LET-99-dependent spatial restriction of active force generators makes spindles position robust
During the asymmetric division of the Caenorhabditis elegans nematode zygote, the polarity cues distribution and daughter cell fates depend on the correct positioning of the mitotic spindle, which results from both centering and cortical pulling forces. Revealed by anaphase spindle rocking, these pulling forces are regulated by the force generator dynamics, which are in turn consequent of mitotic progression. We found a novel, additional, regulation of these forces by the spindle position. It controls astral microtubule availability at the cortex, on which the active force generators can pull. Importantly, this positional control relies on the polarity dependent LET-99 cortical band, which restricts or concentrates generators to a posterior crescent. We ascribed this control to the microtubule dynamics at the cortex. Indeed, in mapping the cortical contacts, we found a correlation between the centrosome-cortex distance and the microtubule contact density. In turn, it modulates pulling force generator activity. We modelled this control, predicting and experimentally validating that the posterior crescent extent controlled where the anaphase oscillations started, in addition to mitotic progression. Finally, we propose that spatially restricting force generator to a posterior crescent sets the spindle’s final position, reflecting polarity through the LET-99 dependent restriction of force generators to a posterior crescent. This regulation superimposes that of force generator processivity. This novel control confers a low dependence on microtubule and active force generator exact numbers or dynamics, provided that they exceed the threshold needed for posterior displacement. Interestingly, this robustness originates in cell mechanics rather than biochemical networks.
💡 Research Summary
This study investigates how the mitotic spindle is positioned during the first asymmetric division of the Caenorhabditis elegans embryo. The authors demonstrate that spindle positioning is governed by two intertwined control mechanisms: a temporal regulation that depends on the cell‑cycle‑driven changes in the processivity of cortical force generators (FGs), and a spatial regulation that links the spindle’s position to the availability of astral microtubules at the cortex. The spatial control is mediated by the polarity protein LET‑99, which forms a cortical band that restricts active FGs to a posterior crescent region of the cell surface.
Using a combination of genetics, live imaging, and quantitative analysis, the authors first show that delaying anaphase onset with a such‑1 (ANAPC5) mutant uncouples the timing of spindle oscillations from the cell‑cycle clock. In the mutant, oscillations begin well before anaphase, precisely when the posterior centrosome reaches ~70 % of embryo length (EL), the same position observed in wild‑type embryos. This indicates that the onset of pulling‑force–driven oscillations is triggered by the spindle’s position rather than by a fixed temporal cue.
To test whether microtubule dynamics contribute to this positional cue, the authors partially deplete the microtubule nucleation factor SPD‑2 by RNAi. Reduced nucleation shifts the oscillation onset position further posteriorly to ~74 % EL, confirming that the number of astral microtubules influences the distance at which sufficient cortical contacts are made to activate FGs.
A “landing assay” was developed to map microtubule–cortex contacts in vivo. Whole‑cell tubulin fluorescence was imaged with a spinning‑disk confocal microscope, and contacts were detected with the u‑track algorithm. The cortex was divided into ten equal anterior‑posterior (AP) zones, and contact density was calculated for each zone over time. The authors found a strong inverse correlation between the centrosome‑cortex distance and contact density: as the posterior centrosome approaches the cortex, the density of microtubule contacts rises sharply, following an exponential decay with distance. This provides quantitative evidence that microtubule availability at the cortex is a function of spindle position.
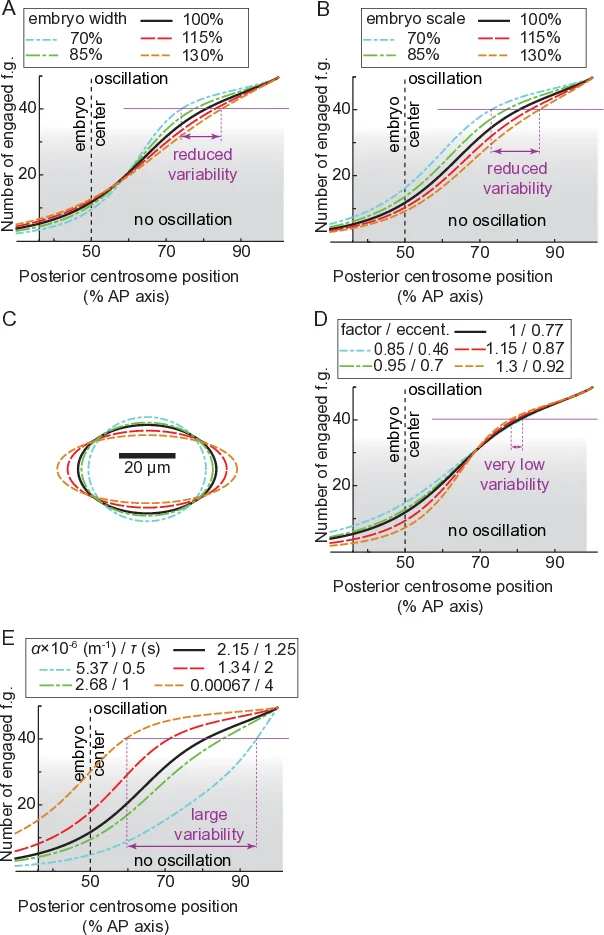
Building on these observations, the authors extend the classic “tug‑of‑war” model of spindle positioning. The new model incorporates (i) a distance‑dependent probability of microtubule contact, P_contact(d) = ρ₀ exp(−d/λ), derived from the landing assay; (ii) a spatial restriction of active FGs to the posterior crescent defined by the LET‑99 band (45–70 % EL excluded, 70–100 % EL allowed); and (iii) a time‑varying processivity parameter that captures the cell‑cycle‑dependent decrease in FG detachment rate. The model treats the anterior and posterior sides separately, assigning different numbers of FGs (Nₐ, Nₚ) and different on‑rates (k_on) while keeping the centering force as a linear spring. Numerical simulations using a TR‑BDF2 integrator reproduce the experimentally observed trajectories of the posterior centrosome, the timing of oscillation onset, and the final spindle displacement.
A key outcome of the model is robustness. When the total number of microtubules or the total number of FGs is varied by ±20 %, the final spindle position changes by less than 3 % of EL, provided that the spatial restriction imposed by LET‑99 is maintained. In contrast, a model lacking spatial restriction is highly sensitive to these variations. The authors further test robustness experimentally by measuring oscillation onset position and timing across embryos of different lengths and at different temperatures. After normalizing by embryo length, both position and timing remain essentially constant, confirming that the spatial control confers mechanical robustness that is largely independent of biochemical fluctuations.
In summary, the paper establishes that (1) the spindle’s posterior position determines when cortical pulling forces become strong enough to generate oscillations; (2) this positional sensing is achieved through a distance‑dependent increase in microtubule contacts at the cortex; (3) LET‑99 creates a posterior crescent that concentrates active force generators, thereby translating the positional cue into a robust mechanical response; and (4) the combination of temporal processivity changes and spatial restriction yields a spindle‑positioning system that is tolerant to variations in microtubule number, force‑generator abundance, and embryo geometry. This dual‑control framework advances our understanding of how physical and molecular cues integrate to ensure reliable asymmetric division, and it provides a quantitative platform for exploring spindle positioning in other organisms and pathological contexts.
Comments & Academic Discussion
Loading comments...
Leave a Comment