre- quently used in dif ferent configurations (e.g., PD or PI only) for different dynamics. Howe ver , recent studies have proved that all three parts are necessary in anesthesia control. F or example, in [269] the deriv ativ e part is eliminated to prevent noise amplification, b ut the performance in the induction phase and disturbance rejection in maintenance phase were less satisfactory for this PI controller than for the PID case, which had a shorter settling-time and less undershoot in the BIS response. 3) Model Predictive Contr ol: Model predictive control (MPC) is a robust and optimal control technique used to handle systems with constraints while optimizing the controller action and predicting system outputs for robustness against noise and disturbances. The applicability of MPC and related strategies (e.g., generalized predicti ve control (GPC)) to medical systems – especially in closed-loop control of anesthesia – has been verified by both simulation and clinical results [270]–[273] . The outstanding features of this controller hav e recently en- couraged control engineers to further de velop MPC controllers to address the complex and nonlinear behavior of closed-loop anesthesia control. One cav eat with standard MPC is the complexity of the computations used to solve the online optimization problem, which pre vents its use in real-time anesthesia control. This limitation can be overcome by using a multi-parametric MPC (mp-MPC) technique, which reduces the complexity of the computations by solving an offline optimization problem on an analytical function instead of numerical solutions [274], [275] . Further , to improve rejection of BIS noise and surgical disturbances, MPC has been implemented in combination with adv anced state estimation techniques such as Kalman filtering and moving horizon estimation (MHE). Simulation results sho w that the MHE method has b e tter accuracy and reduced ov ershoot compared to Kalman filtering [276], [277] . Like wise, studies have also shown that the addition of e vent- based input and state output correction features to MPC can increase its robustness against noise and decrease the amount of anesthetic drugs administered [278], [279] . Another strong point of MPC is its ability to handle intrap- atient and interpatient variability through online estimation of pharmacodynamic parameters [245], [280] , estimation of the slope of the linearized Hill equation at each time-step [281], [282] , and the use of offset-free and state output correction strategies [278], [283] . Piecewise linear PK/PD models can be used to address the problem of nonlinearities in the PD model, which represent one of the main challenges in closed-loop control of anes- thesia. This technique defines se veral linear parts in the hill function to achiev e a more accurate linear approximation of this function [198], [283] . Like wise, multiplying the inv erse of the Hill function C e ( t ) = W C 50 E 0 − B I S ( t ) E max − E 0 + B I S ( t ) 1 γ (42) into the input command and the feedback signal is a common approach to cancel the nonlinear part of the anesthesia system [284] . Other techniques such as linear model predicti ve control (LMPC) [285] . switching the control strategy of mp-MPC to apply bolus treatments in critical BIS indexes [277] , extended prediction self-adapting control (EPSA C) [198] , and online identification of time delays [280] can improv e the perfor- mance of closed-loop anesthesia as evidenced by reduced settling-times (i.e., obtaining a faster response), decreased ov ershoot, and the elimination of unwanted oscillations from the BIS response. 4) Adaptive Contr ol: Adaptive controllers are frequently applied to systems with variable or unknown parameters, and implement algorithms that retune or restructure the effecti ve controller during operation (i.e., online) [286], [287] . As mentioned, the PK/PD model of anesthesia is subjected to large (possibly time varying) uncertainties and its coefficients are dif ferent from patient to patient. Thus, adaptiv e controllers show promise for estimating parameters of the PK/PD model and improving the regulation of anesthesia. Adaptiv e controllers that adjust their control action by directly changing controller gains (rather than an internal model) are kno wn as direct adapti ve controllers. One such method of direct adaptiv e control is to implement an adap- tiv e proportional-integral-deri vati ve control algorithm, which allows adaptive control of anesthesia without directly using the PK/PD model [288] . Howe ver , in practice, this con- troller produced undesirable oscillations and its performance was considered unacceptable. Some studies hav e also shown improv ements to the performance of these controllers by considering the nonlinear uncertainties in the PK model and designing nonlinear adaptiv e controllers [289] . Additionally , neural networks hav e been used to improv e the performance of nonlinear adaptive controllers for dynamic systems with complicated uncertainties [290] . In these controllers, states of the physical system remain in nonnegati ve orthant of the state space [291] . The ef fectiveness of this type of controller has been in vestigated by using sev eral experiments and clinical ev aluation [292] . As an alternative to direct adaptiv e control, indirect adaptiv e control uses parameter updating laws to identify unknown parameters in an internal reference model and thereby retune the ef fective controller gains. Se veral methods are used to obtain such control la ws [234], [293], [294] , and studies hav e sho wn that variations between simulated and real-world control performance can be attenuated by using a minimally parameterized model with an online identification strategy (e.g., an Extended Kalman Filter) to design adaptive nonlinear controllers [212] . Other types of adaptiv e feedback controllers hav e also been designed for anesthesia, including model reference adaptiv e controller (MRAC). Ho wev er, MRA C controllers based on standard models do not show significant improvements in per- formance o ver non-adopti ve techniques. Alternati vely , MRA Cs may also be implemented using fractional order models [295] , and the results of studies using fractional order models hav e confirmed the effecti veness and robustness of fractional order model reference adaptiv e control (FOMRA C), which can also compensate for the time delays in the anesthesia system [296] . Similarly , L1-adaptive methods can achiev e faster adaptation than MRA C [297] and ha ve demonstrated good performance and interpatient robustness [298] . In addition, system iden- tification methods can reduce the order of patient models while also improving performance over standard L1-adaptiv e control [299] . Furthermore, L1-adapti ve controllers can be used to ensure the safe switching between human-in-the-loop and automated close-loop modes of operation [300] . 5) Fuzzy-Logic Contr ol: Since the compartmental models of anesthesia are not exact representations of the human body and are subject to variability in system parameters, closed- loop control of anesthesia can benefit from fuzzy modeling and control. In fact, fuzzy-logic has been applied successfully to all three compartments of anesthesia (i.e., hypnosis [301]– [304] , analgesia [305]–[307] and immobility [308], [309] ). Some comprehensiv e revie ws can be found in [173], [310] . The main benefit of using fuzzy-logic for anesthesia is its ability to express patient models by clustering real patient data without any knowledge of the underlying physiology . Most fuzzy anesthesia systems use re gular (i.e., type-1) fuzzy sets, which have members that take crisp values in [0 1]; howe ver , some systems use type-2 fuzzy sets where the membership values of each element are in another fuzzy set in [0 1] [311] . Finding the best membership function (MF) for such fuzzy sets in [0 1] is one of the main problems with using fuzzy models. T o this end, genetic and neural network algorithms are commonly used to improve performance and optimize the type-2 fuzzy sets’ parameters (e.g., footprint of uncertainty (FOU), centroid, and scaling factors) [312]–[315] . T o dev elop fuzzy control laws, sets of rules are collected according to the expert opinions of anesthesiologists regarding the best correctiv e actions to take under sets of circumstances. Then, these sets of rules are implemented with an aim to create a closed-loop system that mimics their expertise at manual infusion [316] . It should be mentioned that ev en with these rules, fuzzy-logic controllers hav e not demonstrated suitable performance without also being combined with genetic and neural network algorithms. Using these algorithms improves the fuzzy controller by allowing adaptation in the presence of interpatient and intrapatient variability [317] . Frequently , fuzzy-logic controllers are changed to so-called self-organized fuzzy-logic controllers that are capable of adapting to changes in the system [318] . Ho wev er , these controllers are known to cause steady-state error when used with bolus type therapy . T o ov ercome this, simple fuzzy- logic can be used during bolus treatment and the controller can be switched to the self-organized fuzzy-logic type once the system is operating near the desired set-point [302] . As mentioned before, genetic algorithm can then be used for optimizing the fuzzy-logic model [310] . The majority of the fuzzy-logic controllers are based on the type-1 set which cannot handle model uncertainties, and controllers that are based on type-1 sets suf fer from steady- state error in the fuzzy estimation of the desired set-points. Howe ver , these problems can be handled by changing to type- 2 fuzzy sets [319] . Moreover , fuzzy neural network controllers can be used to handle uncertainties in anesthesia [320]–[323] , but this approach has limited effects on the type-1 fuzzy sets. So, type-2 fuzzy neural network controllers are used to solve this problem [324] . Furthermore, controllers based on type-2 fuzzy-logic sets can be improved by switching to a self-organized strategy , resulting in self-org anized type- 2 fuzzy-logic controllers, which can compensate for control uncertainties [2] . By adding genetic algorithms to find the best membership function for the self-organized type-2 fuzzy-logic controller , better results can be obtained in noisy (i.e., real) en vironments at the cost of increased error in the noise-free en vironment [311] . As mentioned, type-2 self-organizing fuzzy-logic controllers are ef fective. Howe ver , signal noise and dynamic uncertainties – such as changes in the PD and PK systems – can de grade controller performance [325] . In most cases, merging fuzzy- logic controllers with other control schemes can ameliorate the final results. For instance, hybridization of fuzzy-logic with MPC can help to predict the effects of uncertainties in system [326], [327] . D. Outlook for Automated Anesthesia As mentioned pre viously , there are four dif ferent types of controllers that are commonly used in the closed-loop control of anesthesia: proportional-integral-deri vati ve, model predictiv e, adaptiv e, and fuzzy-logic controllers. Besides these controllers, strategies such as observer -controllers [213] , ro- bust and robust deadbeat controllers [220], [263], [328] , non- linear H-infinity controllers [329] , non-overshooting tracking controller [330] , sliding mode controllers [331], [332] , and other types of nonlinear controllers [333] ha ve been studied for use in anesthesia. Some of these studies hav e employed per - formance metrics such as settling-time, overshoot, undershoot, lowest observ ed BIS value (BIS-NADIR), performance error (PE), median performance error (MDPE), median absolute performance error (MD APE), total v ariation (TV), integrated absolute error (IAE), and WOBBLE (an inde x of response variations over time) to compare their results and controller effecti veness to other control strategies in attempts to demon- strate adv antages ov er other schemes and find the most suitable class of controllers for anesthesia [272], [334]–[336] . From a control systems perspectiv e, most of the controllers discussed have shown adequate performance. Additionally , some important studies recently hav e been done regarding safety systems for closed-loop of anesthesia [337], [338] . Howe ver , due to the safety critical nature of anesthesia and consequently restrictiv e laws, from the clinical point of vie w , no controller has been completely acceptable for use in a fully closed-loop anesthesia system yet. In fact, none of the four controllers discussed are by themselves able to ov ercome the complex problem of anesthesia due to the presence of intra- and inter-patient variability , surgical disturbances, and nonlin- ear dynamics. As recent papers indicate [326], [327], [339] , combinations of these controllers – optimized to leverage their individual strengths – show promise for improving the per- formance of closed-loop anesthesia and achieving acceptable simulation results. The development of automated anesthesia systems capable of achie ving broad international re gulatory approv al may bene- fit from improved physiological models designed to include the interactions of more organ systems, physiological parameters, and clinical signs for an appropriately div erse cohort of sub- jects and surgical scenarios. If such a model further included all three modes of anesthesia, simulation results might provide a higher degree of confidence in the safety and performance of control algorithms at an early stage. Concurrently , it is also apparent that there is a need for control relev ant anesthesia models with reduced model complexity while maintaining the ability to capture intra- and inter-patient variability . The de- velopment of such models will require collaboration between clinicians, anesthesiologists, mathematicians, and control en- gineers. Furthermore, automated anesthesia would also benefit from dev elopments in clinical monitoring aimed at achieving sig- nificant reductions in measurement noise and time-delays as well as the realization of methods to quantify specific aspects of anesthesia. T o this end, quantification of the depth of anesthesia may provide interesting opportunities to inv estigate the use of sensor fusion to dev elop reliable metrics based on multimodal (and perhaps redundant) monitoring. Since the nonlinear pharmacodynamics of anesthesia are dif ficult to model, the dev elopment of methods to measure the drug concentration in the effect compartment may also help to remov e the need for the corresponding nonlinear model equa- tions and introduce a new set-point to complement or replace BIS values. Again considering the shortcomings of current monitoring technologies, it appears that fully autonomous (unsupervised) anesthesia should not be employed, until a reliable real-time measure of DoA is discovered. Under further consideration of the rapidly developing pharmacological and medical fields, the challenge presented by possible as-of-yet unknown drug interactions and patient variability (with respect to DoA metrics) may , in general, pro vide suf ficient cause to opt for moderated levels of automation (LOA) in PCLC anesthesia devices (i.e., alw ays incorporating the anesthesiologist in a supervisory role). Moving forward, research into the optimal combination of control strategies has the potential to introduce controllers that are feasible for real-world use. Of course, any commercial controller for anesthesia should be able to perform properly in the presence of infusion limitations (e.g., the amount, rate, and frequency of drug infusion), intra- and inter-patient variability , noise, disturbances, and nonlinearities, while also meeting standards for safety and re gulatory appro val. V . D I S C U S S I O N A N D F U T U R E O F P H Y S I O L O G I C A L C L O S E D - L O O P C O N T RO L Considering the historical and current inv estigations in PCLCs, including the artificial pancreas and automated anes- thesia, there are se veral common issues that must be han- dled when implementing control on physiological systems. Chief among these are the ev er-present intra- and inter-patient variations that come with age, health, genetics, hormones, medications, physical activity , and a variety of other inherent and environmental factors. The combination of this variability , its man y pathways, and the complex nature of physiology leads to challenges in both controller design and system validation. The presence of interpatient variability , or differences in the responses observ ed between dif ferent patients, introduces the need for controller robustness and a large collection of high quality clinical data for validation. Howe ver , e ven with a large collection of data, there remains a question of patient dynamics that are not represented by the data or hav e not yet been encountered. Furthermore, the presence of intrapatient variability , or changes to the responses observed within a single patient, introduce time-v arying dynamics which may further introduce either parametric or structural uncertainties. The overwhelming potential for plant-model mismatch has led to a wide acceptance of adapti ve techniques in PCLC formulations, used in combination with robust or predictiv e control strategies. Another key issue in PCLC is the presence of strict con- straints on therapy , which often allo w only positi ve valued control actions (e.g., substances like propofol and insulin- analogs cannot be remov ed once injected) and limit both the rates and amplitudes of applied treatments and estimated physiological states. Of course, one of the most important methods in designing systems that combine these requirements with adaptation and robustness are those based on predic- tiv e control approaches. Then, the application of prediction to physiological control places a significant burden on the observers and estimators to provide reliable approximations of constrained physiological quantities. While there are ar- guments for other control methodologies, predictiv e control methods have been gaining momentum in PCLC and are likely to continue as mathematical models of physiological and pharmaceutical systems develop. Changing our focus to a different portion of the feedback loop, the ability to obtain accurate and on-time measurements is of utmost importance for both safety and performance. Thus, the development and av ailability of improved sensing technologies will, in part, dictate the success of PCLCs. Currently , direct access to many measurable physiological states requires highly inv asiv e approaches, which often can- not be justified for long-term or amb ulatory use. Thus, less accurate, and possibly delayed, measurements are being used as surrogates, for instance in diabetes, which may impose significant performance limitations. While weighing the risks of trade-offs in measurement quality vs. in vasi veness provides a short-term solution in some fields, there is an immediate need for improv ed and miniaturized measurement techniques, perhaps by translation from other fields, that provide rapid and accurate results without jeopardizing patient comfort or health. If we consider the case of long-term PCLC solutions, worn by patients for months or years or in free-li ving conditions, other issues arise as well. For instance, many electrochemical sensors limited lifespans. This case is evident in the CGM systems used for artificial pancreas systems, which last at most around 2 weeks prior to replacement and may provide degraded performance at the early and late stages of use. Additional concerns with long-term use include irritation or infections at attachment sites. Further , the unrestricted en vi- ronments presented by free-living conditions may lead to cases of unforeseen sensor failures or errors, similar to the ef fects of physical activity on subcutaneous glucose measurements, wherein physiological redistrib ution of glucose during ex ercise may bias subcutaneous measurements. On the contrary , the FD A recently approved the first fully implantable glucose sensor , which has a 90-day lifespan and operates on fluorescent sensing technology [340] ; new technologies such as this may provide solutions to some of the classical issues with GCM. Finally , the potential for interactions between multiple drugs or between drugs and sensing elements, as in the case of T ylenol and some CGM systems, should be considered, to the extent possible, in future in vestigations. As PCLCs mature and data becomes av ailable, it may be possible to implement such interactions within the in-silico models used for validation. These models have already vastly improved the rate of con- troller dev elopment in the field of diabetes control and been successful in many other areas as well [341] . The addition of PK-PD models for multiple drugs and known drug interactions within these models may help identify risks or possible failure modes in ambulatory use. Relaxing, slightly , the definition of physiological control to include systems with a primary goal of achieving a target drug concentration (rather than physiological effect), there is additional growing interest in applying control theory to regulate the concentration of pharmaceuticals within certain organs/tissues in the human body [285], [342] . While dosage control alone may reduce the effects of variability in pharma- cokinetics and indirectly improv e regulation of physiological variables, it is also possible that such dosage control could be incorporated as a target in future MIMO physiological control systems. Of particular note in this adjacent field are the recent success of feedback control using electrochemical-aptamer- based (E-AB) sensors [343] ) and the development of long- acting bioresponsiv e pharmaceuticals [344] , which implement a form of analog closed-loop control whereby tailored drugs remain inactive in the body until stimulated by biological feedback conditions. Modern interest in physiological and pharmaceutical control hav e helped bring about the de velopment of a new field distinguished as mathematical pharmacology [345] , which focuses specifically on modeling the pharmacokinetic and pharmacodynamic properties of substances. Future work in this field is likely to pro vide direct support to adv anced automation in medical practice in the form of new and refined pharmacological models as well as insights into the classes of models [346] (and therefore control paradigms) that are well matched to a giv en drug-dosage or physiological control problem. Furthermore, the integration of mathematical phar - macology with systems physiology has the potential to entrust modeling and control design activities with the respective experts in each area and alle viate the present bottleneck wherein control design and validation is faced with significant uncertainty in part due to the complexity of physiological and pharmaceutical interactions. Clearly , the de velopment of mathematical models for control design [347] and e valuation remain important areas of research in physiological closed-loop control. Further , while we have mentioned that some methods of control do not require an explicit mathematical model, these methods may be difficult to ev aluate in a computational setting, and considering the risks in volv ed with physiological systems the immediate future of such methods in PCLC – especially with regard to regulatory approv al – is unclear [9] . Of course, the effecti veness of model-based control is itself subject to the appropriateness of the model(s) used for controller design [347] , and a recent revie w [348] has sho wn that many published methods fall short of completely demonstrating their suitability to the proposed use (e.g., controller design, closed-loop performance ev aluation, hardware-in-the-loop testing) in PCLC devices. This problem is exacerbated by the limited data av ailable to many researchers as well as the e xtraordinary range of potential disturbances, interactions, and variability that affect physiological systems. Ongoing work in the area of credibility assessment for computational patient models [348], [349] and the recently published V&V 40 standard for ”assessing credibility of computational modeling” for medical devices [350] from the American Society of Mechanical Engineers (ASME) seek to provide guidance for establishing model credibility based on the specific context of use (COU), model influence in decision making (e.g., selection of infusion rate), and consequences of suboptimal decisions [348] . W ithin this framew ork, researchers are able to qualify their modeling and control results with appropriate rigor and can establish the credibility of models and techniques as new areas of PCLC emerge. This framework may also have significant value for justifying comparisons between control and safety strate gies. W ith all of these ongoing efforts and the promise of future dev elopments, it seems that the broad introduction of commercially av ailable PCLCs is no w imminent, and the coming decades will likely see many breakthroughs in this regard. Thus, pertinent discussions on appropriate regulatory policy are underway [9] , and similar con versations are being held with regard to ethical implementation of automation in medicine [351], [352] . The continuation of these discussions and refinement of regulatory and ethical guidelines is essential to the future of PCLCs, for establishing both methods of safe practice and guidelines for design, and also for well-informed selection and use of novel medical instruments by hospitals and physicians. V I . C O N C L U S I O N In the years to come, closed-loop medical de vices, artificial intelligence, and networked medical instruments [353] will be- gin to emer ge along e xisting medical robotics [354], [355] ; and physiological closed-loop control systems will become visible within the clinical, home-care, and ambulatory en vironments. Ultimately , it is possible that many of these de vices will be linked to an electronic health network, which could maintain critical patient data or ev en allow physicians to monitor and adjust treatment goals remotely . The integration of feedback control and artificial intel- ligence in medical systems has the potential to improv e adherence to prescribed treatment regimens and protocols, rapidly adapt to new or changing therapy strategies, provide access to treatment in remote areas, optimize the utilization of a vailable resources in scenarios with supply limitations, and increase the capacity of the indi vidual clinician in disaster response and humanitarian crises. The consistency of treatment and av ailability of data that follows automation may be a useful tool for ev aluating the performance of certain clinical practices, v alidating new treatment options, creating innovati ve therapies, and enabling the wide-spread av ailability of per- sonalized medicine. These achiev ements, and others yet to be conceiv ed, will not be obtained without navigating substantial challenges but appear within reach during the 21st century . R E F E R E N C E S [1] C. C. Palerm, “Physiologic insulin deliv ery with insulin feedback: a control systems perspective, ” Computer Methods and Progr ams in Biomedicine , vol. 102, no. 2, pp. 130–137, 2011. [2] F . Doctor, C.-H. Syue, Y .-X. Liu, J.-S. Shieh, and R. Iqbal, “T ype-2 fuzzy sets applied to multivariable self-or ganizing fuzzy logic con- trollers for regulating anesthesia, ” Applied Soft Computing , vol. 38, pp. 872–889, 2016. [3] T . Luspay and K. Grigoriadis, “Robust linear parameter-v arying control of blood pressure using vasoactiv e drugs, ” International Journal of Contr ol , vol. 88, no. 10, pp. 2013–2029, 2015. [4] A. E. Gaweda, A. A. Jacobs, G. R. Aronoff, and M. E. Brier , “Model predictiv e control of erythropoietin administration in the anemia of esrd, ” American J ournal of Kidney Diseases , vol. 51, no. 1, pp. 71–79, 2008. [5] J. J. Cunningham, J. S. Brown, R. A. Gatenby , and K. Sta ˇ nkov ´ a, “Op- timal control to dev elop therapeutic strategies for metastatic castrate resistant prostate cancer , ” Journal of Theoretical Biology , vol. 459, pp. 67–78, 2018. [6] A.-H. H. AlOmari, A. V . Savkin, M. Stevens, D. G. Mason, D. L. T imms, R. F . Salamonsen, and N. H. Lovell, “Developments in control systems for rotary left v entricular assist devices for heart failure patients: a revie w , ” Physiological Measurement , vol. 34, no. 1, pp. R1–R27, 2012. [7] A. Petrou, J. Lee, S. Dual, G. Ochsner, M. Meboldt, and M. Schmid Daners, “Standardized comparison of selected physiological controllers for rotary blood pumps: In vitro study , ” Artificial Organs , vol. 42, no. 3, pp. E29–E42, 2018. [8] J. Rinehart, C. Lee, M. Cannesson, and G. Dumont, “Closed-loop fluid resuscitation: robustness against weight and cardiac contractility variations, ” Anesthesia & Analgesia , vol. 117, no. 5, pp. 1110–1118, 2013. [9] B. Parvinian, C. Scully , H. Wiyor , A. Kumar , and S. W eininger , “Regulatory considerations for physiological closed-loop controlled medical devices used for automated critical care: Food and drug administration workshop discussion topics, ” Anesthesia & Analgesia , vol. 126, no. 6, pp. 1916–1925, 2018. [10] Food and Drug Administration, “Food and drug administration mod- ernization act of 1997: modifications to the list of recognized standards, recognition list number: 036, ” pp. 38 910–38 910, 2014. [11] Health Canada, “List of recognized standards for medical devices, ” 2019. [Online]. A vailable: https://www .canada.ca/ en/health- canada/services/drugs- health- products/medical- devices/ standards/list- recognized- standards- medical- devices- guidance.html [12] European Commission, “Commission communication in the framework of the implementation of the council directive 93/42/eec concerning medical devices (publication of titles and references of harmonised standards under union harmonisation legislation) text with eea relev ance, ” pp. 29–61, 2017. [Online]. A vailable: https://eur- le x.europa. eu/legal- content/EN/TXT/PDF/?uri=OJ:C:2017:389:FULL&from=EN [13] “Medical electrical equipment - part 1-10: General requirements for ba- sic safety and essential performance - collateral standard: Requirements for the development of physiologic closed-loop controllers, ” 2007. [14] J. DiStefano III, Dynamic systems biology modeling and simulation . Academic Press, 2015. [15] S. A.-H. Soliman and A.-A. H. Mantawy , Modern optimization tech- niques with applications in electric power systems . Springer Science & Business Media, 2011. [16] E. H. Mamdani and S. Assilian, “ An experiment in linguistic synthesis with a fuzzy logic controller , ” International Journal of Man-Machine Studies , vol. 7, no. 1, pp. 1–13, 1975. [17] T . T akagi and M. Sugeno, “Fuzzy identification of systems and its applications to modeling and control, ” IEEE T ransactions on Systems, Man, and Cybernetics , vol. SMC-15, no. 1, pp. 116–132, 1985. [18] M. Sugeno and G. Kang, “Structure identification of fuzzy model, ” Fuzzy Sets and Systems , vol. 28, no. 1, pp. 15–33, 1988. [19] P . V . R ¨ oder , B. W u, Y . Liu, and W . Han, “Pancreatic regulation of glucose homeostasis, ” Experimental & Molecular Medicine , v ol. 48, no. 3, p. e219, 2016. [20] S. L. Aronof f, K. Berkowitz, B. Shreiner , and L. W ant, “Glucose metabolism and regulation: beyond insulin and glucagon, ” Diabetes Spectrum , vol. 17, no. 3, pp. 183–190, 2004. [21] American Diabetes Association, “2. classification and diagnosis of diabetes: Standards of medical care in diabetes—2018, ” Diabetes Care , vol. 41, no. Supplement 1, pp. S13–S27, 2018. [22] Z. Punthakee, R. Goldenberg, and P . Katz, “Definition, classification and diagnosis of diabetes, prediabetes and metabolic syndrome, ” Cana- dian Journal of Diabetes , vol. 42, pp. S10–S15, 2018. [23] J. Ozougwu, K. Obimba, C. Belonwu, and C. Unakalamba, “The pathogenesis and pathophysiology of type 1 and type 2 diabetes mellitus, ” J ournal of Physiology and P athophysiology , vol. 4, no. 4, pp. 46–57, 2013. [24] L. A. DiMeglio, C. Evans-Molina, and R. A. Oram, “T ype 1 diabetes, ” The Lancet , vol. 391, no. 10138, pp. 2449–2462, 2018. [25] J. V . Stidsen, J. E. Henriksen, M. H. Olsen, R. W . Thomsen, J. S. Nielsen, J. Rungby , S. P . Ulrichsen, K. Berencsi, J. A. Kahlert, S. G. Friborg et al. , “Pathophysiology-based phenotyping in type 2 diabetes: A clinical classification tool, ” Diabetes/Metabolism Researc h and Reviews , vol. 34, no. 5, p. e3005, 2018. [26] E. Ahlqvist, P . Storm, A. K ¨ ar ¨ aj ¨ am ¨ aki, M. Martinell, M. Dorkhan, A. Carlsson, P . V ikman, R. B. Prasad, D. M. Aly , P . Almgren et al. , “Novel subgroups of adult-onset diabetes and their association with outcomes: a data-driven cluster analysis of six variables, ” The Lancet Diabetes & Endocrinology , vol. 6, no. 5, pp. 361–369, 2018. [27] A. Gedebjerg, T . P . Almdal, K. Berencsi, J. Rungby , J. S. Nielsen, D. R. Witte, S. Friborg, I. Brandslund, A. V aag, H. Beck-Nielsen et al. , “Prev alence of micro-and macrov ascular diabetes complications at time of type 2 diabetes diagnosis and associated clinical characteristics: a cross-sectional baseline study of 6958 patients in the danish dd2 cohort, ” Journal of Diabetes and its Complications , vol. 32, no. 1, pp. 34–40, 2018. [28] D. J. W exler , C. E. Powe, L. A. Barbour , T . Buchanan, D. R. Coustan, R. Corcoy , P . Damm, F . Dunne, D. S. Feig, and A. Ferrara, “Research gaps in gestational diabetes mellitus: ex ecutive summary of a national institute of diabetes and digestiv e and kidney diseases workshop, ” Obstetrics & Gynecology , vol. 132, no. 2, pp. 496–505, 2018. [29] American Diabetes Association, “6. glycemic targets: Standards of medical care in diabetes—2018, ” Diabetes Care , vol. 41, no. Supple- ment 1, pp. S55–S64, 2018. [30] ——, “8. pharmacologic approaches to glycemic treatment: standards of medical care in diabetes-2018, ” Diabetes Car e , vol. 41, no. Supple- ment 1, pp. S73–S85, 2018. [31] P . Adolfsson, C. G. Parkin, A. Thomas, and L. G. Krinelke, “Select- ing the appropriate continuous glucose monitoring system-a practical approach, ” European Endocrinology , vol. 14, no. 1, pp. 24–29, 2018. [32] D. B. Sacks, M. Arnold, G. L. Bakris, D. E. Bruns, A. R. Horv ath, M. S. Kirkman, A. Lernmark, B. E. Metzger , and D. M. Nathan, “Guidelines and recommendations for laboratory analysis in the diagnosis and management of diabetes mellitus, ” Clinical Chemistry , vol. 57, no. 6, pp. e1–e47, 2011. [33] L. Leelarathna and E. W ilmot, “Flash forward: a re view of flash glucose monitoring, ” Diabetic Medicine , vol. 35, no. 4, pp. 472–482, 2018. [34] A. W ood, D. O’neal, J. Furler, and E. I. Ekinci, “Continuous glucose monitoring: a revie w of the evidence, opportunities for future use and ongoing challenges, ” Internal medicine journal , vol. 48, no. 5, pp. 499– 508, 2018. [35] Medtronic, “Enlite: Glucose sensor user guide, ” 2014. [On- line]. A v ailable: https://www .medtronicdiabetes.com/sites/default/files/ library/download- library/user - guides/Mp6025676- 2AF1 a pdf.pdf [36] ——, “Guardian sensor (3) performance, ” 2017. [Online]. A vail- able: https://www .medtronicdiabetes.com/sites/default/files/library/ download- library/user - guides/MP6026113- 2AF1DOC A FINAL.pdf [37] ——, “Guardian connect: System user guide, ” 2018. [Online]. A v ail- able: https://www .medtronicdiabetes.com/sites/default/files/library/ download- library/user - guides/guardian- connect- v3 2/us- manual.pdf. [38] ——, “Guardian sensor(3): User guide, ” 2018. [Online]. A vailable: https://www .medtronicdiabetes.com/sites/default/files/ library/download- library/user - guides/Guardian%20Sensor%203% 20User%20Guide%20- %20June- %202018.pdf [39] Dexcom, “User’ s guide: Dexcom G4® platinum continuous glucose monitoring system receiver with share, ” 2017. [Online]. A vailable: https://s3- us- west- 2.amazonaws.com/dexcompdf/LBL012528+Re v+ 004+User’ s+Guide%2- C+G4+PLA TINUM+with+Share+US+W eb+ with+cover .pdf [40] ——, “User’ s guide: De xcom G5® mobile continuous glucose monitoring system, ” 2018. [Online]. A vailable: https://s3- us- west- 2. amazonaws.com/dexcompdf/G5- Mobile- Users- Guide.pdf [41] ——, “User’ s guide: Dexcom G6® continuous glucose monitoring system, ” 2018. [Online]. A vailable: https://s3- us- west- 2.amazonaws. com/dexcompdf/G6- CGM- Users- Guide.pdf [42] R. L. Houlden, L. Berard, J. M. Lakoff, V . W oo, J.-F . Y ale, and D. C. C. P . G. E. Committee, “Diabetes and driving, ” Canadian Journal of Diabetes , vol. 42, pp. S150–S153, 2018. [43] J. R. Castle and W . K. W ard, “ Amperometric glucose sensors: sources of error and potential benefit of redundancy , ” Journal of Diabetes Science and T echnolo gy , vol. 4, no. 1, pp. 221–225, 2010. [44] A. Basu, M. Q. Slama, W . T . Nicholson, L. Langman, T . Peyser , R. Carter, and R. Basu, “Continuous glucose monitor interference with commonly prescribed medications: a pilot study , ” Journal of diabetes science and technology , vol. 11, no. 5, pp. 936–941, 2017. [45] D. Bruen, C. Delaney , L. Florea, and D. Diamond, “Glucose sensing for diabetes monitoring: recent developments, ” Sensors , vol. 17, no. 8, 2017. [46] J. Kim, A. S. Campbell, and J. W ang, “W earable non-inv asiv e epider- mal glucose sensors: A revie w , ” T alanta , vol. 177, pp. 163–170, 2018. [47] S. Chatterjee, K. Khunti, and M. J. Davies, “T ype 2 diabetes, ” The Lancet , vol. 389, no. 10085, pp. 2239–2251, 2017. [48] E. S. T omaro, R. Pippi, E. Reginato, C. Aiello, L. Buratta, C. Mazzeschi, C. Perrone, C. Ranucci, A. Tirimagni, and A. Russo, “Intensiv e lifestyle intervention is particularly advantageous in poorly controlled type 2 diabetes, ” Nutrition, Metabolism and Car diovascular Diseases , vol. 27, no. 8, pp. 688–694, 2017. [49] “4. lifestyle management: Standards of medical care in dia- betes—2018, ” Diabetes Care , vol. 41, no. Supplement 1, pp. S38–S50, 2018. [50] L. E. Moore, “Insulin, ” in Diabetes in Pr egnancy . Springer , 2018, pp. 87–101. [51] J. Pickup, H. Keen, J. Parsons, and K. Alberti, “Continuous subcu- taneous insulin infusion: an approach to achieving normoglycaemia, ” BMJ , vol. 1, no. 6107, pp. 204–207, 1978. [52] P . Pozzilli, T . Battelino, T . Danne, R. Hovorka, P . Jarosz-Chobot, and E. Renard, “Continuous subcutaneous insulin infusion in diabetes: patient populations, safety , efficacy , and pharmacoeconomics, ” Dia- betes/Metabolism Resear ch and Reviews , v ol. 32, no. 1, pp. 21–39, 2016. [53] S. Oviedo, J. V eh ´ ı, R. Calm, and J. Armengol, “ A review of person- alized blood glucose prediction strategies for t1dm patients, ” Inter- national Journal for Numerical Methods in Biomedical Engineering , vol. 33, no. 6, p. e2833, 2017. [54] K. Fritzen, L. Heinemann, and O. Schnell, “Modeling of diabetes and its clinical impact, ” Journal of Diabetes Science and T echnology , vol. 12, no. 5, pp. 976–984, 2018. [55] P . Colmegna and R. S. Pe ˜ na, “ Analysis of three T1DM simulation mod- els for ev aluating robust closed-loop controllers, ” Computer Methods and Progr ams in Biomedicine , vol. 113, no. 1, pp. 371–382, 2014. [56] C. Cobelli, C. Dalla Man, M. G. Pedersen, A. Bertoldo, and G. T offolo, “ Advancing our understanding of the glucose system via modeling: a perspectiv e, ” IEEE T ransactions on Biomedical Engineering , vol. 61, no. 5, pp. 1577–1592, 2014. [57] I. Ajmera, M. Swat, C. Laibe, N. Le Novere, and V . Chelliah, “The impact of mathematical modeling on the understanding of diabetes and related complications, ” CPT : Pharmacometrics & Systems Pharmacol- ogy , vol. 2, no. 7, pp. 1–14, 2013. [58] M. E. W ilinska and R. Hovorka, “Simulation models for in silico testing of closed-loop glucose controllers in type 1 diabetes, ” Drug Discovery T oday: Disease Models , vol. 5, no. 4, pp. 289–298, 2008. [59] R. N. Bergman, Y . Z. Ider, C. R. Bowden, and C. Cobelli, “Quantitative estimation of insulin sensitivity , ” American J ournal of Physiology- Endocrinology And Metabolism , vol. 236, no. 6, p. E667, 1979. [60] R. N. Bergman, “Minimal model: perspective from 2005, ” Hormone Resear ch in P aediatrics , vol. 64, no. Suppl. 3, pp. 8–15, 2005. [61] C. D. Man, F . Micheletto, D. Lv , M. Breton, B. Kov atchev , and C. Cobelli, “The uva/padov a type 1 diabetes simulator: new features, ” Journal of Diabetes Science and T echnology , vol. 8, no. 1, pp. 26–34, 2014. [62] M. E. W ilinska, L. J. Chassin, C. L. Acerini, J. M. Allen, D. B. Dunger , and R. Hovorka, “Simulation en vironment to ev aluate closed- loop insulin delivery systems in type 1 diabetes, ” Journal of Diabetes Science and T echnolo gy , vol. 4, no. 1, pp. 132–144, 2010. [63] C. Cobelli, C. Dalla Man, G. Sparacino, L. Magni, G. De Nicolao, and B. P . Kov atchev , “Diabetes: models, signals, and control, ” IEEE Reviews in Biomedical Engineering , vol. 2, pp. 54–96, 2009. [64] A. Caumo, R. N. Bergman, and C. Cobelli, “Insulin sensitivity from meal tolerance tests in normal subjects: a minimal model index, ” The Journal of Clinical Endocrinology & Metabolism , vol. 85, no. 11, pp. 4396–4402, 2000. [65] P . G. Fabietti, V . Canonico, M. O. Federici, M. M. Benedetti, and E. Sarti, “Control oriented model of insulin and glucose dynamics in type 1 diabetics, ” Medical and Biological Engineering and Computing , vol. 44, no. 1-2, pp. 69–78, 2006. [66] M. Schiavon, C. Dalla Man, Y . C. Kudva, A. Basu, and C. Cobelli, “Quantitativ e estimation of insulin sensiti vity in type 1 diabetic subjects wearing a sensor-augmented insulin pump, ” Diabetes Care , vol. 37, no. 5, pp. 1216–1223, 2014. [67] A. Cinar and K. T urksoy , “Modeling glucose and insulin concentration dynamics, ” in Advances in Artificial P ancreas Systems . Springer , 2018, pp. 33–50. [68] B. P . K ovatche v , M. Breton, C. D. Man, and C. Cobelli, “In silico preclinical trials: A proof of concept in closed-loop control of type 1 diabetes, ” Journal of Diabetes Science and T echnology , vol. 3, no. 1, pp. 44–55, 2009. [69] C. Dalla Man, R. A. Rizza, and C. Cobelli, “Meal simulation model of the glucose-insulin system, ” IEEE Tr ansactions on Biomedical Engineering , vol. 54, no. 10, pp. 1740–1749, 2007. [70] The Epsilon Group, “T1DMS - the epsilon group, ” 2017. [Online]. A vailable: https://tegvir ginia.com/software/t1dms- 2014/ [71] R. V isentin, E. Campos-N ´ a ˜ nez, M. Schiav on, D. Lv , M. V ettoretti, M. Breton, B. P . Kov atchev , C. Dalla Man, and C. Cobelli, “The uva/pado va type 1 diabetes simulator goes from single meal to single day , ” Journal of Diabetes Science and T echnology , vol. 12, no. 2, pp. 273–281, 2018. [72] L. Magni, D. M. Raimondo, C. Dalla Man, G. De Nicolao, B. Ko- vatche v , and C. Cobelli, “Model predictiv e control of glucose concen- tration in type i diabetic patients: An in silico trial, ” Biomedical Signal Pr ocessing and Contr ol , vol. 4, no. 4, pp. 338–346, 2009. [73] G. P . Incremona, M. Messori, C. T off anin, C. Cobelli, and L. Magni, “Model predictive control with integral action for artificial pancreas, ” Contr ol Engineering Practice , vol. 77, pp. 86–94, 2018. [74] R. Ho vorka, V . Canonico, L. J. Chassin, U. Haueter , M. Massi- Benedetti, M. O. Federici, T . R. Pieber, H. C. Schaller, L. Schaupp, and T . V ering, “Nonlinear model predicti ve control of glucose concentration in subjects with type 1 diabetes, ” Physiological Measur ement , vol. 25, no. 4, pp. 905–920, 2004. [75] M. E. W ilinska and R. Hovorka, “Simulation models for in-silico ev aluation of closed-loop insulin delivery systems in type 1 diabetes, ” in Data-driven Modeling for Diabetes . Springer , 2014, pp. 131–149. [76] D. Boiroux, A. K. Duun-Henriksen, S. Schmidt, K. Nørgaard, S. Mads- bad, N. K. Poulsen, H. Madsen, and J. B. Jørgensen, “Overnight glucose control in people with type 1 diabetes, ” Biomedical Signal Pr ocessing and Contr ol , vol. 39, pp. 503–512, 2018. [77] P . Colmegna, R. S ´ anchez-Pe ˜ na, and R. Gondhalekar, “Linear parameter-v arying model to design control laws for an artificial pan- creas, ” Biomedical Signal Pr ocessing and Control , vol. 40, pp. 204– 213, 2018. [78] L. M. Huyett, E. Dassau, H. C. Zisser, and F . J. Doyle III, “Glucose sensor dynamics and the artificial pancreas: The impact of lag on sensor measurement and controller performance, ” IEEE Contr ol Systems , vol. 38, no. 1, pp. 30–46, 2018. [79] G. Nucci and C. Cobelli, “Models of subcutaneous insulin kinetics. a critical revie w , ” Computer methods and pr ograms in biomedicine , vol. 62, no. 3, pp. 249–257, 2000. [80] M. Schiavon, C. Dalla Man, and C. Cobelli, “Modeling subcutaneous absorption of fast-acting insulin in type 1 diabetes, ” IEEE Tr ansactions on Biomedical Engineering , vol. 65, no. 9, pp. 2079–2086, 2018. [81] A. H. Kadish, “ Automation control of blood sugar a servomechanism for glucose monitoring and control, ” ASAIO J ournal , vol. 9, no. 1, pp. 363–367, 1963. [82] C. Cobelli, E. Renard, and B. K ovatche v , “ Artificial pancreas: past, present, future, ” Diabetes , vol. 60, no. 11, pp. 2672–2682, 2011. [83] R. Hovorka, “Closed-loop insulin deliv ery: from bench to clinical practice, ” Nature Reviews Endocrinology , vol. 7, no. 7, pp. 385–395, 2011. [84] A. Cinar, “ Artificial pancreas systems: An introduction to the special issue, ” IEEE Control Systems , vol. 38, no. 1, pp. 26–29, 2018. [85] K. Turksoy , N. Frantz, L. Quinn, M. Dumin, J. Kilkus, B. Hibner, A. Cinar , and E. Littlejohn, “ Automated insulin delivery-the light at the end of the tunnel, ” The Journal of pediatrics , vol. 186, pp. 17–28, 2017. [86] K. Kumareswaran, H. Thabit, L. Leelarathna, K. Caldwell, D. Elleri, J. M. Allen, M. Nodale, M. E. Wilinska, M. L. Evans, and R. Hovorka, “Feasibility of closed-loop insulin deli very in type 2 diabetes: a randomized controlled study , ” Diabetes Car e , vol. 37, no. 5, pp. 1198– 1203, 2014. [87] L. Bally , H. Thabit, S. Hartnell, E. Andereggen, Y . Ruan, M. E. W ilinska, M. L. Evans, M. M. W ertli, A. P . Coll, C. Stettler et al. , “Closed-loop insulin deliv ery for glycemic control in noncritical care, ” New England Journal of Medicine , vol. 379, no. 6, pp. 547–556, 2018. [88] H. R. Murphy , K. Kumareswaran, D. Elleri, J. M. Allen, K. Caldwell, M. Biagioni, D. Simmons, D. B. Dunger , M. Nodale, and M. E. W ilinska, “Safety and efficac y of 24-h closed-loop insulin deliv ery in well-controlled pregnant women with type 1 diabetes: a randomized crossover case series, ” Diabetes Care , vol. 34, no. 12, pp. 2527–2529, 2011. [89] Z. A. Stewart, M. E. W ilinska, S. Hartnell, L. K. O’Neil, G. Rayman, E. M. Scott, K. Barnard, C. Farrington, R. Hovorka, and H. R. Murphy , “Day-and-night closed-loop insulin delivery in a broad population of pregnant women with type 1 diabetes: a randomized controlled crossover trial, ” Diabetes care , vol. 41, no. 7, pp. 1391–1399, 2018. [90] J.-C. Preiser, J. G. Chase, R. Hovorka, J. I. Joseph, J. S. Krinsle y , C. De Block, T . Desaive, L. Foubert, P . Kalfon, and U. Pielmeier , “Glucose control in the icu: a continuing story , ” Journal of Diabetes Science and T echnolo gy , vol. 10, no. 6, pp. 1372–1381, 2016. [91] A. El Fathi, M. R. Smaoui, V . Gingras, B. Boulet, and A. Haidar , “The artificial pancreas and meal control: An ov erview of postprandial glucose regulation in type 1 diabetes, ” IEEE Control Systems , vol. 38, no. 1, pp. 67–85, 2018. [92] A. Cinar and K. Turkso y , “Multivariable control of glucose concentra- tion, ” in Advances in Artificial P ancreas Systems . Springer , 2018, pp. 65–82. [93] C. M. Ramkissoon, P . Herrero, J. Bondia, and J. V ehi, “Unannounced meals in the artificial pancreas: Detection using continuous glucose monitoring, ” Sensors , vol. 18, no. 3, p. 884, 2018. [94] F . Cameron, G. Niemeyer , and B. W . Bequette, “Extended multiple model prediction with application to blood glucose regulation, ” Journal of Process Control , vol. 22, no. 8, pp. 1422–1432, 2012. [95] P . Colmegna, F . Garelli, H. De Battista, and R. S ´ anchez-Pe ˜ na, “ Au- tomatic regulatory control in type 1 diabetes without carbohydrate counting, ” Control Engineering Practice , vol. 74, pp. 22–32, 2018. [96] M. C. Riddell, D. P . Zahariev a, L. Y avelber g, A. Cinar, and V . K. Jamnik, “Exercise and the development of the artificial pancreas: one of the more difficult series of hurdles, ” Journal of Diabetes Science and T echnology , vol. 9, no. 6, pp. 1217–1226, 2015. [97] J. Bondia, S. Romero-V iv o, B. Ricarte, and J. L. Diez, “Insulin estimation and prediction: A revie w of the estimation and prediction of subcutaneous insulin pharmacokinetics in closed-loop glucose control, ” IEEE Control Systems , vol. 38, no. 1, pp. 47–66, 2018. [98] O. Moser, J. Y ardley , and R. Bracken, “Interstitial glucose and physical exercise in type 1 diabetes: Integrative physiology , technology , and the gap in-between, ” Nutrients , vol. 10, no. 1, p. 93, 2018. [99] T . Peters and A. Haidar, “Dual-hormone artificial pancreas: benefits and limitations compared with single-hormone systems, ” Diabetic Medicine , vol. 35, no. 4, pp. 450–459, 2018. [100] L. Y avelberg, D. Zahariev a, A. Cinar, M. C. Riddell, and V . Jamnik, “ A pilot study validating select research-grade and consumer-based wearables throughout a range of dynamic ex ercise intensities in persons with and without type 1 diabetes: A novel approach, ” Journal of Diabetes Science and T echnology , vol. 12, no. 3, pp. 569–576, 2018. [101] O. M. Buxton, M. Pavlov a, E. W . Reid, W . W ang, D. C. Simonson, and G. K. Adler , “Sleep restriction for 1 week reduces insulin sensitivity in healthy men, ” Diabetes , vol. 59, no. 9, pp. 2126–2133, 2010. [102] E. Donga, M. V an Dijk, J. G. V an Dijk, N. R. Biermasz, G.-J. Lammers, K. V an Kralingen, R. P . Hoogma, E. P . Corssmit, and J. A. Romijn, “Partial sleep restriction decreases insulin sensitivity in type 1 diabetes, ” Diabetes Care , vol. 33, no. 7, pp. 1573–1577, 2010. [103] S. S. Farabi, “T ype 1 diabetes and sleep, ” Diabetes Spectrum , vol. 29, no. 1, pp. 10–13, 2016. [104] J. Qian, C. Dalla Man, C. J. Morris, C. Cobelli, and F . A. Scheer, “Differential effects of the circadian system and circadian misalignment on insulin sensitivity and insulin secretion in humans, ” Diabetes, Obesity and Metabolism , vol. 20, no. 10, pp. 2481–2485, 2018. [105] A. Bertachi, C. M. Ramkissoon, J. Bondia, and J. V eh ´ ı, “ Automated blood glucose control in type 1 diabetes: A revie w of progress and challenges, ” Endocrinolog ´ ıa, Diabetes y Nutrici ´ on (English ed.) , vol. 65, no. 3, pp. 172–181, 2018. [106] S. A. W einzimer, J. L. Sherr, E. Cengiz, G. Kim, J. L. Ruiz, L. Carria, G. V oskanyan, A. Roy , and W . V . T amborlane, “Ef fect of pramlintide on prandial glycemic excursions during closed-loop control in adolescents and young adults with type 1 diabetes, ” Diabetes car e , vol. 35, no. 10, pp. 1994–1999, 2012. [107] National Institutes of Health, “Safety auxiliary feedback element for the artificial pancreas in type 1 diabetes, ” 2015. [Online]. A vailable: https://projectreporter .nih.gov/project [108] ——, “Relative efficacy of single-, bi-, and tri-hormonal closed-loop control systems, ” 2015. [Online]. A vailable: https://projectreporter .nih. gov/project [109] G. M. Steil, “ Algorithms for a closed-loop artificial pancreas: the case for proportional-integral-deri vativ e control, ” Journal of Diabetes Science and T echnolo gy , vol. 7, no. 6, pp. 1621–1631, 2013. [110] G. M. Steil, A. E. Panteleon, and K. Rebrin, “Closed-loop insulin deliv ery-the path to physiological glucose control, ” Advanced Drug Delivery Reviews , vol. 56, no. 2, pp. 125–144, 2004. [111] L. M. Huyett, E. Dassau, H. C. Zisser , and F . J. Doyle III, “Design and ev aluation of a robust pid controller for a fully implantable artificial pancreas, ” Industrial & Engineering Chemistry Researc h , vol. 54, no. 42, pp. 10 311–10 321, 2015. [112] T . MohammadRidha, M. Ait-Ahmed, L. Chaillous, M. Krempf, I. Guil- hem, J.-Y . Poirier , and C. H. Moog, “Model free ipid control for glycemia regulation of type-1 diabetes, ” IEEE T ransactions on Biomed- ical Engineering , vol. 65, no. 1, pp. 199–206, 2018. [113] G. M. Steil, C. C. Palerm, N. Kurtz, G. V oskanyan, A. Roy , S. Paz, and F . R. Kandeel, “The effect of insulin feedback on closed loop glucose control, ” The Journal of Clinical Endocrinology & Metabolism , v ol. 96, no. 5, pp. 1402–1408, 2011. [114] J. L. Ruiz, J. L. Sherr , E. Cengiz, L. Carria, A. Roy , G. V oskanyan, W . V . T amborlane, and S. A. W einzimer , “Effect of insulin feedback on closed-loop glucose control: a crossover study , ” Journal of Diabetes Science and T echnolo gy , vol. 6, no. 5, pp. 1123–1130, 2012. [115] M. de Bock, J. Dart, A. Roy , R. Dav ey , W . Soon, C. Berthold, A. Retterath, B. Grosman, N. Kurtz, and E. Davis, “Exploration of the performance of a hybrid closed loop insulin delivery algorithm that includes insulin delivery limits designed to protect against hypo- glycemia, ” Journal of Diabetes Science and T echnology , vol. 11, no. 1, pp. 68–73, 2017. [116] A. Rev ert, F . Garelli, J. Pic ´ o, H. De Battista, P . Rossetti, J. V eh ´ ı, and J. Bondia, “Safety auxiliary feedback element for the artificial pancreas in type 1 diabetes, ” IEEE Tr ansactions on Biomedical Engineering , vol. 60, no. 8, pp. 2113–2122, 2013. [117] E. Fushimi, N. Rosales, H. De Battista, and F . Garelli, “ Artificial pancreas clinical trials: Moving towards closed-loop control using insulin-on-board constraints, ” Biomedical Signal Processing and Con- tr ol , vol. 45, pp. 1–9, 2018. [118] A. Cinar and K. Turkso y , “Dual-hormone (insulin and glucagon) ap systems, ” in Advances in Artificial P ancreas Systems . Springer, 2018, pp. 83–87. [119] F . Cameron, B. W . Bequette, D. M. Wilson, B. A. Buckingham, H. Lee, and G. Niemeyer , “ A closed-loop artificial pancreas based on risk management, ” Journal of Diabetes Science and T echnology , vol. 5, no. 2, pp. 368–379, 2011. [120] G. P . Forlenza, F . M. Cameron, T . T . L y , D. Lam, D. P . Howsmon, N. Baysal, G. Kulina, L. Messer, P . Clinton, and C. Levister , “Fully closed-loop multiple model probabilistic predictiv e controller artificial pancreas performance in adolescents and adults in a supervised hotel setting, ” Diabetes T echnology & Therapeutics , vol. 20, no. 5, pp. 335– 343, 2018. [121] L. Magni, M. Forgione, C. T offanin, C. D. Man, B. Ko vatche v , G. D. Nicolao, and C. Cobelli, “Run-to-run tuning of model predictive control for type 1 diabetes subjects: In silico trial, ” Journal of Diabetes Science and T echnology , vol. 3, no. 5, pp. 1091–1098, 2009. [122] B. W . Bequette, “ Algorithms for a closed-loop artificial pancreas: the case for model predictive control, ” Journal of Diabetes Science and T echnology , vol. 7, no. 6, pp. 1632–1643, 2013. [123] B. Grosman, E. Dassau, H. C. Zisser, L. Jov anovi ˇ c, and F . J. Doyle III, “Zone model predictiv e control: a strategy to minimize hyper-and hypoglycemic events, ” Journal of Diabetes Science and T echnology , vol. 4, no. 4, pp. 961–975, 2010. [124] R. Gondhalekar, E. Dassau, and F . J. Doyle III, “Periodic zone- mpc with asymmetric costs for outpatient-ready safety of an artificial pancreas to treat type 1 diabetes, ” Automatica , vol. 71, pp. 237–246, 2016. [125] A. Chakrabarty , S. Zavitsanou, F . J. Doyle, and E. Dassau, “Event- triggered model predictive control for embedded artificial pancreas systems, ” IEEE T ransactions on Biomedical Engineering , vol. 65, no. 3, pp. 575–586, 2018. [126] R. Gondhalekar , E. Dassau, and F . J. Doyle, “V elocity-weighting & velocity-penalty mpc of an artificial pancreas: Improved safety & performance, ” Automatica , vol. 91, pp. 105–117, 2018. [127] C. T offanin, M. Messori, F . D. Palma, G. D. Nicolao, C. Cobelli, and L. Magni, “ Artificial pancreas: Model predicti ve control design from clinical experience, ” Journal of Diabetes Science and T echnology , vol. 7, no. 6, pp. 1470–1483, 2013. [128] B. Buckingham, E. Cobry , P . Clinton, V . Gage, K. Caswell, E. Kunsel- man, F . Cameron, and H. P . Chase, “Prev enting hypoglycemia using predictiv e alarm algorithms and insulin pump suspension, ” Diabetes T echnology & Therapeutics , vol. 11, no. 2, pp. 93–97, 2009. [129] C. Ellingsen, E. Dassau, H. Zisser , B. Grosman, M. W . Perciv al, L. Jov anovi ˇ c, and F . J. Doyle III, “Safety constraints in an artificial pancreatic β cell: an implementation of model predictiv e control with insulin on board, ” Journal of Diabetes Science and T echnology , vol. 3, no. 3, pp. 536–544, 2009. [130] I. Hajizadeh, M. Rashid, and A. Cinar , “Considering plasma insulin concentrations in adaptiv e model predictiv e control for artificial pan- creas systems, ” in 2018 40th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC) . IEEE, 2018, Conference Proceedings, pp. 4452–4455. [131] D. Shi, E. Dassau, and F . J. Doyle, “ Adaptiv e zone model predictive control of artificial pancreas based on glucose-and velocity-dependent control penalties, ” IEEE T ransactions on Biomedical Engineering , vol. 66, no. 4, pp. 1045–1054, 2018. [132] G. C. Goodwin, A. M. Medioli, D. S. Carrasco, B. R. King, and Y . Fu, “ A fundamental control limitation for linear positiv e systems with application to type 1 diabetes treatment, ” Automatica , vol. 55, pp. 73–77, 2015. [133] M. Messori, C. T offanin, S. Del Favero, G. De Nicolao, C. Cobelli, and L. Magni, “Model individualization for artificial pancreas, ” Computer methods and progr ams in biomedicine , 2016. [134] J. Garcia-Tirado, C. Zuluaga-Bedo ya, and M. D. Breton, “Identifiability analysis of three control-oriented models for use in artificial pancreas systems, ” Journal of Diabetes Science and T echnology , vol. 12, no. 5, pp. 937–952, 2018. [135] U. Fischer, W . Schenk, E. Salzsieder, G. Albrecht, P . Abel, and E.-J. Freyse, “Does physiological blood glucose control require an adaptive control strate gy?” IEEE T ransactions on Biomedical Engineering , no. 8, pp. 575–582, 1987. [136] M. Messori, G. P . Incremona, C. Cobelli, and L. Magni, “Indi vidualized model predicti ve control for the artificial pancreas: In silico evaluation of closed-loop glucose control, ” IEEE Contr ol Systems , vol. 38, no. 1, pp. 86–104, 2018. [137] K. T urksoy and A. Cinar, “ Adaptiv e control of artificial pancreas systems-a review , ” Journal of Healthcare Engineering , vol. 5, no. 1, pp. 1–22, 2014. [138] A. Cinar and K. T urksoy , “V arious control philosophies for ap systems, ” in Advances in Artificial P ancr eas Systems . Springer , 2018, pp. 55–63. [139] K. T urksoy , E. Littlejohn, and A. Cinar , “Multimodule, multivariable artificial pancreas for patients with type 1 diabetes: Regulating glucose concentration under challenging conditions, ” IEEE Control Systems , vol. 38, no. 1, pp. 105–124, 2018. [140] B. Kov atchev , S. Patek, E. Dassau, I. Francis J. Doyle, L. Magni, G. D. Nicolao, C. Cobelli, and J. D. R. F . A. P . Consortium, “Control to range for diabetes: Functionality and modular architecture, ” Journal of Diabetes Science and T echnology , vol. 3, no. 5, pp. 1058–1065, 2009. [141] S. D. Patek, L. Magni, E. Dassau, C. Karvetski, C. T offanin, G. De Nicolao, S. Del Fa vero, M. Breton, C. Dalla Man, and E. Renard, “Modular closed-loop control of diabetes, ” IEEE Tr ansactions on Biomedical Engineering , vol. 59, no. 11, pp. 2986–2999, 2012. [142] E. Atlas, R. Nimri, S. Miller , E. A. Gurmberg, and M. Phillip, “Md- logic artificial pancreas system: A pilot study in adults with type 1 diabetes mellitus, ” Diabetes Care , vol. 33, no. 5, pp. 1072–1076, 2010. [143] R. Mauseth, I. B. Hirsch, J. Bollyk y , R. Kircher , D. Matheson, S. Sanda, and C. Greenbaum, “Use of a ”fuzzy logic” controller in a closed-loop artificial pancreas, ” Diabetes T echnology & Therapeutics , v ol. 15, no. 8, pp. 628–633, 2013. [144] P . Grant, “ A new approach to diabetic control: fuzzy logic and insulin pump technology , ” Medical Engineering & Physics , vol. 29, no. 7, pp. 824–827, 2007. [145] R. Nimri, N. Bratina, O. K ordonouri, M. A vbelj Stefanija, M. Fath, T . Biester, I. Muller, E. Atlas, S. Miller , and A. Fogel, “Md-logic overnight type 1 diabetes control in home settings: A multicentre, multinational, single blind randomized trial, ” Diabetes, Obesity and Metabolism , vol. 19, no. 4, pp. 553–561, 2017. [146] Dose Safety, 2016. [Online]. A vailable: http://www .dosesafety .com/ [147] F . Doyle, B. Srinivasan, and D. Bon vin, “Run-to-run control strategy for diabetes management, ” in Engineering in Medicine and Biology Society , 2001. Pr oceedings of the 23rd Annual International Confer- ence of the IEEE , vol. 4. IEEE, 2001, Conference Proceedings, pp. 3159–3162. [148] H. Zisser , L. Jov anovic, F . Doyle III, P . Ospina, and C. Owens, “Run- to-run control of meal-related insulin dosing, ” Diabetes T echnology & Therapeutics , vol. 7, no. 1, pp. 48–57, 2005. [149] C. Owens, H. Zisser, L. Jovano vic, B. Sriniv asan, D. Bonvin, and F . J. Doyle III, “Run-to-run control of blood glucose concentrations for people with type 1 diabetes mellitus, ” IEEE Tr ansactions on Biomedical Engineering , vol. 53, no. 6, pp. 996–1005, 2006. [150] C. C. Palerm, H. Zisser , L. Jov anovi ˇ c, and F . J. Doyle III, “ A run- to-run control strategy to adjust basal insulin infusion rates in type 1 diabetes, ” Journal of process contr ol , vol. 18, no. 3-4, pp. 258–265, 2008. [151] J. B. Lee, E. Dassau, and F . J. Doyle III, “ A run-to-run approach to enhance continuous glucose monitor accuracy based on continuous wear , ” IF AC-P apersOnLine , vol. 48, no. 20, pp. 237–242, 2015. [152] I. Contreras and J. V ehi, “ Artificial intelligence for diabetes manage- ment and decision support: Literature revie w , ” Journal of Medical Internet Research , vol. 20, no. 5, 2018. [153] S. Ahmad, N. Ahmed, M. Ilyas, and W . Khan, “Super twisting sliding mode control algorithm for developing artificial pancreas in type 1 diabetes patients, ” Biomedical Signal Processing and Control , vol. 38, pp. 200–211, 2017. [154] The Doyle Group, “ Artificial clinical trial database, ” 2015. [Online]. A vailable: https://www .thedoylegroup.org/apdatabase [155] E. Iturralde, M. L. T anenbaum, S. J. Hanes, S. C. Suttiratana, J. M. Ambrosino, T . T . L y , D. M. Maahs, D. Naranjo, N. W alders-Abramson, and S. A. W einzimer, “Expectations and attitudes of individuals with type 1 diabetes after using a hybrid closed loop system, ” The Diabetes Educator , vol. 43, no. 2, pp. 223–232, 2017. [156] V . Foerster and M. Severn, “ A hybrid closed-loop insulin delivery system for the treatment of type 1 diabetes, ” in CADTH Issues in Emer ging Health T echnologies . Canadian Agency for Drugs and T echnologies in Health, 2017. [157] C. Boughton and R. Hovorka, “Is an artificial pancreas (closed-loop system) for type 1 diabetes effectiv e?” Diabetic Medicine , vol. 36, no. 3, pp. 279–286, 2019. [158] S. Trevitt, S. Simpson, and A. W ood, “ Artificial pancreas device systems for the closed-loop control of type 1 diabetes: what systems are in development?” J ournal of Diabetes Science and T echnology , vol. 10, no. 3, pp. 714–723, 2016. [159] C. M. Ramkissoon, B. Aufderheide, B. W . Bequette, and J. V eh ´ ı, “ A revie w of safety and hazards associated with the artificial pancreas, ” IEEE Reviews in Biomedical Engineering , vol. 10, pp. 44–62, 2017. [160] Food and Drug Administration Center for De vices and Radiological Health, “Infusion pumps total product life cycle: Guidance for industry and fda staff, ” 2014. [Online]. A vailable: https://www .fda.gov/media/ 78369/download [161] ——, “Guidance for industry and food and drug administration staff: The content of inv estigational device exemption (ide) and premarket approv al (pma) applications for artificial pancreas device systems, ” 2012. [Online]. A vailable: https://www .fda.gov/media/80644/download [162] D. Lewis, “What is #openaps?” 2018. [Online]. A v ailable: https: //openaps.org/frequently- asked- questions/. [163] D. Lewis, S. Leibrand, and . O. Community , “Real-world use of open source artificial pancreas systems, ” Journal of Diabetes Science and T echnology , vol. 10, no. 6, pp. 1411–1411, 2016. [164] D. Lewis, “Real-world use of open source artificial pancreas systems - poster presented at american diabetes association scientific sessions, ” 2016. [Online]. A vailable: https://openaps.org/2016/06/11/ real- world- use- of- open- source- artificial- pancreas- systems- poster- \ presented- at- american- diabetes- association- scientific- sessions/ [165] ——, “Frequently asked questions, ” 2018. [Online]. A vailable: https://openaps.org/what- is- openaps/ [166] S. Bibian, “ Automation in clinical anesthesia, ” Ph.D. dissertation, Univ ersity of British Columbia, 2006. [167] I. Nas ¸cu, R. Oberdieck, and E. N. Pistikopoulos, “Explicit hybrid model predictiv e control strategies for intrav enous anaesthesia, ” Computers & Chemical Engineering , vol. 106, pp. 814–825, 2017. [168] A. Borgeat, O. Wilder -Smith, M. Saiah, and K. Rifat, “Does propofol hav e an anti-emetic effect?” Anaesthesia and Intensive Car e , vol. 20, no. 2, p. 260, 1992. [169] M. C. McManus, “Neuromuscular blockers in sur gery and intensive care, part 2, ” American Journal of Health-System Pharmacy , vol. 58, no. 24, pp. 2381–2395, 2001. [170] Z. T . Zhusubaliyev , A. Medvede v , and M. M. Silva, “Bifurcation analysis of pid-controlled neuromuscular blockade in closed-loop anes- thesia, ” Journal of Process Contr ol , vol. 25, pp. 152–163, 2015. [171] S. A. Abdulla, “Depth of anaesthesia control techniques and human body models, ” Ph.D. dissertation, University of Southern Queensland, 2012. [172] P . K. Sinha and T . Koshy , “Monitoring devices for measuring the depth of anaesthesia-an overvie w , ” Indian Journal of Anaesthesia , vol. 51, no. 5, p. 365, 2007. [173] J.-Y . Lan, M. F . Abbod, R.-G. Y eh, S.-Z. Fan, and J.-S. Shieh, “Intelligent modeling and control in anesthesia, ” Journal of Medical and Biological Engineering , vol. 32, no. 5, pp. 293–308, 2012. [174] J. J. Chang, S. Syafiie, R. Kamil, and T . A. Lim, “ Automation of anaesthesia: a review on multi variable control, ” Journal of Clinical Monitoring and Computing , vol. 29, no. 2, pp. 231–239, 2015. [175] H. Mantzaridis and G. Kenn y , “ Auditory evok ed potential index: a quantitativ e measure of changes in auditory evok ed potentials during general anaesthesia, ” Anaesthesia , vol. 52, no. 11, pp. 1030–1036, 1997. [176] B. Musizza and S. Ribaric, “Monitoring the depth of anaesthesia, ” Sensors , vol. 10, no. 12, pp. 10 896–10 935, 2010. [177] S. Bibian, C. R. Ries, M. Huzmezan, and G. A. Dumont, “Clinical anesthesia and control engineering: terminology , concepts and issues, ” in Eur opean Control Conference (ECC), 2003 . IEEE, 2003, Confer- ence Proceedings, pp. 2430–2440. [178] J. M. Bailey and W . M. Haddad, “Drug dosing control in clinical pharmacology , ” IEEE Control Systems , vol. 25, no. 2, pp. 35–51, 2005. [179] M. M. Struys, T . De Smet, S. Greenwald, A. R. Absalom, S. Bing ´ e, and E. P . Mortier , “Performance ev aluation of two published closed-loop control systems using bispectral index monitoringa simulation study , ” Anesthesiology: The Journal of the American Society of Anesthesiolo- gists , vol. 100, no. 3, pp. 640–647, 2004. [180] M. M. Struys, H. V ereecke, A. Moerman, E. W . Jensen, D. V erhaeghen, N. De Neve, F . J. Dumortier, and E. P . Mortier , “ Ability of the bis- pectral index, autoregressiv e modelling with exogenous input-derived auditory evoked potentials, and predicted propofol concentrations to measure patient responsi veness during anesthesia with propofol and remifentanil, ” Anesthesiology: The Journal of the American Society of Anesthesiologists , vol. 99, no. 4, pp. 802–812, 2003. [181] T . Zikov , “Monitoring the anesthetic-induced unconsciousness (hypno- sis) using wa velet analysis of the electroencephalogram, ” Thesis, 2002. [182] S. Bibian, C. R. Ries, M. Huzmezan, and G. Dumont, “Introduction to automated drug delivery in clinical anesthesia, ” European Journal of Contr ol , vol. 11, no. 6, pp. 535–557, 2005. [183] P . Schuller, S. Newell, P . Strickland, and J. Barry , “Response of bispectral index to neuromuscular block in awake volunteers, ” British Journal of Anaesthesia , vol. 115, no. suppl 1, pp. i95–i103, 2015. [184] S. Malviya, T . V oepel-Lewis, A. R. T ait, M. F . W atcha, S. Sadhasivam, and R. H. Friesen, “Effect of age and sedative agent on the accuracy of bispectral index in detecting depth of sedation in children, ” P ediatrics , vol. 120, no. 3, pp. e461–e470, 2007. [185] K. Y amashita, Y . T erao, C. Inadomi, M. T akada, M. Fukusaki, and K. Sumikawa, “ Age-dependent relationship between bispectral index and sedation lev el, ” Journal of Clinical Anesthesia , vol. 20, no. 7, pp. 492–495, 2008. [186] M. Cascella, “Mechanisms underlying brain monitoring during anes- thesia: limitations, possible improvements, and perspectiv es, ” K orean Journal of Anesthesiology , vol. 69, no. 2, p. 113, 2016. [187] L. Prichep, L. Gugino, E. John, R. Chabot, B. Ho ward, H. Merkin, M. T om, S. W olter, L. Rausch, and W . K ox, “The patient state index as an indicator of the level of hypnosis under general anaesthesia, ” British Journal of Anaesthesia , vol. 92, no. 3, pp. 393–399, 2004. [188] X. Chen, J. T ang, P . F . White, R. H. W ender, H. Ma, A. Sloninsky , and R. Kariger, “ A comparison of patient state index and bispectral index values during the perioperative period, ” Anesthesia & Analgesia , vol. 95, no. 6, pp. 1669–1674, 2002. [189] K. Lee, Y . Kim, Y . Sung, and M. Oh, “The patient state index is well balanced for propofol sedation, ” Hippokratia , vol. 19, no. 3, p. 235, 2015. [190] H. Derendorf and B. Meibohm, “Modeling of pharmacoki- netic/pharmacodynamic (pk/pd) relationships: concepts and perspec- tiv es, ” Pharmaceutical Resear ch , vol. 16, no. 2, pp. 176–185, 1999. [191] T . W . Schnider, C. F . Minto, P . L. Gambus, C. Andresen, D. B. Goodale, S. L. Shafer, and E. J. Y oungs, “The influence of method of administration and cov ariates on the pharmacokinetics of propofol in adult volunteers, ” Anesthesiology: The Journal of the American Society of Anesthesiologists , vol. 88, no. 5, pp. 1170–1182, 1998. [192] J. Sch ¨ uttler and H. Ihmsen, “Population pharmacokinetics of propo- fola multicenter study , ” Anesthesiology: The Journal of the American Society of Anesthesiologists , vol. 92, no. 3, pp. 727–738, 2000. [193] C. F . Minto, T . W . Schnider, T . D. Egan, E. Y oungs, H. J. Lemmens, P . L. Gambus, V . Billard, J. F . Hoke, K. H. Moore, and D. J. Hermann, “Influence of age and gender on the pharmacokinetics and pharmaco- dynamics of remifentanili. model development, ” Anesthesiology: The Journal of the American Society of Anesthesiologists , vol. 86, no. 1, pp. 10–23, 1997. [194] C. F . Minto, T . W . Schnider, and S. L. Shafer , “Pharmacokinetics and pharmacodynamics of remifentanilii. model application, ” Anesthesiol- ogy: The J ournal of the American Society of Anesthesiologists , vol. 86, no. 1, pp. 24–33, 1997. [195] T . Hallynck, H. Soep, J. Thomis, J. Boelaert, R. Daneels, and L. Dettli, “Should clearance be normalised to body surface or to lean body mass?” British Journal of Clinical pharmacology , vol. 11, no. 5, pp. 523–526, 1981. [196] L. B. Sheiner, D. R. Stanski, S. V ozeh, R. D. Miller , and J. Ham, “Simultaneous modeling of pharmacokinetics and pharmacodynamics: application to d-tubocurarine, ” Clinical Pharmacology & Therapeutics , vol. 25, no. 3, pp. 358–371, 1979. [197] S. Shafer , “Principles of pharmacokinetics and pharmacodynamics, ” Principles and Practice of Anesthesiology , pp. 1159–1210, 1998. [198] J. Nino, R. De Ke yser, S. Syafiie, C. Ionescu, and M. Struys, “Epsac- controlled anesthesia with online gain adaptation, ” International Jour - nal of Adaptive Contr ol and Signal Processing , v ol. 23, no. 5, pp. 455–471, 2009. [199] I. Constant and A. Rigouzzo, “Which model for propofol tci in children, ” P ediatric Anesthesia , vol. 20, no. 3, pp. 233–239, 2010. [200] S. Dahmani, I. Stany , C. Brasher , C. Lejeune, B. Bruneau, C. W ood, Y . Ni voche, I. Constant, and I. Murat, “Pharmacological pre vention of sevoflurane-and desflurane-related emergence agitation in children: a meta-analysis of published studies, ” British Journal of Anaesthesia , vol. 104, no. 2, pp. 216–223, 2010. [201] V . Mani and N. S. Morton, “Overview of total intravenous anesthesia in children, ” P ediatric Anesthesia , vol. 20, no. 3, pp. 211–222, 2010. [202] A. Rigouzzo, F . Servin, and I. Constant, “Pharmacokinetic- pharmacodynamic modeling of propofol in children, ” Anesthesiology: The Journal of the American Society of Anesthesiologists , vol. 113, no. 2, pp. 343–352, 2010. [203] Z. T . Zhusubaliyev , A. Medvede v , and M. M. Silva, “Bifurcation analysis for pid-controller tuning based on a minimal neuromuscular blockade model in closed-loop anesthesia, ” in 52nd IEEE Confer ence on Decision and Control . IEEE, 2013, pp. 115–120. [204] M. M. Da Silva, T . W igren, and T . Mendonc ¸ a, “Nonlinear identification of a minimal neuromuscular blockade model in anesthesia, ” IEEE T ransactions on Control Systems T echnology , vol. 20, no. 1, pp. 181– 188, 2012. [205] S. W ard, E. Neill, B. W eatherley , and I. Corall, “Pharmacokinetics of atracurium besylate in healthy patients (after a single iv bolus dose), ” BJ A: British Journal of Anaesthesia , vol. 55, no. 2, pp. 113–118, 1983. [206] B. W eatherley , S. Williams, and E. Neill, “Pharmacokinetics, pharma- codynamics and dose-response relationships of atracurium administered iv , ” BJA: British Journal of Anaesthesia , vol. 55, pp. 39S–45S, 1983. [207] R. Hodrea, R. Morar , I. Nascu, and H. V asian, “Modeling of neuromus- cular blockade in general anesthesia, ” in Advanced T opics in Electrical Engineering (ATEE), 2013 8th International Symposium on . IEEE, 2013, Conference Proceedings, pp. 1–4. [208] K. Soltesz, G. A. Dumont, K. van Heusden, T . H ¨ agglund, and J. M. Ansermino, “Simulated mid-ranging control of propofol and remifen- tanil using eeg-measured hypnotic depth of anesthesia, ” in 2012 IEEE 51st IEEE Conference on Decision and Contr ol (CDC) . IEEE, 2012, pp. 356–361. [209] T . Zikov , S. Bibian, G. A. Dumont, M. Huzmezan, and C. R. Ries, “Quantifying cortical activity during general anesthesia using wav elet analysis, ” IEEE T ransactions on Biomedical Engineering , vol. 53, no. 4, pp. 617–632, 2006. [210] S. E. Kern, G. Xie, J. L. White, and T . D. Egan, “ A response sur- face analysis of propofol-remifentanil pharmacodynamic interaction in volunteers, ” The Journal of the American Society of Anesthesiologists , vol. 100, no. 6, pp. 1373–1381, 2004. [211] M. M. Da Silva, T . Mendon, and T . W igren, “Online nonlinear identification of the effect of drugs in anaesthesia using a minimal pa- rameterization and bis measurements, ” in American Contr ol Conference (ACC), 2010 . IEEE, 2010, Conference Proceedings, pp. 4379–4384. [212] M. Silva, “Prediction error identification of minimally parameterized wiener models in anesthesia, ” in Pr oc. 18th IF AC W orld Congress , 2011, pp. 5615–5620. [213] F . N. Nogueira, T . Mendonc ¸ a, and P . Rocha, “Positive state observer for the automatic control of the depth of anesthesia-clinical results, ” Computer Methods and Pro grams in Biomedicine , 2016. [214] T . Mendonc ¸ a, H. Alonso, M. M. da Silva, S. Estev es, and M. Seabra, “Comparing different identification approaches for the depth of anes- thesia using bis measurements, ” IF AC Proceedings V olumes , vol. 45, no. 16, pp. 781–785, 2012. [215] Z. T an, R. Kaddoum, and H. W . Le Y i W ang, “Decision-oriented multi-outcome modeling for anesthesia patients, ” The Open Biomedical Engineering journal , vol. 4, p. 113, 2010. [216] M. Nakayama, H. Ichinose, S. Y amamoto, N. Kanaya, and A. Namiki, “The effect of fentanyl on hemodynamic and bispectral index changes during anesthesia induction with propofol, ” Journal of clinical anes- thesia , vol. 14, no. 2, pp. 146–149, 2002. [217] Z. T an, “Monitoring, diagnosis, and control for adv anced anesthesia management, ” Ph.D. dissertation, W ayne State University , 2011. [218] M. M. Silva, T . W igren, and T . Mendonc ¸ a, “ A reduced mimo wiener model for recursi ve identification of the depth of anesthesia, ” Inter - national Journal of Adaptive Contr ol and Signal Pr ocessing , vol. 28, no. 12, pp. 1357–1371, 2014. [219] M. J. Hall, A. Schwartzman, J. Zhang, and X. Liu, “ Ambulatory surgery data from hospitals and ambulatory surgery centers: United states, 2010, ” National Health Statistics Reports , no. 102, pp. 1–15, 2017. [220] S. Abdulla and P . W en, “Depth of anaesthesia control investigation using robust deadbeat control technique, ” in 2012 ICME International Confer ence on Complex Medical Engineering (CME) . IEEE, 2012, Conference Proceedings, pp. 107–111. [221] J. Bruhn, P . Myles, R. Sneyd, and M. Struys, “Depth of anaesthesia monitoring: what’ s av ailable, what’ s validated and what’ s next?” BJA: British Journal of Anaesthesia , vol. 97, no. 1, pp. 85–94, 2006. [222] F . Padula, C. Ionescu, N. Latronico, M. Paltenghi, A. V isioli, and G. V i- vacqua, “Optimized pid control of depth of hypnosis in anesthesia, ” Computer Methods and Pro grams in Biomedicine , vol. 144, pp. 21– 35, 2017. [223] M. Neckebroek, C. Ionescu, K. van Amsterdam, T . De Smet, P . De Baets, J. Decruyenaere, R. De Ke yser, and M. Struys, “ A comparison of propofol-to-bis post-operative intensive care sedation by means of target controlled infusion, bayesian-based and predictive control methods: an observational, open-label pilot study , ” Journal of Clinical Monitoring and Computing , pp. 1–12, 2018. [224] J. F . Biebuyck, D. A. O’hara, D. K. Bogen, and A. Noordergraaf, “The use of computers for controlling the delivery of anesthesia, ” Anes- thesiology: The Journal of the American Society of Anesthesiologists , vol. 77, no. 3, pp. 563–581, 1992. [225] A. J. Asbury , “Feedback control in anaesthesia, ” International journal of clinical monitoring and computing , vol. 14, no. 1, pp. 1–10, 1997. [226] K. Solt ´ esz, On automation in anesthesia . Department of Automatic Control, Lund University , 2013. [227] A. R. Absalom and G. Kenny , “Closed-loop control of propofol anaesthesia using bispectral index T M : performance assessment in patients receiving computer-controlled propofol and manually con- trolled remifentanil infusions for minor surgery , ” British Journal of Anaesthesia , vol. 90, no. 6, pp. 737–741, 2003. [228] J. Glen, “The development of ’ diprifusor’: a TCI system for propofol, ” Anaesthesia , vol. 53, pp. 13–21, 1998. [229] L. Merigo, M. Beschi, F . Padula, N. Latronico, M. Paltenghi, and A. V isioli, “Event-based control of depth of hypnosis in anesthesia, ” Computer Methods and Pro grams in Biomedicine , vol. 147, pp. 63–83, 2017. [230] N. Liu, T . Chazot, A. Genty , A. Landais, A. Restoux, K. McGee, P .-A. Lalo, B. T rillat, L. Barvais, and M. Fischler, “Titration of propofol for anesthetic induction and maintenance guided by the bispectral index: Closed-loop versus manual controla prospectiv e, randomized, multi- center study , ” Anesthesiology: The Journal of the American Society of Anesthesiologists , vol. 104, no. 4, pp. 686–695, 2006. [231] N. Liu, T . Chazot, S. Hamada, A. Landais, N. Boichut, C. Dussaussoy , B. Trillat, L. Beydon, E. Samain, and D. I. Sessler , “Closed-loop coadministration of propofol and remifentanil guided by bispectral index: a randomized multicenter study , ” Anesthesia & Analgesia , vol. 112, no. 3, pp. 546–557, 2011. [232] C. M. Ionescu, R. De Keyser , B. C. T orrico, T . De Smet, M. M. Struys, and J. E. Normey-Rico, “Robust predictive control strategy applied for propofol dosing using bis as a controlled variable during anesthesia, ” IEEE T ransactions on Biomedical Engineering , vol. 55, no. 9, pp. 2161–2170, 2008. [233] T . De Smet, M. M. Struys, M. M. Neckebroek, K. V an den Hauwe, S. Bonte, and E. P . Mortier, “The accuracy and clinical feasibility of a new bayesian-based closed-loop control system for propofol administration using the bispectral index as a controlled variable, ” Anesthesia & Analgesia , vol. 107, no. 4, pp. 1200–1210, 2008. [234] M. M. Struys, T . De Smet, L. F . V ersichelen, S. V an de V elde, R. V an den Broecke, and E. P . Mortier, “Comparison of closed-loop controlled administration of propofol using bispectral index as the controlled variable versus standard practice controlled administration, ” Anesthesiology: The Journal of the American Society of Anesthesiolo- gists , vol. 95, no. 1, pp. 6–17, 2001. [235] G. A. Dumont, “Closed-loop control of anesthesia-a revie w , ” IF AC Pr oceedings V olumes , vol. 45, no. 18, pp. 373–378, 2012. [236] G. A. Dumont and J. M. Ansermino, “Closed-loop control of anesthe- sia: a primer for anesthesiologists, ” Anesthesia & Analgesia , vol. 117, no. 5, pp. 1130–1138, 2013. [237] M. Ilyas, M. F . U. Butt, M. Bilal, K. Mahmood, A. Khaqan, and R. Ali Riaz, “ A review of modern control strategies for clinical ev aluation of propofol anesthesia administration employing hypnosis lev el regulation, ” BioMed Researc h International , vol. 2017, 2017. [238] R. Chilcoat, “ A review of the control of depth of anaesthesia, ” T rans- actions of the Institute of Measurement and Contr ol , vol. 2, no. 1, pp. 38–45, 1980. [239] T . Sieber, C. Frei, M. Derighetti, P . Feigenwinter , D. Leibundgut, and A. M. Zbinden, “Model-based automatic feedback control versus human control of end-tidal isoflurane concentration using low-flo w anaesthesia, ” British Journal of Anaesthesia , vol. 85, no. 6, pp. 818– 825, 2000. [240] M. Struys, A. Kalmar, L. De Baerdemaeker, E. Mortier, G. Rolly , J. Manigel, and W . Buschke, “Time course of inhaled anaesthetic drug deli very using a ne w multifunctional closed-circuit anaesthesia ventilator . in vitro comparison with a classical anaesthesia machine, ” British Journal of Anaesthesi , vol. 94, no. 3, pp. 306–317, 2004. [241] B. Lortat-Jacob, V . Billard, W . Buschke, and F . Servin, “ Assessing the clinical or pharmaco-economical benefit of target controlled desflurane deliv ery in surgical patients using the zeus® anaesthesia machine, ” Anaesthesia , vol. 64, no. 11, pp. 1229–1235, 2009. [242] C. W . Connor, “Optimizing target control of the vessel rich group with volatile anesthetics, ” Journal of Clinical Monitoring and Computing , vol. 33, no. 3, pp. 445–454, 2019. [243] M. Mahfouf, A. Asbury , and D. Linkens, “Unconstrained and con- strained generalised predictiv e control of depth of anaesthesia during surgery , ” Contr ol Engineering Practice , vol. 11, no. 12, pp. 1501–1515, 2003. [244] A. Krieger , N. Panoskaltsis, A. Mantalaris, M. C. Georgiadis, and E. N. Pistikopoulos, “Modeling and analysis of indi vidualized phar- macokinetics and pharmacodynamics for volatile anesthesia, ” IEEE T ransactions on Biomedical Engineering , vol. 61, no. 1, pp. 25–34, 2013. [245] A. Krieger and E. N. Pistikopoulos, “Model predictiv e control of anesthesia under uncertainty , ” Computers & Chemical Engineering , vol. 71, pp. 699–707, 2014. [246] A. Krieger, “Modelling, optimisation and explicit model predictive con- trol of anaesthesia drug deliv ery systems, ” Ph.D. dissertation, Imperial College London, 2013. [247] S. Das, S. Das, and K. Maharatna, “Control strategy for anaesthetic drug dosage with interaction among human physiological organs using optimal fractional order pid controller , ” in Proceedings of The 2014 International Confer ence on Control, Instrumentation, Ener gy and Communication (CIEC) . IEEE, 2014, Conference Proceedings, pp. 66–70. [248] F . L. Higgen, G. G. Supp, J. F . Hipp, A. K. Engel, and M. Siegel, “Mid-latency auditory evok ed potentials differentially predict sedation and drug level under opioid and hypnotic agents, ” F r ontiers in Phar- macology , vol. 9, p. 1427, 2018. [249] G. Kenn y and H. Mantzaridis, “Closed-loop control of propofol anaes- thesia, ” British Journal of Anaesthesia , vol. 83, no. 2, pp. 223–228, 1999. [250] A. Morley , J. Derrick, P . Mainland, B. Lee, and T . Short, “Closed loop control of anaesthesia: an assessment of the bispectral index as the target of control, ” Anaesthesia , vol. 55, no. 10, pp. 953–959, 2000. [251] T . M. Hemmerling, S. Charabati, C. Zaouter, C. Minardi, and P . A. Mathieu, “ A randomized controlled trial demonstrates that a novel closed-loop propofol system performs better hypnosis control than manual administration, ” Canadian Journal of Anesthesia/Journal cana- dien d’anesth ´ esie , vol. 57, no. 8, pp. 725–735, 2010. [252] N. Liu, M. Le Guen, F . Benabbes-Lambert, T . Chazot, B. T rillat, D. I. Sessler , and M. Fischler, “Feasibility of closed-loop titration of propofol and remifentanil guided by the spectral m-entropy monitor , ” Anesthesiology: The Journal of the American Society of Anesthesiolo- gists , vol. 116, no. 2, pp. 286–295, 2012. [253] N. W est, G. A. Dumont, K. van Heusden, C. L. Petersen, S. Khosravi, K. Soltesz, A. Umedaly , E. Reimer, and J. M. Ansermino, “Rob ust closed-loop control of induction and maintenance of propofol anes- thesia in children, ” P ediatric Anesthesia , vol. 23, no. 8, pp. 712–719, 2013. [254] K. V an Heusden, G. A. Dumont, K. Soltesz, C. L. Petersen, A. Umedaly , N. W est, and J. M. Ansermino, “Design and clinical ev aluation of robust pid control of propofol anesthesia in children, ” IEEE T ransactions on Contr ol Systems T echnology , vol. 22, no. 2, pp. 491–501, 2014. [255] A. Simalatsar, M. Guidi, and T . Buclin, “Cascaded pid controller for anaesthesia delivery , ” in 2016 38th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC) . IEEE, 2016, pp. 533–536. [256] K. Soltesz, J.-O. Hahn, T . H ¨ agglund, G. A. Dumont, and J. M. Ansermino, “Individualized closed-loop control of propofol anesthesia: A preliminary study , ” Biomedical Signal Pr ocessing and Contr ol , vol. 8, no. 6, pp. 500–508, 2013. [257] K. J. ˚ Astr ¨ om, T . H ¨ agglund, and K. J. Astrom, Advanced PID contr ol . ISA-The Instrumentation, Systems, and Automation Society Research T riangle, 2006, vol. 461. [258] K. Ejaz and J.-S. Y ang, “Controlling depth of anesthesia using pid tuning: a comparative model-based study , ” in Pr oceedings of the 2004 IEEE International Conference on Control Applications , vol. 1. IEEE, 2004, Conference Proceedings, pp. 580–585. [259] F . Padula, C. Ionescu, N. Latronico, M. Paltenghi, A. V isioli, and G. V i- vacqua, “Inversion-based propofol dosing for intra venous induction of hypnosis, ” Communications in Nonlinear Science and Numerical Simulation , vol. 39, pp. 481–494, 2016. [260] S. Anna and P . W en, “Depth of anesthesia control using internal model control techniques, ” in IEEE/ICME International Conference on Complex Medical Engineering . IEEE, 2010, Conference Proceedings, pp. 294–300. [261] J.-O. Hahn, G. A. Dumont, and J. M. Ansermino, “Robust closed-loop control of hypnosis with propofol using wavcns index as the controlled variable, ” Biomedical Signal Pr ocessing and Contr ol , vol. 7, no. 5, pp. 517–524, 2012. [262] L. Merigo, F . Padula, A. Pawlo wski, S. Dormido, J. L. G. Snchez, N. Latronico, M. Paltenghi, and A. V isioli, “ A model-based control scheme for depth of hypnosis in anesthesia, ” Biomedical Signal Pr o- cessing and Control , vol. 42, pp. 216–229, 2018. [263] G. A. Dumont, A. Martinez, and J. M. Ansermino, “Rob ust control of depth of anesthesia, ” International Journal of Adaptive Contr ol and Signal Processing , vol. 23, no. 5, pp. 435–454, 2009. [264] K. Bhole and S. Agashe, “ Automating intravenous anesthesia with a fuzzy inference system coupled with a proportional integral derivati ve (pid) controller , ” American Journal of Biomedical Science and Engi- neering , vol. 1, no. 6, pp. 93–99, 2015. [265] L. Merigo, F . Padula, N. Latronico, T . Mendonc ¸ a, M. P altenghi, P . Rocha, and A. V isioli, “Optimized pid tuning for the automatic control of neuromuscular blockade, ” IF AC-P apersOnLine , vol. 51, no. 4, pp. 66–71, 2018. [266] H. Panagopoulos, K. Astrom, and T . Hagglund, “Design of pid con- trollers based on constrained optimisation, ” IEE Pr oceedings-Control Theory and Applications , vol. 149, no. 1, pp. 32–40, 2002. [267] O. Castillo, G. Huesca, and F . V aldez, “Evolutionary computing for optimizing type-2 fuzzy systems in intelligent control of non-linear dy- namic plants, ” in Fuzzy Information Pr ocessing Society , 2005. NAFIPS 2005. Annual Meeting of the North American . IEEE, 2005, Conference Proceedings, pp. 247–251. [268] K. Soltesz, J.-O. Hahn, G. A. Dumont, and J. M. Ansermino, “Indi- vidualized pid control of depth of anesthesia based on patient model identification during the induction phase of anesthesia, ” in 2011 50th IEEE Confer ence on Decision and Control and Eur opean Contr ol Confer ence . IEEE, 2011, pp. 855–860. [269] F . Padula, C. Ionescu, N. Latronico, M. Paltenghi, A. V isioli, and G. V ivacqua, “ A gain-scheduled pid controller for propofol dosing in anesthesia, ” IF AC-P apersOnLine , vol. 48, no. 20, pp. 545–550, 2015. [270] Y . Sawaguchi, E. Furutani, G. Shirakami, M. Araki, and K. Fukuda, “ A model predictive sedation control system under total intravenous anesthesia, ” in IEEE EMBS Asian-P acific Conference on Biomedical Engineering, 2003. IEEE, 2003, pp. 358–359. [271] E. Furutani, Y . Sawaguchi, G. Shirakami, M. Araki, and K. Fukuda, “ A hypnosis control system using a model predictive controller with online identification of individual parameters, ” in Contr ol Applications, 2005. CCA 2005. Proceedings of 2005 IEEE Confer ence on . IEEE, 2005, Conference Proceedings, pp. 154–159. [272] A. Bamdadian, F . T owhidkhah, and M. H. Moradi, “Generalized predictiv e control of depth of anesthesia by using a pharmocokinetic- pharmacodynamic model of the patient, ” in 2008 2nd International Confer ence on Bioinformatics and Biomedical Engineering . IEEE, 2008, Conference Proceedings, pp. 1276–1279. [273] G. Goodwin, A. Medioli, K. Murray , R. Sykes, and C. Stephen, Applications of MPC in the Area of Health Care . Springer, 2019, pp. 529–550. [274] D. Ingole, J. Holaza, B. T akcs, and M. Kvasnica, “Fpga-based explicit model predictiv e control for closed-loop control of intra venous anesthe- sia, ” in 2015 20th International Conference on Pr ocess Control (PC) . IEEE, 2015, Conference Proceedings, pp. 42–47. [275] I. Nas ¸cu and E. N. Pistikopoulos, “Multiparametric model predictiv e control and state estimation of the hypnotic component in anesthesia, ” in 2016 IEEE International Conference on Automation, Quality and T esting, Robotics (AQTR) . IEEE, 2016, pp. 1–6. [276] M. L. Darby and M. Nikolaou, “ A parametric programming approach to moving-horizon state estimation, ” A utomatica , vol. 43, no. 5, pp. 885–891, 2007. [277] I. Nas ¸cu and E. N. Pistikopoulos, “Multiparametric model predictiv e control strategies of the hypnotic component in intravenous anesthe- sia, ” in 2016 IEEE International Confer ence on Systems, Man, and Cybernetics (SMC) . IEEE, 2016, pp. 002 828–002 833. [278] ——, “Modeling, estimation and control of the anaesthesia process, ” Computers & Chemical Engineering , vol. 107, pp. 318–332, 2017. [279] A. Pawło wski, L. Merigo, J. Guzman, A. V isioli, and S. Dormido, “Event-based gpc for depth of hypnosis in anesthesia for efficient use of propofol, ” in 2017 3r d International Confer ence on Event-Based Contr ol, Communication and Signal Pr ocessing (EBCCSP) . IEEE, 2017, pp. 1–7. [280] Y . Sawaguchi, E. Furutani, G. Shirakami, M. Araki, and K. Fukuda, “ A model-predictive hypnosis control system under total intravenous anesthesia, ” IEEE T ransactions on Biomedical Engineering , vol. 55, no. 3, pp. 874–887, 2008. [281] F . Robayo, D. Sendoya, R. Hodrea, C. Ionescu, and R. de K eyser , “Estimating the time-delay for predictive control in general anesthesia, ” in Control and Decision Conference (CCDC), 2010 Chinese . IEEE, 2010, pp. 3719–3724. [282] V . Sartori, P . M. Schumacher, T . Bouillon, M. Luginbuehl, and M. Morari, “On-line estimation of propofol pharmacodynamic parame- ters, ” in 2005 IEEE Engineering in Medicine and Biology 27th Annual Confer ence . IEEE, 2006, pp. 74–77. [283] D. Ingole, J. Drgo ˇ na, and M. Kvasnica, “Offset-free hybrid model predictiv e control of bispectral index in anesthesia, ” in 2017 21st International Confer ence on Pr ocess Control (PC) . IEEE, 2017, pp. 422–427. [284] I. Nas ¸ cu, “ Adv anced multiparametric optimization and control studies for anaesthesia, ” Ph.D. dissertation, Imperial College London, 2016. [285] P . Sopasakis, P . Patrinos, and H. Sarimveis, “Robust model predictive control for optimal continuous drug administration, ” Computer Meth- ods and Pr ograms in Biomedicine , vol. 116, no. 3, pp. 193–204, 2014. [286] W . M. Haddad, T . Hayakawa, and J. M. Bailey , “ Adaptiv e control for non-negati ve and compartmental dynamical systems with applications to general anesthesia, ” International Journal of Adaptive Contr ol and Signal Processing , vol. 17, no. 3, pp. 209–235, 2003. [287] ——, “Nonlinear adaptiv e control for intensive care unit sedation and operating room hypnosis, ” in American Contr ol Confer ence, 2003. Pr oceedings of the 2003 , vol. 2. IEEE, 2003, Conference Proceedings, pp. 1808–1813. [288] A. R. Absalom, N. Sutcliffe, and G. N. Kenny , “Closed-loop control of anesthesia using bispectral indexperformance assessment in patients undergoing major orthopedic surgery under combined general and regional anesthesia, ” Anesthesiology: The Journal of the American Society of Anesthesiologists , vol. 96, no. 1, pp. 67–73, 2002. [289] W . M. Haddad, T . Hayakawa, and J. M. Bailey , “ Adaptiv e control for nonlinear compartmental dynamical systems with applications to clinical pharmacology , ” Systems & Contr ol Letters , vol. 55, no. 1, pp. 62–70, 2006. [290] T . Hayakawa, W . M. Haddad, N. Hovakimyan, and J. M. Bailey , “Neu- ral network adaptiv e dynamic output feedback control for nonlinear nonnegati ve systems using tapped delay memory units, ” in American Contr ol Conference , 2004. Proceedings of the 2004 , vol. 5. IEEE, 2004, Conference Proceedings, pp. 4505–4510. [291] W . M. Haddad, K. Y . V olyanskyy , J. M. Bailey , and J. J. Im, “Neu- roadaptiv e output feedback control for automated anesthesia with noisy eeg measurements, ” IEEE T ransactions on Contr ol Systems T echnology , vol. 19, no. 2, pp. 311–326, 2011. [292] J. M. Bailey , W . M. Haddad, J. J. Im, T . Hayakawa, and P . A. Nagel, “ Adaptive and neural network adaptive control of depth of anesthesia during surgery , ” in American Contr ol Confer ence, 2006 . IEEE, 2006, Conference Proceedings, p. 6 pp. [293] H. Schwilden, J. Sch ¨ uttler , and H. Stoeckel, “Closed-loop feedback control of methohexital anesthesia by quantitativ e eeg analysis in humans. ” Anesthesiology , vol. 67, no. 3, pp. 341–347, 1987. [294] H. Schwilden, H. Stoeckel, and J. Sch ¨ uttler , “Closed-loop feedback control of propofol anaesthesia by quantitativ e eeg analysis in humans, ” BJ A: British Journal of Anaesthesia , vol. 62, no. 3, pp. 290–296, 1989. [295] G. Nav arro-Guerrero and Y . T ang, “ Adaptive control for anesthesia based on a simple fractional-order model, ” in 2015 54th IEEE Confer- ence on Decision and Contr ol (CDC) . IEEE, 2015, pp. 5623–5628. [296] ——, “Fractional order model reference adaptive control for anesthe- sia, ” International Journal of Adaptive Contr ol and Signal Pr ocessing , vol. 31, no. 9, pp. 1350–1360, 2017. [297] M. Ralph, C. Beck, and M. Bloom, “L1-adaptiv e methods for control of patient response to anesthesia, ” in American Control Conference (ACC), 2011 . IEEE, 2011, pp. 1729–1735. [298] E. Kharisov , C. L. Beck, and M. Bloom, “Control of patient response to anesthesia using l 1 adaptive methods, ” IF AC Proceedings V olumes , vol. 45, no. 18, pp. 391–396, 2012. [299] ——, “Regulation of anesthesia deliv ery via l1-adaptiv e control, ” in 2013 Proceedings of the Conference on Control and its Applications . SIAM, 2013, Conference Proceedings, pp. 191–198. [300] ——, “Design of l1 adaptive controllers for human patient anesthesia, ” Contr ol Engineering Practice , vol. 44, pp. 65–77, 2015. [301] O. Simanski, M. Janda, A. Schubert, J. Bajorat, R. Hofmockel, and B. Lampe, “Progress of automatic drug delivery in anaesthesia-the ’ rostock assistant system for anaesthesia control (ran)’, ” International Journal of Adaptive Control and Signal Pr ocessing , vol. 23, no. 5, pp. 504–521, 2009. [302] Y .-C. Chou, M. F . Abbod, J.-S. Shieh, and C.-Y . Hsu, “Multiv ariable fuzzy logic/self-organizing for anesthesia control, ” Journal of Medical and Biological Engineering , vol. 30, no. 5, pp. 297–306, 2010. [303] S. A. Abdulla and P . W en, “Robust internal model control for depth of anaesthesia, ” International Journal of Mechatr onics and Automation , vol. 1, no. 1, pp. 1–8, 2011. [304] J. A. Mendez, A. Leon, A. Marrero, J. M. Gonzalez-Cav a, J. A. Reboso, J. I. Este vez, and J. F . Gomez-Gonzalez, “Impro ving the anesthetic process by a fuzzy rule based medical decision system, ” Artificial Intelligence in Medicine , vol. 84, pp. 159–170, 2018. [305] J.-S. Shieh, L.-W . Chang, T .-C. Y ang, and C.-C. Liu, “ An enhanced patient controlled analgesia (epca) for the extracorporeal shock wave lithotripsy (eswl), ” Biomedical Engineering: Applications, Basis and Communications , vol. 19, no. 01, pp. 7–17, 2007. [306] J.-S. Shieh, C.-Y . Dai, Y .-R. W en, and W .-Z. Sun, “ A novel fuzzy pain demand index deriv ed from patient-controlled analgesia for postoper- ativ e pain, ” IEEE T ransactions on Biomedical Engineering , vol. 54, no. 12, pp. 2123–2132, 2007. [307] A. U. Schubert, M. Janda, O. Simanski, J. Bajorat, B. Pohl, R. Hof- mockel, and B. Lampe, “ A fuzzy system for regulation of the analgesic remifentanil during general anaesthesia, ” in 2008 16th Mediterranean Confer ence on Contr ol and Automation . IEEE, 2008, pp. 1634–1639. [308] D. A. Linkens and S. Hasnain, “Self-organising fuzzy logic control and application to muscle relaxant anaesthesia, ” in IEE Pr oceedings D (Contr ol Theory and Applications) , vol. 138. IET , 1991, Conference Proceedings, pp. 274–284. [309] P . A. Fazendeiro and J. V . de Oli veira, “ A survey of fuzzy control strategies for neuromuscular blockade using continuous infusion of atracurium, ” in 2002 IEEE W orld Congress on Computational Intelli- gence. 2002 IEEE International Conference on Fuzzy Systems. , vol. 1. IEEE, 2002, Conference Proceedings, pp. 547–552. [310] M. Abbod, M. Mahfouf, and D. Linkens, “Multi-objecti ve genetic optimisation for self-organising fuzzy logic control, ” 1998. [311] Y .-X. Liu, J.-S. Shieh, S.-Z. Fan, F . Doctor, and K.-K. Jen, “Genetic type-2 self-organising fuzzy logic controller applied to anaesthesia, ” in 2015 Confer ence on T echnologies and Applications of Artificial Intelligence (TAAI) . IEEE, 2015, Conference Proceedings, pp. 83– 88. [312] J.-S. Shieh, M.-H. Kao, and C.-C. Liu, “Genetic fuzzy modelling and control of bispectral index (bis) for general intravenous anaesthesia, ” Medical Engineering & Physics , vol. 28, no. 2, pp. 134–148, 2006. [313] J. Muthuswamy and R. J. Roy , “The use of fuzzy integrals and bispectral analysis of the electroencephalogram to predict movement under anesthesia, ” IEEE T ransactions on Biomedical Engineering , vol. 46, no. 3, pp. 291–299, 1999. [314] J.-S. Shieh, S.-Z. F an, and W .-L. Shi, “The intelligent model of a patient using artificial neural networks for inhalational anaesthesia, ” Journal of the Chinese Institute of Chemical Engineers , vol. 33, no. 6, pp. 609–620, 2002. [315] F . T aheriyan, M. sadat Ghafourian, and A. Noori, “ Applying ga optimization algorithm for interval type-2 fuzzy logic controller pa- rameters of multi variable anesthesia system, ” in Iranian Conference on Electrical Engineering (ICEE) . IEEE, 2018, pp. 1613–1618. [316] X.-S. Zhang, J. W . Huang, and R. J. Roy , Depth of anesthesia contr ol with fuzzy logic . Springer , 2002, pp. 83–112. [317] A. Marrero, J. M ´ endez, J. Reboso, I. Mart ´ ın, and J. Calvo, “ Adaptive fuzzy modeling of the hypnotic process in anesthesia, ” Journal of Clinical Monitoring and Computing , vol. 31, no. 2, pp. 319–330, 2017. [318] J.-S. Shieh, M. F . Abbod, C. Hsu, S.-J. Huang, Y .-Y . Han, and S.-Z. Fan, Monitoring and control of anesthesia using multivariable self- or ganizing fuzzy logic structure . Springer, 2009, pp. 273–295. [319] M. El-Bardini and A. M. El-Nagar, “Direct adaptive interval type-2 fuzzy logic controller for the multi variable anaesthesia system, ” Ain Shams Engineering Journal , vol. 2, no. 3-4, pp. 149–160, 2011. [320] R. Allen and D. Smith, “Neuro-fuzzy closed-loop control of depth of anaesthesia, ” Artificial Intelligence in Medicine , vol. 21, no. 1-3, pp. 185–191, 2001. [321] C. S. Nunes, M. Mahfouf, D. A. Linkens, and J. E. Peacock, “Modelling and multiv ariable control in anaesthesia using neural- fuzzy paradigms: Part i. classification of depth of anaesthesia and dev elopment of a patient model, ” Artificial Intelligence in Medicine , vol. 35, no. 3, pp. 195–206, 2005. [322] M. Mahfouf, C. S. Nunes, D. A. Linkens, and J. E. Peacock, “Mod- elling and multi variable control in anaesthesia using neural-fuzzy paradigms: Part ii. closed-loop control of simultaneous administration of propofol and remifentanil, ” Artificial Intellig ence in Medicine , vol. 35, no. 3, pp. 207–213, 2005. [323] M. T osun and R. G ¨ unt ¨ urk ¨ un, “ Anesthetic gas control with neuro-fuzzy system in anesthesia, ” Expert Systems with Applications , vol. 37, no. 3, pp. 2690–2695, 2010. [324] A. M. El-Nagar and M. El-Bardini, “Interval type-2 fuzzy neural network controller for a multivariable anesthesia system based on a hardware-in-the-loop simulation, ” Artificial Intelligence in Medicine , vol. 61, no. 1, pp. 1–10, 2014. [325] Y .-N. Y u, F . Doctor, S.-Z. Fan, and J.-S. Shieh, “ An adaptive monitoring scheme for automatic control of anaesthesia in dynamic surgical en vironments based on bispectral index and blood pressure, ” Journal of Medical Systems , vol. 42, no. 5, p. 95, 2018. [326] J. A. M ´ endez, A. Marrero, J. A. Reboso, and A. Le ´ on, “ Adaptiv e fuzzy predictiv e controller for anesthesia deli very , ” Contr ol Engineering Practice , vol. 46, pp. 1–9, 2016. [327] Y . W ang, Q. Jin, and R. Zhang, “Improved fuzzy pid controller design using predictiv e functional control structure, ” ISA T ransactions , vol. 71, pp. 354–363, 2017. [328] N. Sadati, M. Hosseinzadeh, and G. A. Dumont, “Multi-model robust control of depth of hypnosis, ” Biomedical Signal Processing and Contr ol , vol. 40, pp. 443–453, 2018. [329] G. Rigatos, E. Rigatou, and N. Zervos, “ A nonlinear h-infinity approach to optimal control of the depth of anaesthesia, ” in AIP Conference Pr oceedings , vol. 1790, no. 1. AIP Publishing, 2016, p. 060006. [330] R. Padmanabhan, N. Meskin, C. M. Ionescu, and W . M. Haddad, “ A nonov ershooting tracking controller for simultaneous infusion of anesthetics and analgesics, ” Biomedical Signal Pr ocessing and Control , vol. 49, pp. 375–387, 2019. [331] O. Iskrenovic-Momcilovic and A. Momcilovic, “Sliding mode control for depth of anesthesia, ” Annals of the F aculty of Engineering Hune- doara , vol. 15, no. 4, pp. 41–46, 2017. [332] L. H. Abood, E. H. Karam, and A. H. Issa, “Design of adaptive neuro sliding mode controller for anesthesia drug deli very based on biogeography based optimization, ” International Journal of Advanced Computer Research , vol. 9, no. 42, pp. 146–155, 2019. [333] A. Khaqan, M. Bilal, M. Ilyas, B. Ijaz, and R. Ali Riaz, “Control law design for propofol infusion to regulate depth of hypnosis: a nonlinear control strategy , ” Computational and Mathematical Methods in Medicine , vol. 2016, 2016. [334] A. Khaqan, Q. ul Hasan, S. A. Malik, M. Bilal, M. F . U. Butt, and R. A. Riaz, “Comparison of two nonlinear control strategies for hypnosis regulation, ” Arabian Journal for Science and Engineering , vol. 42, no. 12, pp. 5165–5178, 2017. [335] R. Hodrea, I. Nascu, I. Nascu, R. De Ke yser, and H. V asian, “Epsac versus pid control of neuromuscular blockade, ” in 2014 IEEE Inter- national Conference on Automation, Quality and T esting, Robotics . IEEE, 2014, Conference Proceedings, pp. 1–6. [336] S. Y elneedi, L. Samavedham, and G. Rangaiah, “ Advanced control strategies for the regulation of hypnosis with propofol, ” Industrial & Engineering Chemistry Researc h , vol. 48, no. 8, pp. 3880–3897, 2009. [337] M. Y ousefi, K. van Heusden, I. M. Mitchell, J. M. Ansermino, and G. A. Dumont, “ A formally-verified safety system for closed-loop anesthesia, ” IF AC-P apersOnLine , vol. 50, no. 1, pp. 4424–4429, 2017. [338] M. Y ousefi, K. van Heusden, N. W est, I. M. Mitchell, J. M. Ansermino, and G. A. Dumont, “ A formalized safety system for closed-loop anesthesia with pharmacokinetic and pharmacodynamic constraints, ” Contr ol Engineering Practice , vol. 84, pp. 23–31, 2019. [339] M. J. Khodaei, A. Mehrvarz, N. Jalili et al. , “ An adaptiv e multi-critic neuro-fuzzy control framework for intravenous anesthesia administra- tion, ” IF AC-P apersOnLine , vol. 51, no. 34, pp. 202–207, 2019. [340] Senseonics, Inc, “FDA executi ve summary - senseonics ev ersense continuous glucose monitoring system, ” 2018. [Online]. A vailable: https://www .fda.gov/do wnloads [341] R. B. Colquitt, D. A. Colquhoun, and R. H. Thiele, “In silico modelling of physiologic systems, ” Best Practice & Researc h Clinical Anaesthe- siology , vol. 25, no. 4, pp. 499–510, 2011. [342] G. T . T ucker, “Personalized drug dosage-closing the loop, ” Pharma- ceutical Research , vol. 34, no. 8, pp. 1539–1543, 2017. [343] N. Arroyo-Curr ´ as, G. Ortega, D. A. Copp, K. L. Ploense, Z. A. Plaxco, T . E. Kippin, J. P . Hespanha, and K. W . Plaxco, “High-precision control of plasma drug levels using feedback-controlled dosing, ” ACS Pharmacology & T ranslational Science , vol. 1, no. 2, pp. 110–118, 2018. [344] J. Y u, Y . Zhang, J. Y an, A. R. Kahkoska, and Z. Gu, “ Advances in bioresponsiv e closed-loop drug delivery systems, ” International Journal of Pharmaceutics , vol. 544, no. 2, pp. 350–357, 2018. [345] P . H. van der Graaf, N. Benson, and L. A. Peletier, “T opics in mathematical pharmacology , ” Journal of Dynamics and Differ ential Equations , vol. 28, no. 3-4, pp. 1337–1356, 2016. [346] P . Sopasakis, H. Sarimv eis, P . Macheras, and A. Dokoumetzidis, “Fractional calculus in pharmacokinetics, ” Journal of Pharmacokinetics and Pharmacodynamics , vol. 45, no. 1, pp. 107–125, 2018. [347] R. S. Parker and F . J. Doyle III, “Control-relev ant modeling in drug deliv ery , ” Advanced Drug Delivery Revie ws , vol. 48, no. 2-3, pp. 211– 228, 2001. [348] B. Parvinian, P . Pathmanathan, C. Daluwatte, F . Y aghouby , R. A. Gray , S. W eininger, T . M. Morrison, and C. G. Scully , “Credibility evidence for computational patient models used in the development of physiological closed-loop controlled de vices for critical care medicine, ” F r ontiers in Physiology , vol. 10, 2019. [349] L. Mulugeta, A. Drach, A. Erdemir, C. A. Hunt, M. Horner , J. P . Ku, J. G. Myers Jr, R. V adigepalli, and W . W . L ytton, “Credibility , replicability , and reproducibility in simulation for biomedicine and clinical applications in neuroscience, ” F rontier s in Neur oinformatics , vol. 12, 2018. [350] “ Assessing credibility of computational modeling through verification and validation: Application to medical devices, ” 2018. [351] G. S. V irk, K. Chinzei, and M. Brossoit, “Emergence of medical robots and autonomy in medical electrical equipment, ” Biomedical Instrumentation & T echnolo gy , vol. 52, no. 2, pp. 156–159, 2018. [352] G.-Z. Y ang, J. Cambias, K. Cleary , E. Daimler, J. Drake, P . E. Dupont, N. Hata, P . Kazanzides, S. Martel, and R. V . Patel, “Medical robotics- regulatory , ethical, and legal considerations for increasing lev els of autonomy , ” Science Robotics , vol. 2, no. 4, p. 8638, 2017. [353] N. Dey , A. S. Ashour, F . Shi, S. J. Fong, and J. M. R. T avares, “Medical cyber -physical systems: A surve y , ” Journal of Medical Systems , vol. 42, no. 4, p. 74, 2018. [354] M. Crawford, “T op 6 robotic applications in medicine, ” 2016. [Online]. A vailable: https://www .asme.org/engineering- topics/articles/ bioengineering/top- 6- robotic- applications- in- medicine [355] R. A. Beasley , “Medical robots: current systems and research direc- tions, ” Journal of Robotics , vol. 2012, 2012.
Original Paper
Loading high-quality paper...
Comments & Academic Discussion
Loading comments...
Leave a Comment