Deep learning analysis of coronary arteries in cardiac CT angiography for detection of patients requiring invasive coronary angiography
In patients with obstructive coronary artery disease, the functional significance of a coronary artery stenosis needs to be determined to guide treatment. This is typically established through fractional flow reserve (FFR) measurement, performed during invasive coronary angiography (ICA). We present a method for automatic and non-invasive detection of patients requiring ICA, employing deep unsupervised analysis of complete coronary arteries in cardiac CT angiography (CCTA) images. We retrospectively collected CCTA scans of 187 patients, 137 of them underwent invasive FFR measurement in 192 different coronary arteries. These FFR measurements served as a reference standard for the functional significance of the coronary stenosis. The centerlines of the coronary arteries were extracted and used to reconstruct straightened multi-planar reformatted (MPR) volumes. To automatically identify arteries with functionally significant stenosis that require ICA, each MPR volume was encoded into a fixed number of encodings using two disjoint 3D and 1D convolutional autoencoders performing spatial and sequential encodings, respectively. Thereafter, these encodings were employed to classify arteries using a support vector machine classifier. The detection of coronary arteries requiring invasive evaluation, evaluated using repeated cross-validation experiments, resulted in an area under the receiver operating characteristic curve of $0.81 \pm 0.02$ on the artery-level, and $0.87 \pm 0.02$ on the patient-level. The results demonstrate the feasibility of automatic non-invasive detection of patients that require ICA and possibly subsequent coronary artery intervention. This could potentially reduce the number of patients that unnecessarily undergo ICA.
💡 Research Summary
This paper addresses the clinical need to identify patients with functionally significant coronary artery stenosis who require invasive coronary angiography (ICA) and fractional flow reserve (FFR) measurement, using only non‑invasive cardiac CT angiography (CCTA) data. Current practice relies on CCTA for anatomical assessment, but its low specificity leads to many unnecessary ICA procedures. Existing non‑invasive FFR estimation methods either simulate blood flow, requiring accurate lumen segmentation, or analyze myocardial tissue, both of which have practical limitations.
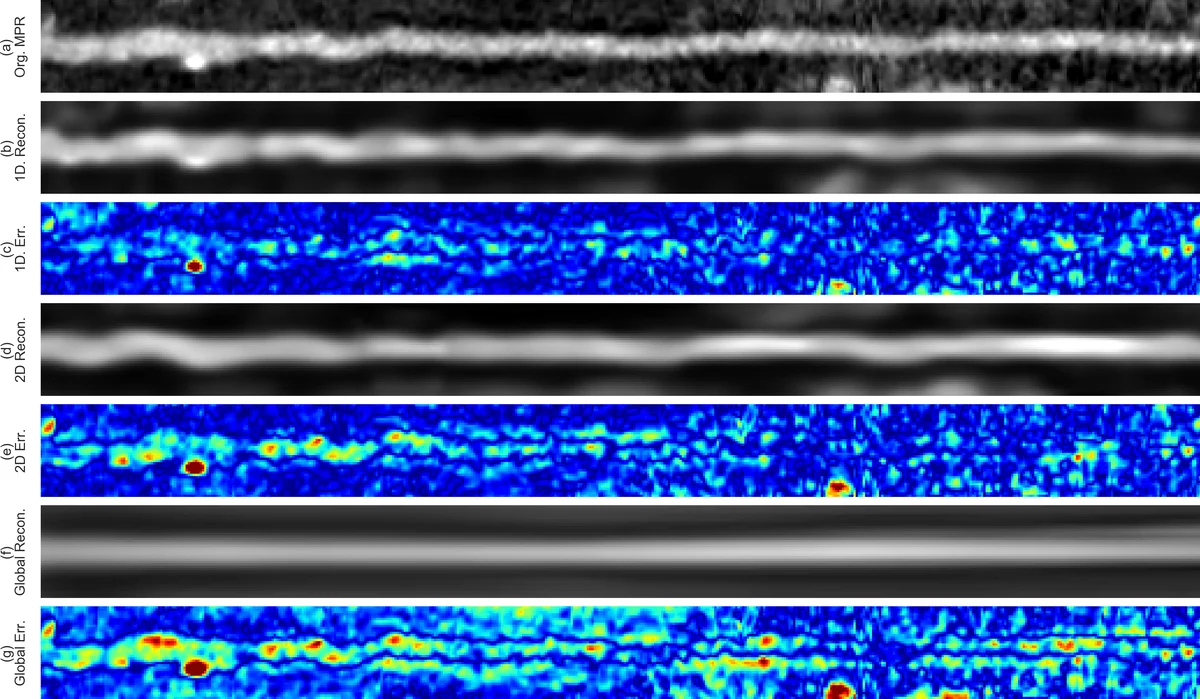
The authors propose a fully automated pipeline that requires only the coronary artery centerline, which can be extracted automatically or semi‑automatically. From the centerline, a straightened three‑dimensional multi‑planar reformatted (MPR) volume is generated for each artery. Because MPR volumes vary widely in length and contain millions of voxels, a single convolutional autoencoder (CAE) cannot efficiently encode them. Therefore, a two‑stage unsupervised encoding strategy is introduced. First, a 3‑D variational CAE (3D‑VCAE) processes small overlapping sub‑volumes along the artery, producing a compact set of latent features for each sub‑segment. Second, a 1‑D CAE (1D‑CAE) receives the sequence of sub‑segment encodings and compresses the entire artery, regardless of its length, into a fixed‑size latent vector.
These latent vectors capture both local spatial characteristics and global sequential patterns of the artery without any explicit lumen segmentation. The vectors are then fed to a support vector machine (SVM) classifier that predicts whether the artery’s minimal FFR value (recorded during ICA) is below the clinical threshold of 0.80, indicating a functionally significant stenosis that warrants invasive evaluation.
The study retrospectively collected CCTA scans from 187 patients (average age 58.6 ± 8.7 years, 145 male) acquired between 2012 and 2016. Of these, 137 patients (192 arteries) underwent invasive FFR measurement up to one year after the CCTA. All scans were performed on a 256‑detector row scanner with isotropic voxel sizes of 0.3 mm³ after straightening.
Performance was assessed using repeated 10‑fold cross‑validation (five repetitions). At the artery level, the method achieved an area under the ROC curve (AUC) of 0.81 ± 0.02; at the patient level, the AUC was 0.87 ± 0.02. These results demonstrate that the proposed unsupervised feature extraction combined with a simple linear classifier can reliably identify arteries and patients that need ICA, matching or surpassing more complex flow‑simulation approaches that require labor‑intensive lumen segmentation.
Key contributions include: (1) a novel dual‑CAE architecture that handles variable‑length 3‑D coronary artery volumes efficiently, and (2) a practical, segmentation‑free workflow that only depends on centerline extraction, making it robust to calcifications, stents, and motion artifacts. Limitations are the single‑center data set, lack of external validation, and reliance on a linear SVM that may not capture highly nonlinear relationships. Future work should explore multi‑center cohorts, deeper neural classifiers, and integration of myocardial tissue features to further improve diagnostic accuracy and clinical translation.
Comments & Academic Discussion
Loading comments...
Leave a Comment