Tensor-driven extraction of developmental features from varying paediatric EEG datasets
Objective. Consistently changing physiological properties in developing children's brains challenges new data heavy technologies, like brain-computer interfaces (BCI). Advancing signal processing methods in such technologies to be more sensitive to d…
Authors: Eli Kinney-Lang, Loukianos Spyrou, Ahmed Ebied
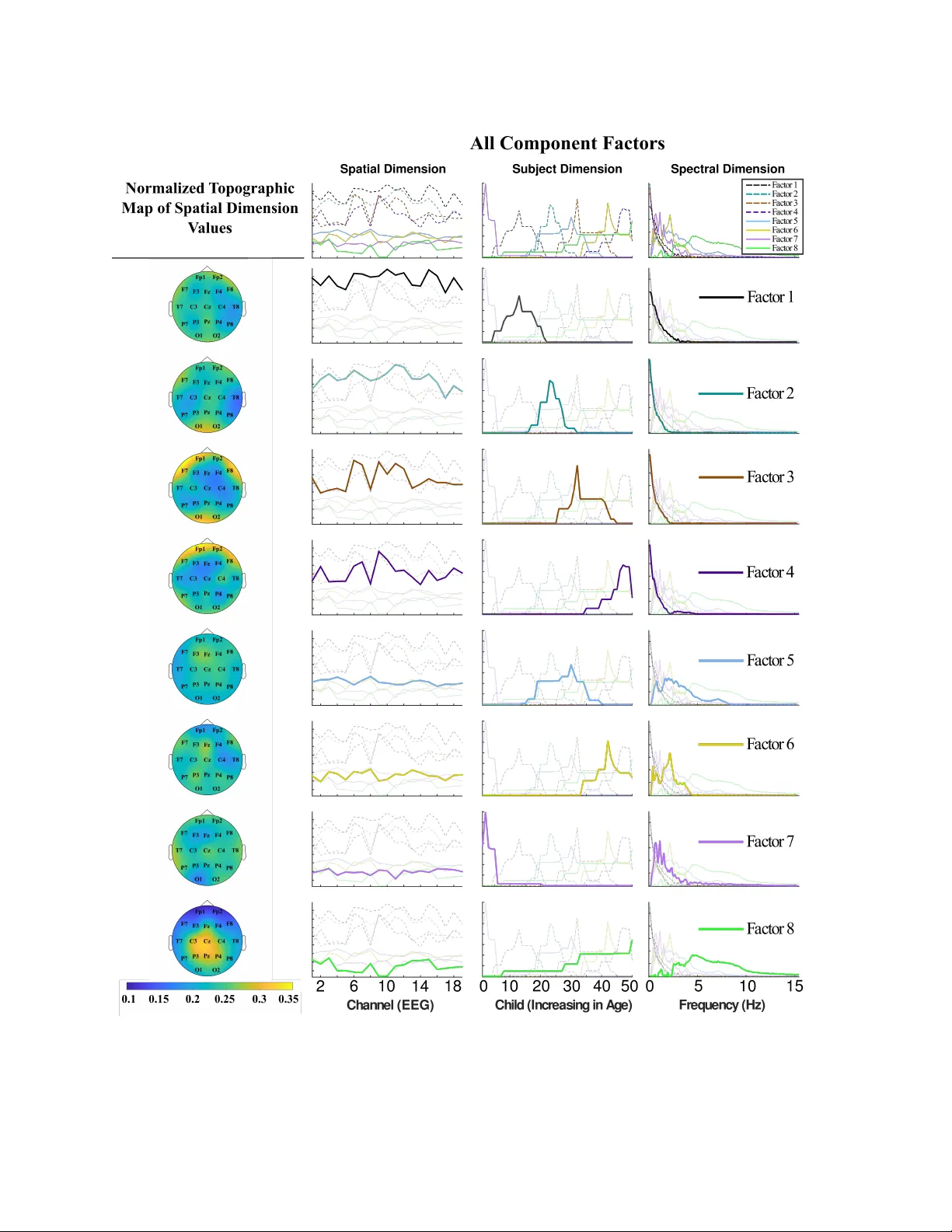
T ensor-driv en extraction of dev elopmen tal features from v arying paediatric EEG datasets E. Kinney-Lang 1 , 2 , ∗ , L. Sp yrou 1 , A. Ebied 1 , R. FM Chin 2 , 3 , and J. Escudero 1 , 2 1 Sc ho ol of Engineering, Institute for Digital Comm unications, The Universit y of Edin burgh, Edin burgh EH9 3FB, United Kingdom 2 The Muir Maxw ell Epilepsy Cen tre, The Univ ersity of Edin burgh, Edin burgh EH8 9XD, United Kingdom 3 Ro y al Hospital for Sic k Children, Edin burgh EH9 1LF, United Kingdom ∗ Corresp onding Author E-mail: e.kinney-lang@ed.ac.uk Abstract. Obje ctive . Consisten tly c hanging ph ysiological properties in dev eloping c hildren’s brains c hallenges new data hea vy tec hnologies, lik e brain-computer in terfaces (BCI). Adv ancing signal pro cessing metho ds in such technologies to b e more sensitiv e to developmen tal changes could help improv e their function and usability in paediatric p opulations. T aking adv an tage of the multi-dimensional structure of EEG data through tensor analysis offers a framew ork to extract relev ant developmen tal features presen t in paediatric resting-state EEG datasets. Metho ds . Three paediatric datasets from v arying dev elopmental states and p opulations were analyzed using a developed t w o-step constrained Parallel F actor (P ARAF AC) tensor decomp osition. The datasets included the Muir Maxwell Epilepsy Cen tre, Children’s Hospital Boston-MIT and the Child Mind Institute, outlining tw o impaired and one healthy p opulation, resp ectiv ely . Within dataset cross-v alidation used supp ort vector machines (SVM) for classification of out-of-fold data predicting sub ject age as a proxy measure of developmen t. t-distributed Sto chastic Neighbour Em b edding (t-SNE) maps complemen ted classification analysis through visualization of the high-dimensional feature structures. Main R esults Developmen t-sensitive features w ere successfully iden tified for the developmen tal conditions of eac h dataset. SVM classification accuracy and misclassification costs were improv ed significan tly for b oth health y and impaired paediatric populations. t-SNE maps rev ealed suitable tensor factorization was key in extracting developmen tal features. Signific anc e The describ ed metho ds are a promising to ol for incorporating the unique dev elopmental features presen t throughout c hildho o d EEG in to new tec hnologies lik e BCI and its applications. Keywor ds: P ARAF A C, feature selection, brain-machine interface, paediatric EEG T ensor-driven extr action of developmental fe atur es fr om varying p ae diatric EEG datasets 2 In tro duction Diseases and injuries sustained in childhoo d are a ma jor public health issue worldwide. The resultan t acute and/or c hronic motor disability affect millions, with early-life sustained motor insult potentially leading to learned non-use of afflicted regions and p otential complications later in life [1, 2]. While traditional therap eutic options for improving motor disabilit y often inv olv e exercise-based techniques [3, 4] lik e constraint-induced mo vemen t therapy (CIM) [1, 5, 6], these exercise-based tec hniques ha ve a trade-off in requiring residual mo vemen t and con trol in the patien t’s afflicted app endage [1, 7]. Brain-computer in terfaces (BCI) and other emerging technologies are strong candidates for non-m uscular neurorehabilitation options in clinical settings [8, 9, 10, 11], with promising early results in adults [12, 13]. Accoun ting for v arying electrophysiological prop erties in dev eloping c hildren, ho wev er, p oses a h urdle for many of these data driven technologies, including BCI. Blending together engineering and medicine, BCIs pro vide direct comm unication c hannels b etw een the brain and an output device, i.e. computer [14], through adv anced signal processing. Man y p opular BCI applications measure and deco de electric p otentials created in the brain, such as sp ecific though t patterns used to in vok e motor imagery (MI) [15, 16, 17]. Hardw are lik e electroencephalography (EEG) record the electric potentials o ver the scalp [14, 15], with user in tent then determined through signal analysis, feature identification, extraction and classification. These BCI signal pro cessing chains often target relatively static electroph ysiological signal features common in adult EEG recordings, relying on their a priori predictabilit y in spatial/temp oral/sp ectral feature selection and determining data outliers. Ho wev er, assuming the adult features ma y not b e appropriate for analyzing p opulations with more v ariable signal features, like children [18]. Signal properties and profiles of the brain are contin ually c hanging from birth through adultho o d [18, 19, 20]. F or example, the lo cation and frequency of the well established EEG alpha rhythm in adults is though t to migrate throughout c hildho o d from appro ximately 6-9 Hz un til reac hing 8-13 in adulthoo d [20, 21, 22]. EEG signal recordings from y oung c hildren are further confounded by high background noise alongside the shifting EEG signal bands [19, 21, 22], resulting in obfuscated and weak er signals of interest. Therefore a means to iden tify and extract EEG features sensitiv e to the changing developmen tal profiles of c hildren w ould b e a critical to ol in constructing paediatric BCI rehabilitation paradigms. T ensor (or m ulti-wa y) analysis [23, 24] provides a p otential framework to capture the dynamic developmen tal profiles in paediatric EEG, through inv estigating the relationships presen t in the multi-dimensional EEG data [25, 26]. T ensor analysis is a higher-order (i.e. m ulti-dimensional/multi-w ay) extension of standard matrix analysis techniques, whic h retains informativ e structural relationships b etw een dimensions (domains or w a ys) in the data [26]. T ensor analysis has already bee n adapted to adult BCI paradigms [26, 23, 27], and thus offers a structure to build to ols tow ards effective paediatric BCI paradigms. This pap er is an extension to our previous conference submission [28] which provided a pro of-of-concept for characterizing developmen tal feature profiles of children using tensor analysis on an EEG dataset. Here an extended, robust feature selection paradigm is presen ted, in tro ducing tensor comp onent selection and mo del v alidation. F urthermore, the improv ed paradigm is demonstrated on several resting-state paediatric EEG datasets which span 1.) a rapidly dev eloping preschool p opulation with p otential dev elopmental impairmen ts; 2.) a p opulation spanning c hildho o d to adultho o d with p oten tial developmen tal impairments; 3.) a healthy p opulation during a stable dev elopmental p erio d of childhoo d. Successful c haracterization of key age-sp ecific features within each dataset supp orts this approach as T ensor-driven extr action of developmental fe atur es fr om varying p ae diatric EEG datasets 3 a p otential adaptive to ol for dev elopment-sensitiv e feature selection in paediatric EEG for applications lik e BCI. Materials and Metho ds Datasets Muir Maxwel l Epilepsy Centr e A retrosp ective analysis of an epileptic preschool cohort ( < 5 y ears) from the Muir Maxw ell Epilepsy Cen tre was included in this study , henceforth referred to as the MMEC dataset. The original cohort w as prosp ectively recruited from National Health Service (NHS) hospitals in Fife and Lothian as part of the NEUR OPROFILES study [29]. A 32-c hannel, unipolar mon tage captured routine EEG in the standard 10-20 system for each child. Of 64 children av ailable, 14 w ere excluded from this study due to corrupted EEG data, inconsistent or incompatible EEG acquisition parameters and irregular recordings, resulting in a dataset of routine EEG from n = 50 presc ho ol c hildren. If multiple resting-state EEG recordings existed, only the first recording was selected for each c hild to a void weigh ting results tow ard children with more recordings and to select from the same a wak e resting-state data across all c hildren. Childr en ’s Hospital Boston-MIT Publicly av ailable data from a study at the Children’s Hospital Boston-MIT of epileptic patien ts from infancy to early adultho o d [30] w as used in this study , do wnloaded through Ph ysiobank.org [31] and henceforth referred to as the CHB- MIT dataset. A 28-channel, bip olar montage captured EEG recordings contin uously ov er t wo days of monitoring. Of the 23 patients a v ailable, 6 sub jects were determined to hav e inconsisten t EEG acquisition parameters for this study , due to discrepancies in the mon tages and unsuitable recordings. This resulted in n = 17 sub jects (age 2-19 y .o.) for analysis. The 48-hour contin uous recordings were separated into 4-hour time blo c ks, with an equal n umber of trials at eac h time selected for processing. Results were a veraged across all time bins to render a holistic representation of the resting-state data for each sub ject. Child Mind Institute Resting-state EEG data for health y control participan ts was taken from the op en science resource provided by [32] and the Child Mind Institute. Data captured from high-densit y 129-channel resting-state paradigms of pre-adolescen t sub jects (one age 6, the rest age 8-11 y .o.) w as used. Of 45 sub jects a v ailable, one sub ject w as excluded (age 11) due to abnormalities in EEG processing, resulting in n = 44 sub jects for analysis. The single 6-y ear-old in the dataset was grouped with the 8-y ear-old class to allow for cross-v alidated classification. A summary of sub ject distribution p er age for each dataset is included in T able 3 of the supplemen tary data. Pr e-pr o c essing Ra w EEG data w as processed using the Fieldtrip to olb o x [33] in Matlab 2015a. A tw o-pass (zero-phase forw ard and rev erse) bandpass filter betw een [0.5-31] Hz w as applied to EEG time- series signals. The filter was detrended, and signals w ere separated into 10-second (5-second) long trials for the MMEC/CHB-MIT (CMI) datasets. EEG c hannels were re-referenced to a common av erage reference and auxiliary/reference sp ecific c hannels were remov ed. The EEG channel montages in the MMEC and CHB-MIT w ere matc hed through adapting the T ensor-driven extr action of developmental fe atur es fr om varying p ae diatric EEG datasets 4 bip olar EEG electrode information from the CHB-MIT to the unip olar MMEC setup. The high-densit y set-up of the CMI data was not adapted to this same conv en tion to av oid p oten tial information loss. Channels in the CMI data with NAN v alues were remov ed. T rials with an y seizure activit y w ere immediately excluded from pro cessing. A m ulti-pass artifact rejection system remo v ed m uscle, jump and o cular artifacts automatically , follow ed by man ual insp ection of data to verify and remo ve any remaining artifacts. Automatic rejection was based on recommended thresholding v alues giv en in Fieldtrip. T ensor Construction Three-w ay tensors consisting of [ S patial ] × [ S pectr al ] × [ S ubj ect ] dimensions were created using the EEG channel, p ow er sp ectra and sub ject age data for eac h dataset, resulting in (19) × (301) × (50), (19) × (301) × (17) and (105) × (61) × (44) elemen ts for the MMEC, CHB-MIT and CMI datasets resp ectively . Figure 1(a) provides a general illustration of the tensor construction. Time-frequency analysis of clean trials using Fieldtrip’s m ultitap er metho d with a 0.5s Hanning windo w provided p ow er sp ectra for each sub ject in the MMEC and CHB-MIT at 0.1 Hz resolution, and 0.5 Hz resolution for the CMI with data normalized. The p o wer sp ectra in eac h sub ject was av eraged across all trials providing a general spectral profile of the resting- state EEG for the [ S pectr al ] domain. The [ S ubj ect ] domain in each dataset was sp ecifically ordered so all sub jects were strictly increasing in age from youngest to oldest. The structure of this domain is critical as a dev elopmental proxy , through whic h features can b e iden tified as age-sp ecific or pan- dev elopmental as indicated by their general asso ciation to age in years. T ensor F actorization: Par al lel F actor Analysis T ensor factorization (decomp osition) is a m ultiwa y extension of standard matrix analysis tec hniques (e.g. principal/indep enden t component analysis) which describ es a mo del of the structural relationship b etw een tensor mo des [23, 25, 34]. A common tensor factorization mo del for EEG data is P arallel F actor Analysis (P ARAF A C, also kno wn as Canonical P oly adic Decomp osition CANDECOMP) [35, 36, 37], which decomposes a tensor X in to a linear com bination of rank-1 tensors coupled with a sup er-diagonal core [35, 36]. EEG data readily supp orts a P ARAF A C mo del due to its inheren t higher-order structure, e.g. relationship b et w een the time-series, c hannels and p ow er sp ectra [37]. Equation (1) illustrates the general R -comp onent P ARAF A C mo del of a 3-dimensional array X ( I × J × K ): x ij k = R X r =1 a ir b j r c kr + e ij k (1) with x ij k , a ir , b j r , c kr ( i = 1 ...I ; j = 1 ...J , k = 1 ...K ) and e ij k as elements of X , domains A ( I × R ), B ( J × R ), C ( K × R ) and residual E ( I × J × K ), resp ectively . Figure 1 B) illustrates tensor decomp osition for v arying comp onen ts R for a 3-dimensional tensor. T ensor datasets were factored using an adapted P ARAF AC function from the NW A Y- to olb o x for Matlab [38]. Sev eral domain constraints were used in analysis to improv e in terpretation of results, and accoun t for domain-specific properties lik e strictly non-negativ e comp onen ts in the p ow er sp ectra. Non-negativit y constraints w ere applied to the [ S patial ] and [ S pectr al ] domains, with unimodality applied to the [ S ubj ect ] domain. Unimo dality was imp osed in order to extract comp onen ts that are b ound to sp ecific age groups within a dataset. T ensor-driven extr action of developmental fe atur es fr om varying p ae diatric EEG datasets 5 The [ S ubj ect ] domain structure p ermitted unimodal constrain ts since no sub jects had rep eated ages (data analyzed at ‘months-old’, group ed into ‘years-old’ for classification). The P ARAF AC mo del decomposition guarantees a 1:1 interaction b etw een extracted factors across domains due to its super-diagonal core [26, 36]. Through imp osing strict zero-v alues in the core tensor on all but the diagonal components (i.e. making the core sup er-diagonal), any given component in a domain can only interact with the corresp onding comp onen t in other domains. In example, in Figure 2 the first factor ( black ) in the [ S patial ] domain corresponds directly with only the first factor ( black ) in the [ S ubj ect ] and [ S pectr al ] domains as a result. Therefore, examining comp onent interactions across domains in P ARAF AC provides direct insight in to the structural relationships within the data. In the presen ted work, this amoun ts to information on the extracted developmen tal features presen t throughout c hildho o d. Exploiting the mild conditions required for uniqueness of the P ARAF A C mo del guarantees that the lo w-rank factor matrices of the P ARAF AC decomp osition retain their meaning [23, 34]. In this prop osal, retaining decomp osition uniqueness allows in terpretation of how the developmen tal [ S ubj ect ] domain influences [ S patial ] / [ S pectr al ] factors. Ev en if the underlying relationships are not obvious in the original EEG data, they are assured to b e viable through the uniqueness condition. Generic uniqueness of P ARAF AC holds under the sufficien t condition [34]: N X n =1 k A ( n ) ≥ 2 R + ( N − 1) (2) for an N -w ay tensor with k elements for eac h domain matrix A ( n ) (i.e. mo de- n rank of A) and R factor comp onents [34]. T ensor F actorization: Comp onent Sele ction Comp onen t selection is a critical step in tensor factorization. Choosing the optimal n umber of comp onent factors for P ARAF A C decomp osition balances mo del suitabilit y with prop er represen tation of latent structural information. The unkno wn underlying developmen tal profiles in the data tensors are b est captured b y an unkno wn n umber of components, e.g. some non-minimal rank R decomp osition. In example, assume that there is a kno wn n umber of EEG ‘sources’, s underlying the p ow er sp ectra of the [ S pectr al ] domain. Each source s could be describ ed b y exactly one rank-1 tensor b y selecting r = s comp onen ts for tensor decomp osition. Ho wev er, if there are more sources s than comp onents r (i.e. to o few comp onents c hosen) then the mo del ma y obscure the less obvious, but still important, structural relationships. Similarly , if to o man y components are c hosen, this may lead to o ver-fitting the model. T o help gauge viable comp onent selection of a P ARAF A C mo del, the core consistency diagnostic (corcondia) is used [39]. Corcondia pro vides a direct measure of the suitability of a specific P ARAF A C mo del in an easy to in terpret fashion b y describing the degree to whic h the appro ximate tensor mo del deviates from its ‘true’ sup er-diagonal core [39]. The v alidity of the mo del can then b e determined, with mo dels under 40% corcondia considered as generally non-viable [39]. Corcondia may also provide insigh t into the p oint in which laten t structural relationships ha ve b een matc hed uniquely to underlying component ‘sources’. Corcondia has a prop erty of p ermanen tly and sharply decreasing after some maxim um n um b er of comp onents is chosen [39]. This decrease is not necessarily monotonic, but rather assured to nev er b e b etter than the corcondia giv en at said maxim um n umber of comp onents. This w as considered to be the p oin t in which the maximu m num b er of p ossible underlying relationships hav e b een taken T ensor-driven extr action of developmental fe atur es fr om varying p ae diatric EEG datasets 6 in to account, i.e. there is likely maximum matching b etw een comp onents and underlying sources. Therefore, the n umber of comp onents immediately prior to the downfall can serve as an inflection p oint for determining viable versus non-viable mo dels, as done in this study . A tw o-step analysis c hain ov er a v arying set of comp onents ( r = 1 : 20) w as constructed for this study to iden tify P ARAF A C models whic h accoun ted for the maximum n umber of pan- dev elopmental structures in the tensor while maintaining acceptable model viability . First, for eac h num b er of components r , five P ARAF A C decomp ositions were run sim ultaneously with their corcondia and explained v ariance recorded. Using multiple simultaneous runs help ed accoun t for p otential mo del conv ergence to lo cal minimums. Any mo del with corcondia b elow 70% was considered to b e non-viable and w as remo ved. The b est fit mo del w as considered to b e the maximum rank decomp osition mo del still ab ov e the 70% corcondia. Then, a thresholding metho d based on our proof-of-concept w ork [28] w as applied to select only the subset of factors whic h spanned the unimo dal [ S ubj ect ] domain. Reduction of the factors to a dev elopment- sp ecific subset help ed remo ve any features reflecting prop erties of a single c hild, which could o ccur due to the unimo dal constrain t [28]. The reduced P ARAF A C model then had its new corcondia ev aluated, and the reduced mo del with the largest R -comp onents maintaining corcondia ab o v e 70% in b oth steps was selected for analysis and training. Figure 1(b) and Figure 1(c) outline the grid search, corcondia ev aluation and optimal mo del selection pro cess. Supplemen tal Figure 5 shows full and reduced tensor model viabilit y based on corcondia calculations for m ultiple comp onents r for one dataset. Classific ation A mo dified direct pro jection of the extracted training factors on to the testing comp onents w as used for prediction v alidation, stemming from [40]. Direct pro jection traditionally includes a pseudo-in verse step, whic h inherently introduces negative testing comp onent v alues [40]. Giv en the non-negative constrain ts on the training tensor decomp osition, to retain meaningful prediction the pro jected test comp onen ts must also b e non-negativ e. Therefore, the non- negativ e least square (NNLS) solution of the Khatri-Rao pro duct ( ) betw een the non- [ S ubj ect ] dimensions (3) from the training tensor w as in tro duced as an alternative to the pseudo-in verse step. Using the NNLS maintains the [ S patial ] and [ S pectr al ] non-negativ e domain constraints while still fulfilling the same approximate function as the pseudo-inv erse. The NNLS solution w as then m ultiplied b y the [ S ubj ect ]-domain matricized test tensor (4), resulting in a predicted [ S ubj ect × F actor ] matrix for v alidation (5). Results from (3) w ere then multiplied by the [ S ubj ect ]-domain matricized test tensor (4), resulting in a new predicted [ S ubj ect × F actor ] non-negativ e matrix for v alidation classification. P r oj ectedF actor train = N N LS ([ T r ain spectral ] T [ T r ain spatial ]) (3) T est matrix = [ T est subj ect ] × [ T est spatial · spectr al ] (4) P r edicted test = P r oj ectedF actor train × T est matrix (5) T o maintain stringent in tegrit y for classification, data was split into training and testing cross-v alidation folds prior to tensor decomp osition. A m ulti-class, ordinal classification sc heme w as devised to ev aluate the tensor extracted factor’s ability to predict sub ject age using the W ek a to olb o x [41, 42]. Sub ject age (in y ears) was used for within dataset class lab els. An ordinal cost-matrix was used to account for the multi-class, ordinal nature of eac h data constructed tensor. The ordinal cost-matrix p enalized misclassification through linearly weigh ted differences based on class age, thereb y increasing classification penalties for T ensor-driven extr action of developmental fe atur es fr om varying p ae diatric EEG datasets 7 Figure 1. P ARAF A C tensor construction and decomposition flow chart. A) Constructing a 3-dimensional tensor from raw resting-state time-series EEG with [ S patial ] × [ S pectr al ] × [ S ubj ect ] domains on the X , Y , Z axes resp ectively . The [ S ubj ect ] domain is ordered to increase strictly with age. B) Illustration of R = 1 : N comp onen t grid search, where eac h R -factorized mo del is used to reconstruct the original data to test corcondia and mo del suitability . C) Reduction of R N -comp onen ts to a optimal subset R M -comp onen ts (with M < N ) based on a threshold [ S ubj ect ] domain and mo del v alidation via corcondia. F ull resolution figure a v ailable up on request. T ensor-driven extr action of developmental fe atur es fr om varying p ae diatric EEG datasets 8 predicting sub jects as drastically older/younger compared to their actual age. The cost-matrix w as matc hed for each dataset to the unique sub ject ages in that dataset. Using the ordinal cost-matrix, a non-linear radial basis function (RBF) supp ort v ector mac hine (SVM) w as trained using the decomp osed P ARAF A C factors for eac h cross-v alidation fold in each dataset. The RBF-SVM w as optimized using a grid search in W ek a to find C and γ whic h pro vided the highest classification accuracy . Results w ere ev aluated on their o verall c lassification accuracy and total p enalty costs (e.g. the sum of all misclassification p enalties based on the ordinal cost-matrix). Random classification and naive classification (e.g. choosing a single class for all sub jects) is included for comparison. Results are reported as a v erages across all training folds with standard deviation and a tw o-tailed Student’s t -test to infer differences from random and naive classification. The distribution of sub jects p er age in the MMEC and CMI datasets allow ed for 4-fold and 5-fold stratified cross-v alidation resp ectively . The CMI classification included the single six-y ear-old as a mem b er of the ‘Age 8’ class to retain stratified cross-v alidation. Comparative classification within the CHB-MIT data was not p ossible due to the limited sub jects p er age. Visualization T o complemen t classification of the extracted high-dimensional tensor features across the [ S patial ] , [ S pectr al ] , [ S ubj ect ] domains, results from factorized training folds for the MMEC and CMI datasets are display ed using t-distributed Sto chastic Neighbour Embedding (t-SNE) [43]. Using t-SNE, high-dimensional data can be visualized to capture both the lo cal and global structure of the data through presence of clusters at sev eral scales. Demonstrating t-SNE maps on individual training folds in the data offers a visual companion to the classification analysis, sho wing the lo cal and global structure underlying a single fold used in analysis. Simulations A simulation of pseudo-EEG data accompanies the real-world datasets. The Berlin Brain Connectivit y Benc hmark (BBCB) sim ulation co de [44] w as modified to include a shifting sp ectral frequency band of in terest, similar to the alpha frequency seen in developmen t [19, 21, 20, 22]. The band of in terest lo w er bound was set with mean ± 1 standard deviation as the sim ulated age, up to 8 years-old. Afterwards, each additional simulated year increased the sampling mean thereby gating the lo wer b ound tow ards 8-Hz. The upp er bound was set at 3-Hz ± 2 standard deviations higher than the lo wer b ound. The upp er b ound v ariation was at least 1-Hz ab o ve the low er b ound. T en children were simulated p er age using the mo dified co de, from 5 to 11 years-old. Simulated EEG was conv erted to Fieldtrip for processing, with a sim ulated tensor constructed in an identical fashion to the real-world datasets. Results The prop osed tensor analysis successfully identified laten t dev elopmental features across sub jects indep enden tly for each dataset. A detailed visual breakdo wn of the P ARAF A C mo del decomp osition and its resulting ‘developmen tal profile’ snapshots is given in Figure 2 as an example, using one entire dataset (MMEC). Qualitative developmen tal feature profiles are illustrated in each tensor domain, where actual weigh ted v alues in the testing folds were used for classification purposes. Individual factor con tributions are sho wn in the extended profiles to help clarify the latent dev elopmen tal relationships in each domain. Comp onen t profiles in T ensor-driven extr action of developmental fe atur es fr om varying p ae diatric EEG datasets 9 the [ S ubj ect ] domain reflect in which ages the extracted factor (feature) is most dominant and influen tial. F eatures of the [ S ubj ect ] domain are ordered to matc h highest to low est explained v ariance from the [ S patial ] domain. The normalized top ographic map of the [ S patial ] domain sho ws relative regional con tributions of EEG c hannels for each ‘dev elopmental feature profile’ (note this is not a top ographic map of EEG activit y). The [ S pectr al ] domain is shown up to 15 Hz, as higher frequencies for preschool children in resting-state data has little activit y of in terest and remains fairly flat. Pr escho ol childr en with epilepsy: The Muir Maxwel l Epilepsy Centr e data Classification w as significantly improv ed using the iden tified developmen tal factor profiles as features in the MMEC dataset. T able 1 contains the av erage classification results for the MMEC extracted features across all folds. The P ARAF AC extracted features improv ed up on random classification by appro ximately 50%, and reduced total p enalty cost by 9.5 p oints or 37% compared to naive classification (Student’s t -test, p < 0 . 05). T o put the reduced p enalt y cost in another context, if every test sub ject w as misclassified b y appro ximately 2 y ears using naiv e c lassification, P ARAF AC reduced misclassification to approximately only 1.3 y ear for each test sub ject. Naive classification accuracy w as impro ved up on using SVM and the extracted features by appro ximately 37% as w ell (Student’s t -test, p < 0 . 01). Average corcondia across training folds was 85 . 74 ± 4 . 86. Classification Penalt y Cost Accuracy (%) SVM 16.0 ± 4 . 1 30.0 ± 3 . 5 Random – 20.1 ± 0 . 7 ? Naiv e 25.5 ± 1 . 7 ? 22.0 ± 3 . 6 ?? T able 1. Average classification results across all MMEC cross-v alidation folds for SVM, Random and Naive classification for epileptic children 0-5 years of age. No p enalt y costs a v ailable for random classification due to its random nature. ? Indicates significan t difference from SVM using studen t’s t -test at p < 0 . 05. ?? Indicates significan t difference from SVM using studen t’s t -test at p < 0 . 01. While only a third of sub jects w ere on av erage correctly classified in the MMEC dataset, the significantly reduced cost p enalties indicate a mo v e tow ard reductions in gross age misclassification (e.g. classifying a sub ject age 0 as age 3, 4, or 5). This is critical, as impro ved misclassification penalties indicate mistakes trended more to wards closely related ages (i.e. ± 1 or 2 years) more often. These impro vemen ts along with the factor profiles imply success in iden tifying developmen tally imp ortant features of preschool children’s EEG. Child-to-A dult epilepsy sp e ctrum: The CHB-MIT data Results of the CHB-MIT dataset w ere used to demonstrate the scalable nature of the proposed analysis across a broad age range, for sub jects under otherwise similar conditions (e.g. epilepsy afflicted sub jects with matc hed EEG montages). Due to the limited n umber of sub jects for eac h age (only one to tw o - see Supplemental T able 3), meaningful classification w as not p ossible for the CHB-MIT dataset. Instead a qualitativ e illustration of the general developmen tal profile trends is presen ted in Figure 3. The extracted factor profiles reflecting dominating influences at sp ecific ages whic h matc h exp ected dev elopmental patterns [20, 21] indicate T ensor-driven extr action of developmental fe atur es fr om varying p ae diatric EEG datasets 10 Figure 2. Detailed visualization of P ARAF AC extracted dev elopmental feature profiles of epileptic children from 0-5 years old from the MMEC dataset. Normalized top ographic maps of the [ S patial ] domain in column 1 show EEG c hannel regions with higher/lo w er relative contribution for eac h individual feature. The [ S ubj ect ] domain x-axis is the child’s num b er (e.g. Child 1, Child 2), organized by increasing age. The combined and separated feature profiles for the [ S patial ] × [ S ubj ect ] × [ S pectr al ] domains are sho wn in columns 2-4. F ull resolution figure av ailable up on request. T ensor-driven extr action of developmental fe atur es fr om varying p ae diatric EEG datasets 11 Figure 3. A compact visualization of the key feature profiles from the CHB- MIT dataset for epileptic sub jects from age 2 to 19. A normalized top ographic map reflecting relative contributions of each EEG channel region to the extracted factor is sho wn in the first column. Combined and separated feature profiles for the [ S patial ] × [ S ubj ect ] × [ S pectr al ] domains are shown in columns 2-4. F ull resolution figure a v ailable up on request. successful characterization of developmen t sensitiv e features. These results reinforce the likely generalizable nature of the tensor analysis. Pr ofiles Figure 3 demonstrates the P ARAF A C model decomp osition of the CHB-MIT dataset in a condensed format. Key developmen tal feature profiles are emphasized across the extracted feature domains, with probable ‘bac kground’ profiles unaccen ted. The key feature profiles hav e b een organized b y the [ S ubj ect ] domain, with influen tial features prominen t in early childhoo d to early adultho o d ordered from top to b ottom. The exact age of each sub ject is presen t on the [ S ubj ect ] domain axis. Sharp p eaks in the [ S pectr al ] domain at 16, 19, 28 Hz are p otentially residual artifacts from the time-frequency analysis and NAN-a veraging across time-bins. Corcondia was 73.49 for the extracted factors, with 85% explained v ariance. Scho ol-age c ontr ols: The Child Mind Institute Data Dev elopmental features w ere successfully iden tified for the health y control CMI dataset via P ARAF A C. While the a v ailable control data could not b e age-matched to the MMEC for T ensor-driven extr action of developmental fe atur es fr om varying p ae diatric EEG datasets 12 direct comparison, factor profiles found in the CHB-MIT data (seen in Figure 3) at ages similar to CMI sub jects supp orted analyzing the CMI dataset for feasibilit y in identifying dev elopmentally sensitiv e features in normally developed c hildren. T able 2 con tains the av erage classification results across the training/testing folds for CMI sub jects. P ARAF A C extracted features impro v ed significan tly upon random classification by appro ximately 25%, and reduced total p enalty cost by 2 p oints or 19% compared to naive classification. Average corcondia across training folds was 78 . 25 ± 6 . 57. Although classification results had smaller improv ements in classifier p erformance and p enalt y reduction compared to the epileptic p opulation of the MMEC dataset, significant impro vemen ts were still presen t using the P ARAF AC model for SVM classifier training. Extra difficult y in discerning dev elopmental differences in the CMI data was to be expected, giv en the smaller dev elopmental window and higher homogeneity of sub jects in the CMI dataset. Therefore, despite only marginal b o ons in accuracy and p enalt y costs, the classification impro vemen ts in the CMI dataset under P ARAF AC are imp ortan t. The p ositive results serv e as evidence that the prop osed tensor analysis is accessible to ‘normal’ developing c hildren. The CMI dataset results also further illustrates the scalable nature of the tensor analysis, demonstrating successful feature extraction on sub jects more similar dev elopmentally (i.e. health y , b etw een the ages of 8-11 y .o.) compared to the other datasets. Physiological changes across this age span are significantly less drastic compared to b oth the MMEC developmen t windo w from infancy to early childhoo d (age 0-5 y .o.), and the CHB-MIT dev elopmen t windo w from infancy to early adult ho o d (age 2-19 y .o.). Classification Penalt y Cost Accuracy (%) SVM 9.6 ± 0 . 9 34.2 ± 1 . 9 Random – 27.4 ± 1 . 3 ?? Naiv e 11.8 ± 1 . 1 ? 29.8 ± 3 . 6 T able 2. Average classification results across all CMI cross-v alidation folds for SVM, Random and Naive classification b etw een healthy children 8-11 years of age. No p enalt y costs are a v ailable for random classification due to its random nature. ? Indicates significan t difference from SVM using studen t’s t -test at p < 0 . 05. ?? Indicates significan t difference from SVM using studen t’s t -test at p < 0 . 01. t-SNE Visualization F eatures from the first training fold tensor decomposition of the MMEC and CMI datasets are display ed as t-SNE maps in Figure 4(b) and Figure 4(d) resp ectiv ely . Both t-SNE maps demonstrate strong lo cal grouping of different age groups. Maps of EEG data prior to tensor factorization (Figure 4(a)) and when the decomp osition has a randomly ordered [ S ubj ect ] domain in the MMEC data (Figure 4(c)) are included for comparison. The results demonstrate significan tly impro v ed feature grouping and clusters using the prop erly ordered tensor decomp osition metho dology compared to using the raw EEG or random ordered [ S ubj ect ] domain. The t-SNE maps help illustrate how SVM classification from the raw EEG time-series data using all frequency and spatial features do not p erform b etter than random classification in the MMEC. Similar patterned results were found for CMI data. T ensor-driven extr action of developmental fe atur es fr om varying p ae diatric EEG datasets 13 Figure 4. t-SNE Map visualizations with features grouped b y sub ject age. (a) Map of EEG data prior to tensor decomp osition of the MMEC dataset. (b) Map of P ARAF A C extracted dev elopmental features of the MMEC dataset, using the first cross-v alidaiton training fold. (c) Map of P ARAF AC extracted developmen tal features of the MMEC dataset when the original tensor has a randomly ordered [ S ubj ect ] domain. (d) Map of P ARAF AC extracted features from the CMI dataset using the first cross-v alidation training fold. F ull resolution figure av ailable up on request. Simulation A 3-comp onent P ARAF A C mo del revealed the underlying ‘ground truth’ developmen tal profiles built in to the simulation tensor data. Resulting component factors are presented in Supplemen tal Figure 6. Corcondia w as 99% for the mo del, with approximately 23% explained v ariance. These v alues are understandable for the mo del, as the BBCB pseudo-EEG simulation w as designed to retain trilinearity in the data while introducing multiple levels of noise at the frequency band, brain bac kground and sensor la y ers. Replicating developmen tal profile extraction results in the sim ulated data grants further support to our conclusions in real-w orld datasets. Discussion The tensor analysis outlined in this pap er lays a foundational framework capable of extracting latent structures and features associated with dev elopment in paediatric EEG. The unsup ervised nature of this framework op ens the do or to encourage b etter p ersonalized paradigms for data-driv en technologies aimed at paediatric populations. Capitalizing on these developmen ts could help translate new tec hnologies to children, which are sensitive to dev elopmental features in EEG. T ensor-driven extr action of developmental fe atur es fr om varying p ae diatric EEG datasets 14 T ensor analysis derives informative ‘development fe atur e pr ofiles’ of p ae diatric subje cts The factor profiles deriv ed in this study confirm the prop osed P ARAF A C decomp osition sim ultaneously accoun ts for bac kground EEG noise and shifting frequency bands across sub ject age, often explaining more than 85% of the data v ariance. Lo w-frequency , high p o wer sp ectral activit y typically asso ciated with bac kground EEG noise can b e seen in Figure 2, with factors 1-4 reflecting decaying p ow er curv es in the [ S pectr al ] domain, coupled with relativ ely strong con tributions across all channels in the [ S patial ] domain. These factors are likely characterizing general ‘bac kground’ EEG noise indep endent to p oten tial signals of interest (e.g. factors 5- 8). Critically , the [ S ubj ect ] domain demonstrates the tensor analysis has sensitivity to subtle dev elopmental differences, since the features uniquely corresp ond to distinct age groupings, ev en within the p otential background noise. Shifts in spectral p ow er and frequency due to developmen t [19, 21, 22] are seen in the qualitativ e factor profiles of b oth the MMEC and CHB-MIT datasets. In the MMEC dataset, for example, the v ery low frequency , high p ow er dominated sp ectral profile asso ciated with infan t and early-life EEG recordings seem to b e reflected b y F actor 7 in the [ S ubj ect ] and [ S pectr al ] domains. Meanwhile, factor 5 (ligh t blue) is centrally lo cated in the [ S ubj ect ] domain (co vering approximately ages 1-3) and spans the 3-7 Hz range of the [ S pectr al ] domain. F actor 5 therefore highlights the lik ely dominant frequency range for those ages, while also reflecting a shift in p ow er tow ards higher frequencies, whic h is exp ected with gro wing. F actor 8 illustrates a further shift to wards higher frequencies contributing more to the sp ectral profile, as it steadily increases for sub jec ts 31-50 (appro ximately ages 3-5 y .o.) in the [ S ubj ect ] domain. Also, factors 6 and 8 may represen t the b eginning separation b etw een classical EEG bands of in terest, i.e. the delta/theta bands and the alpha/b eta bands resp ectively . Similar developmen tal shifts are also seen in the CHB qualitative feature profiles. The k ey component factors show significantly reduced spectral pow er contributing more in higher [ S ubj ect ] domain ages, alongside a shift in the [ S pectr al ] dimension to wards higher frequencies. These extracted profiles are reflectiv e of the traditional mo vemen t and prev alence of the classical EEG bands, like alpha, throughout child developmen t into adultho o d [21, 22]. Impr ove d classific ation r esults verify tensor extr acte d fe atur es’ sensitivity to development Impro vemen ts in classification coupled with the obtained tensor profiles of b oth impaired (epileptic) and healthy children indicate age-sp ecific factors unco vered from EEG via P ARAF A C con tain structural information on latent dev elopmental relationships. The scalable nature of the proposed analysis sho wed promise in iden tifying relev ant features to dev elopmen t across v arying dev elopmental conditions, including b oth afflicted/health y p opulations, and at slo w/rapid developmen tal windows. With b etter characterization of such features for the paediatric sub jects comes a stronger case for translating signal pro cessing and machine learning applications to children. Clear supp ort for this is seen in the comparison b etw een t-SNE maps for the full feature ra w EEG time-series data and the P ARAF AC pro cessed data. Using the feature-full ra w EEG time-series without processing led to complete failure in iden tifying developmen tal features. The resulting t-SNE map has no discernible structure with random clusters and groupings. Characterization of the underlying developmen tal profile was rendered completely imp erceptible, whic h is likely reflected in the classification b eing no b etter than chance. On the other hand, the highly- structured t-SNE maps of the tensor extracte d features for b oth the MMEC and CMI dataset T ensor-driven extr action of developmental fe atur es fr om varying p ae diatric EEG datasets 15 reflect w ell characterized developmen tal profiles, which can b e utilized in mac hine learning applications. Imp ortan tly , the randomly ordered [ S ubj ect ] domain t-SNE map suggests that successful iden tification of key developmen tal features is not inherent to tensor factorization itself. Although the ov erarching global structure is capable of b eing iden tified, as seen in the similarit y of global shap es of Figure 4(b) and Figure4(c)), the lo cal grouping is completely lost. Rather, the viability of determining developmen tally sensitiv e features relies on prop er construction of the [ S ubj ect ] domain e.g. having an inheren t proxy to growing like strictly increasing sub ject age. F uture work could in vestigate the effects of altering the inheren t structure of the [ S ubj ect ] domain to reflect other developmen tal mark ers, suc h as cognitiv e or b ehavioural scores. Exploiting the structural information from higher-order tensors constructed with careful construction of the [ S ubj ect ] domain as a proxy measure for child developmen t provides a metho dological framew ork designed to enhance sensitivit y to ph ysiological c hanges common throughout childhoo d. Higher sensitivity to these unique developmen tal profiles could b e useful in applications lik e BCI. Through using a framew ork built to determine a curren t child’s dev elopmental state at an electroph ysiological lev el, the BCI could b e automatically tuned and w eighted appropriately for sub jects at differen t p oints along developmen t. Additionally , the results could pro vide ‘health y dev elopment curves’ in studies for comparison to p oten tially dev elopmentally impaired c hildren. T ogether these findings expand up on our previous results [28]. Our improv ed metho dology is v erified using m ultiple datasets. Results indicate the new prop osed metho ds can account for dev elopmental differences in bac kground EEG and shifting sp ectral signals for children under a v ariety of differen t developmen tal conditions. The classification results illustrate a means for dev elopmental feature extraction sensitive to progressive changes, while the profiles provide informativ e context regarding the relationship b etw een [ S patial ] and [ S pectr al ] structures relativ e to sub ject age and developmen t. Limitations Limitations in this study included restricted access to age/acquisition-matc hed paediatric datasets and the heterogeneit y asso ciated with epilepsy in sub jects. Due to limited resources no direct age/acquisition-matc hed health y control data was a v ailable for analysis to compare directly . T o mitigate these drawbac ks, how ev er, m ultiple publicly av ailable datasets were used to demonstrate the prop osed metho dology in multiple settings. The CHB-MIT dataset built up on our analysis from the MMEC across a wider age range using a similar disease condition, while the CMI dataset represented healthy con trol within the b ounds of c hildho o d (but not age-matc hed). F uture work using a more homogeneous p opulation with age-matc hed controls could help further v alidate the results, along with data from b oth resting-state and even t- related EEG. Conclusion Adv anced signal pro cessing, lik e P ARAF A C, com bined with machine learning can help distinguish non-ob vious developmen tal patterns from c hild EEG data. This study prop oses tensor analysis can provide an intuitiv e sense of the latent dev elopmen tal relationships in paediatric EEG data, and pro vide a wa y for developmen t-sensitive feature extraction. The results indicate successful identification of factor profiles and b enefits to classification analysis for a wide v ariet y of developmen tal conditions, including b oth afflicted and health y paediatric T ensor-driven extr action of developmental fe atur es fr om varying p ae diatric EEG datasets 16 p opulations. This study la ys a metho dological framew ork whic h could improv e applications for c hildren reliant on EEG pro cessing and analysis, lik e BCI. F urther dev elopment on this framew ork could help impro ve BCI application sensitivit y to dev elopmen tal changes b y setting the groundwork for a ‘dev elopmen tal domain’ for tensor-based EEG classification in BCIs (e.g. [45]). These adv ances could help immensely in translating the BCI tec hnology to paediatric p opulations and pa ve the w ay for dev elopment of more readily accessible, effectiv e rehabilitation strategies in ternationally . Ac knowledgemen t F unding supp ort for this pro ject was provided by the RS McDonald T rust, Thomas Theo dore Scott Ingram Memorial F und and the Muir Maxwell T rust. The authors would like to thank the c hildren, parents, clinicians and researchers who volun teered and comprised each of the datasets, and the Muir Maxwell Epilepsy Cen tre for providing access the MMEC dataset. The authors also thank Ephrem Zewdie for his comments and suggestions on figures. References [1] Kathryn Y Manning, Darcy F ehlings, Ronit Mesterman, Jan Willem Gorter, Lauren Switzer, Craig Campb ell, and Ravi S Menon. Resting State and Diffusion Neuroimaging Predictors of Clinical Impro v emen ts F ollowing Constraint-Induced Mo vemen t Therap y in Children With Hemiplegic Cerebral P alsy . J. Child Neur ol. , 30(11):1507–14, 2015. [2] Michael V. Johnston. Clinical disorders of brain plasticity. Br ain Dev. , 26(2):73–80, 2004. [3] Martin Fl¨ uc k. F unctional, structural and molecular plasticit y of mammalian skeletal m uscle in resp onse to exercise stim uli. J. Exp. Biol. , 209(Pt 12):2239–48, 2006. [4] Diane L Damiano. Activity , Activit y , Activity: Rethinking Our Ph ysical Therapy Approach to Cerebral P alsy. Phys. Ther. , 86(11):1534–1540, Nov ember 2006. [5] Edward T aub, Gitendra Usw atte, and Thomas Elb ert. New T reatments in Neurorehabilitation F ounded on Basic Researc h. Nat. R ev. Neur osci. , 3(3):228–236, 2002. [6] Diane L. Damiano. Rehabilitative Therapies in Cerebral Palsy: The Go o d, the Not As Go o d, and the P ossible. J. Child Neur ol. , 24(9):1200–1204, 2009. [7] Jos´ e Del R. Mill´ an, R Rupp, G R M ¨ uller-Putz, R Murray-Smith, C Giugliemma, M T angermann, C Vidaurre, F Cincotti, A K ¨ ubler, R Leeb, C Neup er, K-R M ¨ uller, and D Mattia. Combining Brain-Computer Interfaces and Assistive T echnologies: State-of-the-Art and Challenges. F r ont. Neur osci. , 4(September):1–15, 2010. [8] Kai Keng Ang, Karen Sui Geok Ch ua, Kok So on Phua, Chuanc hu W ang, Zheng Y ang Chin, Christopher W ee Keong Kuah, Wilson Low, and Cuntai Guan. A Randomized Controlled T rial of EEG-Based Motor Imagery Brain-Computer In terface Robotic Rehabilitation for Strok e. Clin. EEG Neur osci. , 46(4):310–20, 2014. [9] Natalie Mrachacz-Kersting, Ning Jiang, Andrew James Thomas Stev enson, Imran Khan Niazi, Vladimir Kostic, Aleksandra Pa vlovic, Sasa Radov anovic, Milica Djuric-Jo vicic, F ederica Agosta, Kim Dremstrup, and Dario F arina. Efficien t neuroplasticity induction in c hronic stroke patien ts b y an asso ciativ e brain-computer in terface. J. Neur ophysiol. , page jn.00918.2015, 2015. [10] Jonathan R. W olpaw. Brain-computer interfaces as new brain output path w a ys. J. Physiol. , 579(3):613–619, 2007. [11] L. E H v an Dokkum, T. W ard, and I. Laffont. Brain computer interfaces for neurorehabilitation its current status as a rehabilitation strategy p ost-stroke. A nn. Phys. R ehabil. Me d. , 58(1):3–8, F ebruary 2015. [12] Brittany M Y oung, Zac k Nigogosy an, L ´ eo M W alton, Jie Song, V eena A Nair, Scott W Grogan, Mitc hell E Tyler, Doroth y F Edwards, Kristin Caldera, Justin A Sattin, Justin C Williams, and T ensor-driven extr action of developmental fe atur es fr om varying p ae diatric EEG datasets 17 Viv ek Prabhak aran. Changes in functional brain organization and b eha vioral correlations after rehabilitativ e therap y using a brain-computer in terface. F r ont. Neur o eng. , 7:26, July 2014. [13] Surjo R. So ek adar, Niels Birbaumer, Marc W. Slutzky , and Leonardo G. Cohen. Brain-machine in terfaces in neurorehabilitation of strok e. Neur obiol. Dis. , 83:172–179, 2015. [14] Jonathan R W olpaw, Niels Birbaumer, Dennis J McF arland, Gert Pfurtsc heller, and Theresa M V aughan. Braincomputer interfaces for communication and control. Clin. Neur ophysiol. , 113(6):767–791, 2002. [15] Luis F ernando Nicolas-Alonso and Jaime Gomez-Gil. Brain computer in terfaces, a review. Sensors (Basel). , 12(2):1211–79, 2012. [16] Minkyu Ahn, Mijin Lee, Jin young Choi, and Sung Chan Jun. A Review of Brain-Computer In terface Games and an Opinion Survey from Researchers, Developers and Users. Sensors , 14(8):14601–14633, 2014. [17] Aleksandra V uc k ovic, Jaime A Pineda, Kristen Lamarca, Disha Gupta, Christoph Guger, and Laura Ballerini. Interaction of BCI with the underlying neurological conditions in patien ts: pros and cons. 2014. [18] E Kinney-Lang, B Auy eung, and J Escudero. Expanding the (k aleido)scop e: exploring current literature trends for translating electro encephalography based braincomputer in terfaces for motor rehabilitation in c hildren. J. Neur al Eng. , 13(6):061002, 2016. [19] Peter J Marshall, Y air Bar-Haim, and Nathan A F ox. Developmen t of the EEG from 5 months to 4 y ears of age. Clin. Neur ophysiol. , 113(8):1199–1208, 2002. [20] Theo Gasser, Rolf V erleger, Petra B¨ acher, and Lothar Srok a. Developmen t of the EEG of school- age c hildren and adolescen ts. I. Analysis of band pow er. Ele ctr o enc ephalo gr. Clin. Neur ophysiol. , 69(2):91–99, F ebruary 1988. [21] M Matsuura, K Y amamoto, H F ukuzaw a, Y Okub o, H Uesugi, M Moriiwa, T Ko jima, and Y Shimazono. Age developmen t and sex differences of v arious EEG elemen ts in healthy c hildren and adults–quantification b y a computerized wa ve form recognition metho d. Ele ctr o enc ephalo gr Clin Neur ophysiol , 60(5):394–406, 1985. [22] Vladimir Misk ovic, Xinpei Ma, Chun-An Chou, Miaolin F an, Max Owens, Hiroki Sa y ama, and Brandon E. Gibb. Dev elopmental changes in sp ontaneous electro cortical activit y and net work organization from early to late c hildho o d. Neur oimage , 118:237–47, September 2015. [23] Andrzej Cichocki, Danilo Mandic, Lieven De Lathauw er, Guo xu Zhou, Qibin Zhao, Cesar Caiafa, and Huy Anh Phan. T ensor decomp ositions for signal pro cessing applications: F rom t wo-w ay to m ultiw a y comp onen t analysis. IEEE Signal Pr o c ess. Mag. , 32(2):145–163, 2015. [24] Evrim Acar and B ¨ ulent Y ener. Unsup ervised m ultiwa y data analysis: A literature surv ey. IEEE T r ans. Know l. Data Eng. , 21(1):6–20, 2009. [25] Anh Huy Phan and Andrzej Cic ho cki. T ensor decomp ositions for feature extraction and classification of high dimensional datasets. Nonline ar The ory Its Appl. IEICE , 1(1):37–68, 2010. [26] Andrzej Cichocki, Y oshik azu W ashizaw a, T omasz Rutk owski, Hov agim Bak ardjian, Anh-Huy Phan, Seung jin Choi, Hy ekyoung Lee, Qibin Zhao, Liqing Zhang, and Y uanqing Li. Noninv asive BCIs: Multiw a y Signal-Processing Arra y Decompositions. Computer (L ong. Be ach. Calif ). , 41(10):34– 42, Octob er 2008. [27] Y e Liu, Mingfen Li, Hao Zhang, Hang W ang, Junhua Li, Jie Jia, Yi W u, and Liqing Zhang. A tensor-based sc heme for strok e patients’ motor imagery EEG analysis in BCI-FES rehabilitation training. J. Neur osci. Metho ds , 222:238–249, 2014. [28] E. Kinney-Lang, L. Sp yrou, a. Ebied, R. Chin, and J. Escudero. Elucidating age-sp ecific patterns from bac kground electro encephalogram p ediatric datasets via P ARAF AC. In 2017 39th Annu. Int. Conf. IEEE Eng. Me d. Biol. So c. , pages 3797–3800. IEEE, July 2017. [29] Hunter M.B., Sumpter R., V erity K., Y o ong M., MClellan A., SHetty J., and Chin R.F. NEUR Odev elopmen t in PResc ho ol Children Of FIfe and Lothian Epilepsy Study: NEUR OPR OFILES - A p opulation-based study. 57:56–57, 2015. [30] Ali Hossam Sho eb. Application of mac hine learning to epileptic seizure onset detection and T ensor-driven extr action of developmental fe atur es fr om varying p ae diatric EEG datasets 18 treatmen t. pages 157–162, 2009. [31] A. L. Goldb erger, L. A. N. Amaral, L. Glass, J. M. Hausdorff, P . Ch. Iv anov, R. G. Mark, J. E. Mietus, G. B. Mo o dy , C.-K. Peng, and H. E. Stanley . PhysioBank, Ph ysioT o olkit, and Ph ysioNet : Components of a New Researc h Resource for Complex Ph ysiologic Signals. Cir culation , 101(23):e215–e220, June 2000. [32] Nicolas Langer, Erica J. Ho, Lindsa y M. Alexander, Helen Y. Xu, Renee K. Jozano vic, Simon Henin, Agustin Petroni, Samantha Cohen, Enitan T. Marcelle, Lucas C. Parra, Michael P . Milham, and Simon P . Kelly . A resource for assessing information pro cessing in the dev eloping brain using EEG and ey e trac king. Sci. Data , 4:170040, April 2017. [33] Rob ert Oostenv eld, Pascal F ries, Eric Maris, and Jan Mathijs Sc hoffelen. FieldT rip: Open source soft w are for adv anced analysis of MEG, EEG, and in v asive electroph ysiological data. Comput. Intel l. Neur osci. , 2011, 2011. [34] T amara G. Kolda and Brett W. Bader. T ensor Decomp ositions and Applications. SIAM R ev. , 51(3):455–500, 2008. [35] Richard a Harshman. F oundations of the P ARAF AC procedure: Mo dels and conditions for an explanatory m ultimo dal factor analysis. UCLA Work. Pap. Phonetics , 16(10):1– 84, 1970. [36] Nicolaas Klaas, M F ab er, Rasmus Bro, and Philip K Hopke. Recen t dev elopments in CANDECOMP / P ARAF AC algorithms : a critical review. Chemom. Intel l. L ab. Syst. , 65:119 – 137, 2003. [37] F Cong, Q H Lin, L D Kuang, X F Gong, P Astik ainen, and T Ristaniemi. T ensor decomp osition of EEG signals: A brief review. J. Neur osci. Metho ds , 248:59–69, 2015. [38] Claus A. Andersson and Rasmus Bro. The N-w a y T o olb ox for MA TLAB. Chemom. Intel l. L ab. Syst. , 52(1):1–4, 2000. [39] Rasmus Bro and Henk A. L. Kiers. A new efficient metho d for determining the num b er of comp onen ts in P ARAF A C mo dels. J. Chemom. , 17(5):274–286, June 2003. [40] Javier Escudero, Evrim Acar, Alb erto F ern´ andez, and Rasmus Bro. Multiscale entrop y analysis of resting-state magneto encephalogram with tensor factorisations in Alzheimer’s disease. Br ain R es. Bul l. , 119:136–144, Octob er 2015. [41] Mark Hall, Eib e F rank, Geoffrey Holmes, Bernhard Pfahringer, Peter Reutemann, and Ian H Witten. The WEKA data mining softw are. ACM SIGKDD Explor. , 11(1):10–18, 2009. [42] Eib e F rank, Mark A Hall, and Ian H Witten. The WEKA W orkb ench. In Mor gan Kaufmann, F ourth Ed. , pages 553–571. 2016. [43] Laurens V an Der Maaten and Geoffrey Hinton. Visualizing Data using t-SNE. J. Mach. L e arn. R es. , 9:2579–2605, 2008. [44] Stefan Haufe and Arne Ewald. A Simulation F ramework for Benchmarking EEG-Based Brain Connectivit y Estimation Metho dologies. Br ain T op o gr. , 2016. [45] R Zink, B Huny adi, SV Huffel, and M V os. T ensor-based classification of an auditory mobile BCI without a sub ject-sp ecific calibration phase. J. Neur al Eng. , 13(2):26005, 2016. Supplemen tary Information T ensor-driven extr action of developmental fe atur es fr om varying p ae diatric EEG datasets 19 MMEC Dataset # of Sub j. Age (Mon ths) 11 0-12 14 12-24 4 24-36 8 36-48 10 48-60 3 60-72 CHB-MIT Dataset # of Sub j. Age (Y ears) 1 2 2 3 1 6 2 7 2 9 2 11 2 12 1 13 1 14 1 16 1 18 1 19 CMI Dataset # of Sub j. Age (Y ears) 1 ∗ 6 11 8 12 9 13 10 7 11 ∗ Included as 8 y .o. for classification T able 3. Distribution of sub jects p er age for each dataset. T ensor-driven extr action of developmental fe atur es fr om varying p ae diatric EEG datasets 20 Figure 5. F ull and threshold reduced P ARAF AC mo del corcondia for each fold in a factorization of MMEC dataset. Only a decomp osition maintaining 70% corcondia for b oth mo dels w as b e considered viable, with the reduced tensor used for classification. F ull resolution figure a v ailable up on request. T ensor-driven extr action of developmental fe atur es fr om varying p ae diatric EEG datasets 21 Figure 6. The 3-comp onent P ARAF AC mo del decomp osition of the simulated tensor data. Raw p o w er v alues are shown for the [ S patial ] domain, while comp onent v alues in the [ S pectr al ] and [ S ubj ect ] dimension hav e been normalized. The 3-comp onent mo del accurately identifies the underlying shifting sp ectral frequencies set to v ary from approximately 5-8 Hz to 8-12 Hz based on age in the [ S ubj ect ] domain. Sharp p eaks in the [ S ubj ect ] domain reflect the simulated child with the b est signal-to-noise ratio. F ull resolution figure av ailable up on request.
Original Paper
Loading high-quality paper...
Comments & Academic Discussion
Loading comments...
Leave a Comment