Uncertainty Factors for Stage-Specific and Cumulative Results of Indirect Measurements
Evaluation of a variable Yd from certain measured variable(s) Xi(s), by making use of their system-specific-relationship (SSR), is generally referred as the indirect measurement. Naturally the SSR may stand for a simple data-translation process in a …
Authors: B. P. Datta
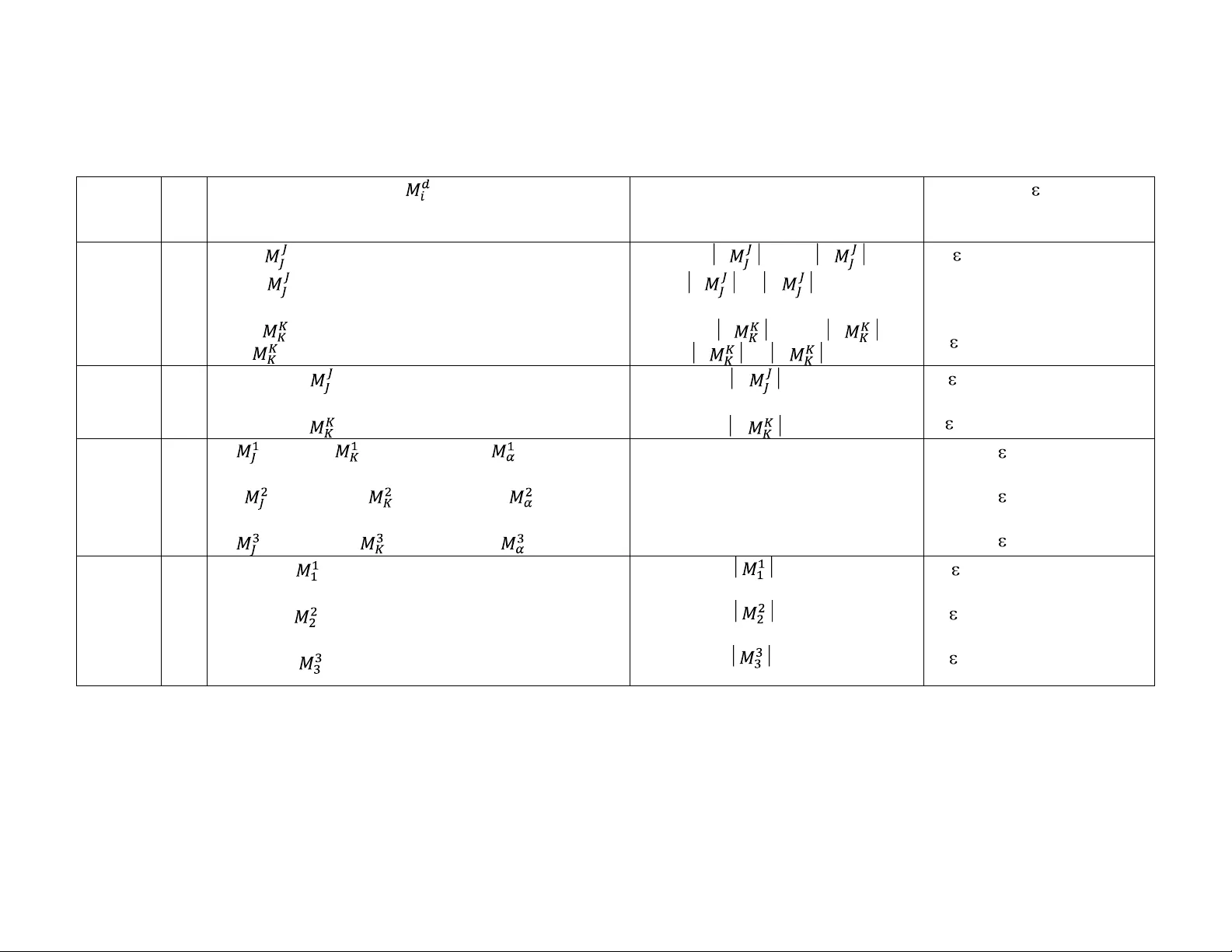
1 Uncertainty Factors for Stage-Specific and Cumulative Results of Indirect Measurements B. P. Datta Radiochemistry Laboratory, Variable Energy Cyclotron Centre, Kolkata 700 0064, India ABSTRACT Evaluation of a variable Y d from certain measured variable(s) X i (s) , by makin g use of their system-specific-relationship (SSR), is generally referred as the indirec t measurement. Naturall y, the SSR may stand for a simple data-transformation process ( X i (s) Y d ) in a given c ase, but a set of equations ( f i ({ Y d }) = X i , d, i = 1, 2, …, N ), or even a cascade of different such process es ( “ X i (s) I Y d (s) II Y d (s) … Y d (s) ”, or “ X J I Y d II Y d … Y d ” , or so), in some other case. Further , though the measurements are a p riori ensured to be accurate, there is no definite method for examining whether the result obtained at the end of an SSR, specifically a cascade of SSRs, is really representative as the measured X i -value(s) . Of course, it was recentl y shown that the uncertainty ( d ) in the estimate ( y d ) of a sp ecified Y d , viz. : X i ’s Y d , is given by a spec ified linear combination of corresponding measurement- uncertainties ( u i ’s ). Here, f urther insi ght into this principle is provided by its application to the cases represented by cascade- SSR s. It is exemplified how the different stage-wise uncertainties ( I d , II d … d ), that is to say th e requirements for the evaluation to be su ccessful, could even a priori be p redicted. The theoretical tools ( SSR s) have r esemblance with the real world measuring devices (M Ds), and hence are referred as also the data tr ansformation scales (DTSs) . However, non-uniform behavior appears to be the feature of the DTSs rather than of the MDs. 2 1. INTRODUCTION The measurement, which needs to be a ccomplish ed through a theoretical process such as the below, is generally labeled as the indirect one: 1 y d = f d ( x i ) = f d ( X i + i ) , d = i = 1 (1a) Or, y d = f d ({ x i }) = f d ({ X i + i }) , d = 1 and i = 1, 2, …, N = J, K, …, N (1b) Or, f i ({ y d }) = x i = X i + i , d, i = 1, 2, …, N (1c) where y d and x i stand for the estimates of the desired and the a ctually meas ured variables, Y d and X i , respectively, and i for the experimental e rror (the deviation in x i from its unknown true value X i ). Clearly, that Y d is known to so correlate with X i (s) is the basis for such an evaluation: Y d = f d ( X i ) , d = i = 1 (1a / ) Or, Y d = f d ({ X i }) , d = 1 and i = 1, 2, …, N = J, K, …,N (1b / ) Or, f i ({ Y d }) = X i , d, i = 1, 2, …, N (1c / ) Then any ph ysicochemical evaluation (viz. determination of a characteristic parameter 2,3 or even simply the concentration, 4-6 of a chemical species) should be, it may be pointed out, an example of the indirect measurement only. Further, as indi cated above, a given example would be distinguished from another b y generall y the nature o f the s y stem-specific-relationship (SSR ) of desired Y d (s) with the measured X i (s). However, it is possible 6 that one example differs from another onl y with respect to the desired variable Y d . Thus the evaluations represented, e.g. b y the SSRs as “ Y P = X J / 2 ” , “ Y Q = X J ”, and “ Y R = ( – 1) ” , are different for alone the theoretical tasks described b y them are different. However, such an observation makes us enquiring whether all the corresponding results ( y P , y Q , and y R ) should be equall y accurate as the mea sured estimate ( x J ). In other words, the question is : can the accuracy ( u i ), to be required in X i – measurements for achieving a preset accuracy ( d ) in the desired result ( y d ), be dictated by the SSR shaping y d ? 3 However, it ma y be reminded, the evaluation of error in an unknown-estimate (either x i , or y d ) is impossible. Again, there could be no alternative to the assessment of the possible error for ensuring whether the corresponding me asurement is useful. Thus, for a ny measurement ( X i ) to be carried out, it is a we ll-known norm to first d evelop the method to the extent that the maximum possible value (MPV) of experimental error ( i ) should at least be a cceptable. The MPV of err or is referred to here as , it should be noted, either 1 uncertainty or 7 accurac y (inaccurac y). Further, we consider the error as relative only, i.e. : i = ( X i / X i ) = ([ x i – X i ] / X i ), and then : u i = Max i . Similarly, denoting the error in the result y d as d , we me an that : d = ( Y d / Y d ) = ([ y d – Y d ] / Y d ), and : d = Max d . Now, returning to above, it may be mentioned that the point at issue ha s already been evaluated 7 . It wa s thus clarified that the shaping of a re sult y d should also mean the fixation of its uncertainty d by the SSR involved. That is to say that, as a ny oxidation reaction but without the complementary reduction process is inconceivable , the desi red (i.e. a given SSR dictated systematic ) change as “ x i (s) y d ” but without reall y the compl ementary uncertaint y(s) transformation “ u i (s) d ” is also unthinkable. In short, it has previously been explained 7 why the uncertainty d can even numerically be differe nt from the uncertainty u i , i.e. wh y the SSR can have a say in planning the required experiments. At present, the idea is simpl y to elaborate on the implications of such facts for cases of somewhat involved SSRs. Actually, the result shaping : x i (s) y d is in man y a case represented b y (instead of Eq. 1a, or Eq. 1b, or Eq. 1c, alone) a cascade of computational processes (COCP) comprising one or more types as Eqs. 1a-1c. Moreover, a stud y ma y require di fferent kinds of experiments (variables : X J , X K , … X N ), and hence the different estimates ( x J , x K , … x N ) would likel y be subject to different uncertainties ( u J , u K , … u N ). Even, different x i ’s might conform the inputs to different sta ges of a COCP (viz. : K X I I I J d d X Y Y … N X d Y ). Further, as point ed out above, 7 an y output 4 (e.g. I y d , but which should be an input to the 2 nd stage of the COCP ) will even for purel y random sources of errors in the estimates ( x i ’s ) of X i ’s b e subject to : (i) systematic-error, and (ii) to th e extent decided b y the corresponding (1 st stage) SSR. That is, assessing (predicting) uncertainty in a result obtained via a COCP appeals difficult, but is defined the objective here. In fact, an evaluation as Eq. 1 might be (for computational convenience and/ or for specific objectives) designed as a COCP. For ex ample, in the case of determinin g a light element isotopic ratio ( Y d ) by mass spectrometr y (IRMS), t he : X i (s) Y d (s) transformation is with the idea to avoid the reporting of results on non-corresponding scales 8 carried out via a C OCP. That is, as recommended, 8 ,9 the input(s) for and th e output(s) from an Eq. 1 representing the IRMS sy stem are made to be subject to certain scale-conversions. However, it is generally diffi cult to ascertain whether the scale-conversion proce sses used ther e in I RMS really assure t he purpose. Howe ver, the uncertainty-consideration here is believed to help crosscheck the pros and cons f or involving a ny data-translation process (an Eq. 1 or a COCP) in a basically experimental study. The work is organized as follows. First, we make a simplifying consideration of terminologies cum principles (section 2). Subsequently (section 3), we discuss our findings. Here, it may be mentioned that a given SSR (an Eq. 1) is sometimes for convenience referred to by alone Y d , viz. “ Y P = X J / 2 ” by “ Y P ”. 2. PRINCIPLES 2.1 Data Transformation Scale (DTS) It may be pointed ou t that, by the behavior, an SSR is indistinguishable from a measuring device (MD). For ex ample, as the response of an MD depends on the qu antit y be ing measured, Y d (or estimate : y d ) will alway s vary as a functio n of X i (or estimate : x i , cf. Eq. 1). More over, as 5 the response of an MD towards a given quanti ty is decided by it s own feature, the output ( Y d ) for any given input ( X i (s) ) here will depend on the SSR itself, e.g. X J gets projected as however three different outputs b y the above SSRs : Y P = X J /2 , Y Q = X J , and Y R = (X J 2 – 1) , which are also therefore referred to as the three different data transformation scales (DTSs). 2.1.1 Characteristics of a DTS: non-uniform behavior A DTS can like an y usual MD be shown bracketed with certain parameter(s) (s) dictatin g its behavior, 7 e.g. an Eq. 1b / could be marked with: , d = 1, and i = 1, 2, … N (or, i = J, K, … N ) (2) Similarly an Eq. 1 c / , which consists of N different S SRs (with : d, i = 1, 2 … N ), will naturall y have so many parameters ( ’s ) as N 2 . Of course, “ N ” is unity for cases r epresented b y Eq. 1a / . Yet, it should generally be possible to distinguish a given Eq. 1 a / , in terms the characteristic rate ( ) of relative variation in Y d as a function of X i , from another. For example, Y P , Y Q and Y R above are predicted to differently vary ( with X J ) : = 1.0 (cf. Y P ); = ½ (cf. Y Q ); and = . Th at is to sa y that Y P will (for a given change in X J ) vary at a rate different from that of Y Q , but the rate is in either case ever f ixed . However, the response of Y R will be decided by X J it self. For illustration, let : X J = 0.1 . Th en, one can verif y that : = - 0.0202 (cf. Eq. 2). Now, say : X J = 1.1 , which however yields : = 11.5238. Further, it may be mentioned that uniformity- in -response stands generally as a useful criterion for recognizing an MD. In addition, the results of measuring a given quantit y b y two appropriate but different kinds of MDs should by and large be expected the same. However , the behavior of even alone the DTS as Y R (viz . depends on X J ) should suffice explaining wh y non-uniformity as a feature be better attributed to the DTS than to the MDs. 6 2.1.2 Uncertainty transfer via a DTS (an Eq. 1) The signature for a relationship (Eq. 1) between X i (s) and Y d (s) is that the substitution for X i (s) by an y desired True x i (s) should y ield the corresponding True y d (s) , and the vice versa. However, this had led to the qu ery 7 wh ether x i (s) having subject to say 0.05% uncertainty will cause the desired y d (s) to be at e xactly 0.05% uncertaint y (i. e. should d equ al u i ?) , which w as in turn evaluated by introducing the above term “ behavior of a DTS ( , cf. Eq. 2)”. Thus , it was clarified that: 7 ε d = (3) Or, if all inputs ( x i ’s ) are subject to a given uncertainty ( G u ), i.e. if u i = G u ( i = J, K, … N ), then: d = ( ) u i = ( ) G u (4) Here, it ma y also be pointed out that Eq. 2 defines (s) as the theoretical constant(s) for a given DTS, thereb y enabling even a priori predicti on of the uncertaint y ( d ). Further, for a simple case as Eq. 1a / ( d = i = N = 1), Eq. 3/ 4 reduces to : d = u i = G u (3a) It ma y be noted th at neither the evaluation of the uncertaint y d requir es the knowledge of the u i -sources, nor can d var y depending on whether u i stands for random and/ or s ystematic causes . Further, Eq. 3 should stand exact for all cases r epresented b y linear S SRs onl y. 7 Howev er, as th e factors o f “ (u i ) P , with : P 2 ” or so are i gnored 7 in Eq. 3, the uncerta int y ( d ) corresponding to a non-linear DTS (and for finite u i (s) ) might in reality var y from t hat given by Eq. 3. Nevertheless, the ex periments are always so designed that : u i zero. Therefore, Eq. 3 should also suffice explaining the non-linear cases. In support, we offer the evaluation below. Let the test-systems to be represented by the SSRs Y P , Y Q and Y R above, and an X J -standard by “ T X J = True x J = 0.1 (i.e. say : Y P = 0.05 ; Y Q = 0.3162278; and : Y R = -0.99 )” . Further, let the method of measurement be so established that : u J = 0.05% (i.e. sa y the estimates of T X J were observed to 7 be so restricted as : 0.100 05 T x J 0.09995). Then, on the one, Eq. 3a predicts : P = u J = u J = 0.05% ( as : = 1, cf. section 2.1.1); Q = u J = u J /2 = 0.025% ; and R = u J = 0.0202 u J = 0.00101% . On the other, taking e. g. the lowest estimate : T x J = 0.09995, one obtains : y P = 0.049975, y Q = 0.316149, and : y R = -0.99001. Thus, it may be noted that not alone “ P = Max P = ( y P - Y P )/ Y P = 0.05% = P ” ; but “ Max Q = 0.025003% Q ” ; or “ Max R = 0.00101% = R ”. Similarly, let the m easurement-technique be so independent of X J that, against “ T X J = 1.1 ”, it yields : 1.10055 T x J 1.09945 (i.e. again suppose that : u J = 0.05%). Then, taking e.g. th e highest estimate ( T x J = 1.10055), one obtains : y P = 0.550275 (i.e. : Max P = 0.05% = P ); y Q = 1.049071 (i.e. : Max Q = 0.024997% Q ); and : y R = 0.21121 (i.e. : Max R = 0.5763% R ), which further verify the theory that (though : P = u J , or : Q = u J / 2 ) , and hence R , will vary with X J ( R = 11.5238 u J = 0.5762% , cf. Eq. 3a). 2.2 u i (s) d via a COCP: stage-wise uncertainty factors (UFs) Suppose that the parameter III Y d of an arbitrary sy stem is determined via a COCP as: I Y d = f d ( X J , X K ) = X J – X K (5) II Y d = f d ( I Y d ) = α ( I Y d + 1) (5a) III Y d = f d ( II Y d , X L ) = II Y d + X L (5b) where the prefix as “I” refers to the stage -number , and α is a constant . Clearly, the desired variable ( III Y d ) could be obtained as a single -stage-process. How ever, the id ea is to also represent the cases, where : (i) not alone the COCP output (here, III Y d ) but even an inter-stage on e ( I Y d or II Y d ) is a desired variable, 2,3 (ii) a sin gle-step proce ss is, say simpl y for conv enience, spli t up int o more than one, 10,11 etc. An yway, the basic purpose here is to examine whether the inter -stage 8 theoretical-processes can cause their output -parameters as I d , II d , and III d , to numerically differ from one another, and/ or whether should it be a fact that : u i < I d < II d < III d , or so. The 1 st stage process, it may be point ed out, can in no wa y b e different from an individual DTS, and hence the uncertainty ( I d ) in the result ( I y d ) should be obtained as (cf. Eq. 3): I d = I u J + I u K = ( I F J + I F K ) G u = I [UF ] d G u (6 ) where : I and I stand for the 1 st stage SSR -specific parameters (cf. Eq. 2), u i ‘s for the measurement-uncertainties, G u for any preset value (e.g. 0.01%) of “ u i ” so that : F J = ( u J / G u ), F K = ( u K / G u ), and/ or the 1 st stage uncertainty factor ( I [UF ] d ) is a priori predicted a s: I [UF ] d = K i = J I F i = I F J + I F K (7 ) The 2 nd stage has only one input ( I y d ) but is generated by the 1 st stage. Otherwise, the 2 nd -stage (i.e. the 1 st cumulative) uncertainty II d should also be obtained as Eq. 3/ 3a: II d = II I d = II I [UF ] d G u = II [UF ] d G u (6 a) where : II is the characteristic theoretical constant of the 2 nd stage SSR, and II [UF ] d is called as the 1 st cumulative (or, 2 nd stage) uncertainty-factor and is also a theoretical parameter: II [UF ] d = II I [UF ] d = II ( K i = J I F i ) (7 a) The 3 rd stage (Eq. 5b ) is analogous to the 1 st stage, but its inputs comprise the int er-stage- dependent variable II Y d , and an independent one ( X L ). Hence, the output -uncertaint y ( III d ) should here again be obtained as a linear combination of input uncertainties ( II d and u L ): III d = III II d + III u L = III II [UF ] d G u + III F L G u = III [UF ] d G u (6 b) where : III and III stand for the predicted rates of variations (c f. Eq. 2) of III Y d as a function of II Y d and X L , respectively , F L = ( u L / G u ), and III [UF] d is the 2 nd cumulative unce rtainty-factor: 9 III [UF ] d = III II [UF ] d + III F L (7 b) Thus, as shown, Eq. 3 stands as the fundamental expression of uncertainty ( d ) in the result y d . It ma y however b e mentioned that, only for indicating the pro cess involve d is a COCP, the final output is also prefixed here (cf. III Y d ). Further, it ma y be noted that d c an, depending upon the value(s) of the SSR-specific constant(s) as (s) an d/ or the COCP-parameters as [UF] d , turn out > G u (or, > u i ), equal to G u , or even < G u ,. Now, let us im agine that : u J = u K = u L = G u , so that the factors as F J , F K and F L be all unit y, and/ or that the unc ertaint y-factors ( UFs ) be dec ided b y the stage-related ’s only(cf. Eq. 7c below ). Clearly, even then, no trend as either : III d = II d = I d = G u , or : G u < I d < II d < III d , or so, should stand for a general fact. III [UF ] d = III II [UF ] d + III F L = III II I [UF] d + III F L = III II ( I F J + I F K ) + III F L = III II ( I + I ) + III (7 c) 3. COCP SYSTEMS: VERIFICATION OF THE UNCERTAINTY THEORY 3.1 The arbitrary COCP system as Eqs. 5-5b Whether our treatment above is cor rect or not c an be ascertained provid ed the s ystem-specific X i -values , and hence the I Y d , II Y d , etc., are known. Therefore, inst ead of a real world s y stem (with unknown X i ’s ), we first consider the C OCP system as Eqs. 5-5b to be r epresented b y, say : α = 0.25; and by the X J , X K and X L -standards as : T X J = 5.0, T X K = 3.0 and T X L = 0.50, respectively. We also presume that all measurement s were desired to be 0.05% accurate (i.e. G u = 0.05%). However sa y, while the measurements of X J and X K had actuall y ensured : u J = u K = 0.05% (i.e. tough the repetitive measurements of T X J and T X K had yielded : 4.9975 T x J 5.0025, and : 2.9985 T x K 3.0015, respectivel y), the X L -measurement was tricky as that : u L = 0.4% (i.e. : 10 0.5002 T x L 0.498). Then, it should be noted that : F L = ( u L / G u ) = 8, but : F J = F K = 1. However the stage-specific parameters ( (s) , [UF ] d , and d ) are presented in T able 1 (cf. Block No. 1 ) , which clarifies that ’ s (e.g . I , and I ), and hence [UF] d ’s and/ or d ’s , depend on X i ’s . That is to say that, if X i ’s vary fr om their chosen true values ( T X i ’s ) above, then I d , II d and III d would also vary from their values predicted in Table 1. However, how should we verify, e.g. that the 1 st stage output-uncertaint y ( I d ) is in reality 0.2%? Clearly, the same is true, provided 7 : (i) only two specific combinations of the (highest and the lowest) estimates of T X J and T X K above should cause the r esult ( I y d ) to be at ±0.2% error ; a nd (ii) all other ( T x J and T x K ) combinations should imply “ I d < 0.2% ”. Nevertheless, it could be shown that : (1) ( x J = T x J = 5.0025 and x K = T x K = 2.9985) give “ I y d = 2.004, i.e. : I d = 0.2% ” ; and (2) ( x J = 4.9975 and x K = 3.0015) yield “ I y d = 1.996, i.e. : I d = -0.2% ”; but even (3) ( x J = 5.0025 and x K = 3.0015) g ive an output equall y accurate as them ( I y d = 2.001, i.e. : I d = 0.05% = i ) . Further : (4) x J = 5.001 (with : J = 0.02%) and x K = 3.001 (w ith : K = 0.0333%), y ield “ I y d = 2.0 = I Y A ” . Th is , it may be noted, clarifies the fact 7 that “ i ’s 0.0 ” ca n cause “ d = 0.0 ” . Actuall y, the corresponding predic ted requirement 7 is : J / K = ( I / I ) = 0.6 (cf. Table 1), which is satisfied in the present case ( as : J / K = 0.6). Ov er and abo ve, the uncertaint y factor has also the value as predicted : I [UF] d = ( I d / G u ) = (0.2 / 0.05) = 4.0 (cf. Table 1). Now, coming to the 2 nd stage process (Eq. 5a), it could be readily seen that : (i) I y d = 2.004 (cf. example no. 1 above) yields “ II y d = 0.751 (i.e. : II d = 0.133%) ” ; (ii) I y d = 1.996 gives “ II y d = 0.749 (with : II d = - 0.133%) ” ; but (iii) the example nos. 3 and 4 give “ II y d = 0.75025 ” , and “ II y d = 0.75 ” , respectively (i.e. II d < 0.133% ). These verif y that : II d = 0.133%, and/ or : II [UF ] d = II d / G u = 11 2.667 (cf. Table 1). Even this prediction ( II d = 0.1 33% ) could be, as indicated above, shown tru e for the stages I and II together to be the single DTS : II Y d = f II ( X J , X K ) = α ( X J - X K + 1). Similarly it can be su pplement ed that the output III y d will turn out more inaccurate than either the 1 st ( I y d ) or the 2 nd ( II y d ) stage-output ( III d = 0.24% , cf. Table1 ), viz. only the two pairs of input-estimates as ( II y d = 0.751 and x L = 0.502 ) and ( II y d = 0.749 and x L = 0.498 ) y ield : III y d = ( III Y d 0.0024 III Y d ). However, all other possible pairs of II y d and x L imply : III d < 0.24% (e.g. II y d = 0.75025 and x L = 0 .502 , gives : III y d = 1.252 25 i.e. III d = 0.18%). Here again, the facts ( III d = 0.24%, and/ or : III [UF] d = III d / G u = 4.8) could b e shown to re main un changed for considering : III Y d = f III ( X J , X K , X L ) = (α ( X J - X K + 1) + X L ). However, if : F J = F K = F L = 1 (i.e. if also : u L = G u ), then Eq. 7c predicts : III [UF ] d = 2.0 , which in turn gives : III d = 2 G u = 0.1%. That is to say that the estimate III y d would then be more accurate than either the estimate I y d or II y d (as, it may be pointed out, Eq. 5 or Eq. 5a does not involve X L , i.e. as F L can not aff ect I d ( 0.2% ) and II d ( 0.133% )). This further clar ifies the basic fact that any output uncertainty d (and/ or [UF] ) would jointl y be decided by the corresponding SSR and its input-uncertainties ( u i (s) and/ or d (s), i.e. as the case may be ). I n addition, the finding such as that neither : I d = u i ( = G u = 0.05%), nor : I d = II d = III d , is a confirmation that even a linear DTS may cause its output to vary by accuracy from its input . 3.2 Standard free energy ( Y G ) of micellization The free energy of mecellization Y G was evaluated 2 via the critical concentration ( Y C ) as: Y C = f C ( X J , X K , X L ) = X J / ( X K – X L ) (8) and, Y G = f G ( Y C ) = R T ln ( V 0 Y C / [1 + V 0 Y C ]) (8 a) where X i ’s stand for rel evant experimental v ariables, R for the gas constant, T for a bsolute temperature, and V 0 is a constant of reaction medium. 12 However we enquire whe ther Y G , which wa s obtained as a func tion of Y C alone, is e xactly as accurate as the estimate ( y C ) of Y C . We therefore work out the parameters of the COCP -sy stem here, and present them in Table 1 (cf. BLOC K No. 2 ). The t able clarifies that t he behavior (i.e. : (s )) of eithe r the 1 st or the 2 nd stage process would depend on X i ’ s but which are unknown. However, for the speci fic experimental condition s referred 2 to as “R w = [water]/[ surfactant] = 1” in chloroform ( V 0 = 0802 dm 3 mol -1 ), the X i -values obtained : x J = 0.22 0.04, x K = 18.4 0.1 (in dm 3 mol -1 ) and x L = 10. 6 0.6 (in dm 3 mol -1 ). These give : y C = 28.20 (reported 2 : 28.3) mmol dm -3 , which in turn yields : y G = -15.1 kJ mol -1 . It should be noted that such a set of the measured estimates ( x i ’s ) are take n here as the true x i ’s (i.e. : X i = x i , and hence : Y d = y d ), and the ir scatters as the measurement-uncertainties (i.e. : u J = [ (0.04 / x J ) = (0.04 / 0.22)] = 18.2%, u K = 0.54%, and u L = 5.7%), and accordin gly the behavior of th e s ystem is elabora ted in T able 1. Further, as u i ’s are known, the consideration and non-consideration of the parameters as F i ’s (cf. section 2.2) should make no difference. Yet, in Table 1 (Block no. 2), the uncertaint y-evaluation is illustrated for the preset measurement-uncertaint y ( G u ) of 0.5% (i.e. G u u K ), and hence for : F J = ( u J / G u ) = 36.4, F K = 1.08, and F L = 11.4. Table 1 predicts the estimates of Y C and Y G to be subject to the uncertainties as high as 50 and 9 times the least experimental-uncertaint y u K , respectively. Thus, for example, it could be shown that “ x J = ( X J + 18 .2%), x K = ( X K - 0.54%), and x L = ( X L + 5. 7 %)” yield “ y C = ( Y C + 30 %), i.e. │ Max C │ = 30%, and hence [UF] C = ( Max C / G u ) = 60”; which in turn yields : y G = ( Y G – 4.28%), i.e. [UF ] G = (4.28 / 0.5) = 8.56 . Clearly, the reason is that the u i ’s are generall y high. Nevertheless, that the predictions (Table 1) are s ound could be better v erified by s caling down the errors by sa y a fact or of 100 (i.e. say : G u = 0.005%, so that : C = 0.272% a nd : G = 0.0445% ), viz. : (1) x J = ( X J + 0.182 %), x K = ( X K - 0.0054%), and x L = ( X L + 0.057 %), yields “ y C 13 = ( Y C + 0.27% ), i.e. [UF] C = (0.27 / G u ) = 54 ”, which in turn gives “ y G = ( Y G – 0.045% ), i.e. [UF ] G = 9 ” ; and (2) x J = ( X J - 0.182%), x K = ( X K + 0.0054%), and x L = ( X L - 0.057%), impl y “ y C = ( Y C - 0.27% ), i.e. [UF] C = 54” , and “ y G = ( Y G + 0.045% ), with [UF ] G = 9”. Fu rther, all other error-combinations with “│ J │ ≤ 0.182%, │ K │ ≤ 0.0054%, and │ L │ ≤ 0.057%” could be shown to cause : │δ C │ < 0.27% and, │δ G │ < 0.045% , e.g. (3) x J = ( X J + 0.182%), x K = ( X K - 0.0054%), and x L = ( X L - 0.057%), yield “ y C = ( Y C + 0.1 2%), a nd “ y G = ( Y G - 0.02 %) . However, in order for ha ving a better picture as to how u i ’s affect the desired results, we now consider all the th ree e stimates ( x i ’s ) above as equ ally accurate ( u J = u K = u L = G u = 0.5%), i.e. : F J = F K = F L = 1. Then, one can v erify : [UF] C = L i = J = 4.72, a nd : [UF] G = [UF ] C = 0.77 (cf. Eq. 7c). That is, the estimate y C would even th en be more in accurate ( C = 4.72 G u = 2.36 %) , but y G be b etter accurate ( G = 0.77 G u = 0.39%), than the measu red estimates ( x i ’s ). For example : x J = ( X J - 0.5%), x K = ( X K + 0.5%), and x L = ( X L - 0.5%), yield “ y C = ( Y C – 2.32%)”, which in turn g ives “ y G = ( Y G + 0.38%) ” . Howev er, x J = ( X J + 0.5%), x K = ( X K + 0.5%), and x L = ( X L + 0.5%), yield : y C = Y C , and hence : y G = Y G , which thus confirm the finding 7 that an y DTS of the type as Eq. 1b / (here ) has got the possibility of leading “ i ’s 0” to “ d = 0”. 3.3 Rate constants ( Y F , Y D and X J ) for an enzyme ( E) catalyzed reactions of myoglobin (S) E + S F D Y Y ES J X E + P The reaction rate constant ( X J ), and some other parameters ( X K , X L , a nd X M ), were obtained 3 by experiments-cum-curve-fitting. However, the formation constant ( Y F ) and the dissociation constant ( Y D ) of the ES complex were evaluate d 3 as: 0 ( , , ) KL F F J K L JE XX Y f X X X XC (9 ) 0 ( , , , ) D D J K L F K L J F E Y f X X X Y X X X Y C (9 a) 14 where 0 E C is a constant (i nitial concentration of enz y me). Further, the res ults of all kinetic parameters were reported 3 to contain 10% of experimental errors. Therefore, if the uncertainty ( u J ) in the determined v alue ( x J ) of X J sh ould be 10%, then the un certainties ( F and D ) in the estimates ( y F and y D ) of Y F and Y D (respectively) should also be 10%. At least, it appears that D , F and u J , were meant 3 to be equal. However, the idea he re is to ascertain the fact. It ma y here be mentioned that, though “ X K ” and “ X L ” (cf. Eq. 9 -9a) were implied 3 to have obtained experimentally, neither the ir estimates ( x K and x L ) nor the corresponding un certainties ( u K and u L , respectivel y) were re ported. Y et, it may be noted , we first make our consideration (cf. Block No. 3 in Table 1 for the DTS-specific predicted para meters) for : (i) not alone “ X J ” but X K and X L as the measured variables, and (ii) u J = u K = u L = G u (i.e. fo r : F J = F K = F L = 1). W e, of course, also clarify below the implications for both X K and X L to be the constants. The present system (Eqs. 9-9a) could, if the n umber N of e xperimental variables ( X i ’s) is any yardstick, be conside red analogous to the COCP case ( Eq s. 8-8a) above. However it should be noted that the features of the 1 st stage process as Eq. 9 are predicted to be, unlike those for Eq. 8, independent of X i ’s (i.e. = 1 , with : i = J, K and L, cf. Table 1 ). Further , that thi s is the fact can be verified as follows. The actual error ( F ) in the result ( y F ) should be obtained as 7 : = ( - J + K + L ), i.e. if and when : J = ( K + L ), the result should be absolutely accurate. In fact, it can be seen that a data-set of the type “ x J = ( X J + 0.05%), x K = ( X K + 0.02%), and x L = ( X L + 0.03%) ” b ut irrespective of whatever might be the true valu es of the X i ’s yield : y F = Y F (i.e. error : F = 0). These, in addition to confirming that a ny DTS as Eq. 1b / can under certain circumstances cause : ( i ’s 0) ( d = 0), exemplify the f act 7 tha t the characteristics of a DTS can (even drastically) vary with its description. Now, we come to the 2 nd stage process (Eq. 9 a) but which reduces as : 15 Y D = X K + X L X J – ( X K X L / X J ) = f D ( X J , X K , X L ) f D ( Y F ) (9a / ) Eq. 9a / cla rifies th at the dissociation constant ( Y D ) is governed thou gh differentl y by the same set of experimental variables ( X J , X K , and X L ) which define the formation constant ( Y F ). That is, as : F = f F ( u J , u K , u L ), the uncertainty D c an also be independently evaluated as : D = f D ( u J , u K , u L ). It ma y in fact be pointed out that, in Table 1 (Block no. 3), the 2 nd stage par ameters are furnished against Eq. 9a / , and the r eason why is that Y D though which mi ght be com puted via Eq. 9a is not really a function of Y F . That is to say that, in principle and unli ke “ Y G ” above (cf. Eq. 8a : Y G = f G ( Y C ) = f G ( X J , X K , X L )) , neither Y D should be referred as the 2 nd stage variable, nor the unc ertainty ( D ) in its estimate ( y D ) c ould be obtained as an inter-stage dependent (i.e. COCP) parameter. However, unlike the case of Y F , the rates ( ’ s , cf. Eq. 2) of variations of Y D with X i ’s are predicted to depend on X i ’s (cf. Table 1 ). Thus, it ma y be noted, the tabulated -values w ere obtained b y considering the true X i ’s to be : X J = 1.1 s -1 (i.e. as reported 3 ), X K = 1.3 , and X L = 2.7077x10 -8 (i.e. as obtained here b y solving Eq. 9 and Eq. 9a / for 3 : 0 E C = 3.2 x10 -6 M, Y F = 0.01 M -1 s -1 , and Y D = 0.20 s -1 ). However, though Y D is prac tically inv ariant with X L ( = -2.5x10 -8 , cf. Table 1), the uncertaint y in its estimate is predicted to be 4 times and 12 times higher than the uncertainties in the estimates of the forward ( Y F ) and reaction rate ( X J ) constants, respectively ( [UF ] D : [UF] F : u J = 12 : 3 : 1, cf. Table 1). Thus, for verification, let the evaluation process be so established that : u i = G u = 1%. However, thou gh true errors ca n never be known, imagine that two different sets of expe riments have yielded : ( 1 ) x J = ( X J 0.01 X J ), x K = ( X K + 0.01 X K ), and x L = ( X L + 0.01 X L ); and ( 2 ) x J = ( X J + 0.01 X J ), x K = ( X K 0.01 X K ), and x L = ( X L 0.01 X L ). Then, it could be shown that the set no. 1 gives : y F = ( Y F + 0.0304 Y F ), and : y D = ( Y D + 0.12 Y D ); and the set no. 2 yields : y F = ( Y F 0.0296 Y F ), and : y D = ( Y D 0.12 Y D ). Further, one can verif y that all other possible sets of x i ’s (with : Max i = 1% ) impl y : F < 3% , and : D < 12% . 16 Above, we have considered : u J = u K = u L = G u . However, if either or both u K and u L differ from u J , then the uncertainty factor will also be different . For ex ample, let : F J = 1 , but : F K < 1 and/ or : F L < 1 . Then, Eq. 7 predicts : 1 < [UF] F < 3 , and : 5.5 < [UF] D < 12 . S imilarly, say “ X K ” and “ X L ” are constants , i.e. : Y F = f F ( X J ); and : Y D = f D ( X J ). T hen the d etermined 3 X J and Y F should certainly be equall y accurate ( F = u J = u J , c f. Eq. 3a). Yet, as : 1, D can not be taken to equal u J , i.e. “ y D ” should be (for the X i values above, 5.5 times more) inaccurate than “ x J ”. The behavior of the SSR Y F (Eq. 9), it may here again be emphasized, is independent of X i ’s . 3.4 Simultaneous determination of carbon and oxygen isotopic abundance r atios as The constituent 13 C/ 12 C, 17 O/ 16 O and 18 O/ 16 O abundance ratios (say, III Y 1 , III Y 2 and III Y 3 , respectively), or even only 13 C/ 12 C and 18 O/ 16 O ratios, in CO 2 are generally det ermined via the (inputs II Y d ’s , and the outputs Y d ’s , shaping) COCP as below: 10,11 I Y d = f d ( S X i , A X i ) = ( S X i + 1) ( A X i + 1) ( D K i +1) – 1, d = i = 1, 2 = J, K (10 ) II Y d = f d ( I Y i ) = D R i ( I Y i + 1), d = i = 1, 2 = J, K (11) f i ({ III Y d }) = II Y i , (with : d = 1-3 , but : i = 1, 2 = J, K) (12) Y d = f d ( III Y d ) = ( III Y d / D E d ) – 1, d = 1, 3 (or, d = 1, 2, 3) (13) where the prefix es S, A and D refer to the s ample CO 2 , an auxil iary-reference CO 2 and a desired- reference CO 2 , respectivel y; and D K i , D R i , and D E d , are known constants. Clearly, either Eq. 10 or Eq. 11 or even Eq. 13 represents 2-3 independe nt relationships. However, Eq. 12 is generally referred 10 - 12 to the following set of relationships: III Y 1 + 2 III Y 2 = II Y J (12 a) III Y 2 (2 III Y 1 + III Y 2 ) + 2 III Y 3 = II Y K (12b) III Y 2 = [ D E 2 / ( D E 3 ) ] ( III Y 3 ) = ( III Y 3 ) (12 c) 17 where D E 1 (cf. Eq. 13), D E 2 and D E 3 stand for the known (attributed) values of 13 C/ 12 C, 17 O/ 16 O and 18 O/ 16 O abundance ratios, respec tivel y, in the CO 2 gas D; and thus are chosen constants (e.g. : = 0.5, 10 - 12 , = 0 .528, 13 etc.), so that the solutions for three unknowns ( III Y d ’s ) but from (only two di fferent data on the sample isotopic- molecular abundance ratios II Y J and II Y K , and hence from) a set of two equations (nos. 12 a-12b) should be possi ble. Here , it may also be mentioned that the X i -measurement is generally carried out 10,11 as the relative difference between the isotopic molecular abundanc e ratio ( S R i , or A R i ) of the CO 2 gas (S, or A) of interest and that ( W R i ) of a working-reference CO 2 gas (W). Thus, here, we refer (cf. Eq. 10) : 10 S X J = [( S R J / W R J ) – 1] = [( S R 45/44 / W R 45/44 ) – 1] ; and A X J = [( W R 45/44 / A R 45/44 ) – 1]; S X K = [( S R 46/44 / W R 46/44 ) – 1]; and A X K = [( W R 46/44 / A R 46/44 ) – 1]). Similarly : D K i = [( A R i / D R i ) – 1]. Further, even for given D E 2 and D E 3 (cf. Eq. 12c), will va ry with the sample (i.e . III Y 2 and III Y 3 ). That is to say that there can be no wa y to exactly preset “ ” . In other words, not alone the measured estimates ( S x J , A x J , S x K , and A x K ) should be at certain uncertainties ( S u J , A u J , S u K , and A u K , respectively ) but even the chosen value of would be subject to some uncertainty u . We th erefore tr eat as a measured cum input variable ( i = L ), thereby offering a means to study the effec ts of its variation on the desired results. The COCP-parameters ( ’s , [UF] d ’s, and d ’s ) are furnished in T able 2. The case of the somewhat involved p rocess as Eq. 12 is eve n ela borated in APPENDI X 1 . How ever, as clarified (cf. T able 2 and Append ix 1), the characteristics of all the SSRs (Eqs. 10- 13) will var y with the measured variables and the constants as well. Thu s, it may be noted that the numerical values of ’s in Table 2 are r eferred to 10 : S X J = -0.020, A X J = -0.0070, D K J = 1.756367272x10 -3 , and S X K = 0.020, A X K = -0.0010, D K K = -2.193768974x10 -3 ( cf. 1 st stage) , D R J = 11.99880669x10 -3 , and D R K = 41.85401492x 10 -4 ( cf. 2 nd stage) , and D E 1 = 11.2372x10 -3 , D E 2 = 38.0803342x 10 -5 , D E 3 = 18 20.88349077x10 -4 , and = 0.5 ( cf. 3 rd and 4 th stages) . In addition, the [UF] d , and hence the d , values are predicted for : S u J = A u J = S u K = A u K = u = G u , i.e. for : F i (s) = 1.0 . Therefore, “ G u ” in Table 2 could be read as “ u i (i = J, K, or α)” , e.g. : I J = 1.06 u i . Further, if : G u = 1%, then, for a specified Y d , “ d ” will numerically equal to “ [UF] d ”. Such a fact is exemplified in Table 3, which furnishes, for : x i = ( X i ± 0.01 X i ), all the different stage-outputs ( y d ’s ) and their actual er rors ( d ’s) . It ma y be noted that onl y t wo specific examples (nos. 1 and 2 ) yield, li ke the cases discussed above : d = d = [UF] d , but corresponding other s (nos. 3-5 ) give : d d . What ma y however be worth pointing out is that , even for a given input-uncertainty ( u i = G u , with : i = J , K and, ), d III d II d I d u i (cf. Table 2 ), which supplement the previous finding 7 that no DTS (Eq. 1 or COCP) can be wit hout checking its properties taken to yield the desired output(s) exactl y so accurate as its input(s). In other words, the study here cla rifies that the comparison b etween labs on the measured d ata ( X i -values ), and that on the corresponding results (scale-converted data : Y d -values ), ma y not stand the same. For illustration, suppose that two different labs : (i) are equipped for measurements with 1% accura cy ( G u = u i = 1%), and (ii) have report ed the measured data, and h ence the result s, on their independentl y collected samples, as those described by the example nos. 1 and 2, respectively , in Table 3. Then, it ma y be noted that the measured data, e. g. S x J = ( S X J + 0.01 X J ) = -0.0202 and, S x J = ( S X J 0.01 X J ) = -0.0198 (cf. example nos. 1 and 2, respectively), imply a variation of 2% be tween labs. However, it ma y b e pointed out, the lab to la b output-variation d epends on the stage no. and the output- variable as well, e.g. the COCP results as y 1 differ b y 2.18% , but y 2 -values b y 4.5% and the y 3 -values b y 2.5 %. On the contrary, the varia tion between the outputs for a specified 2 nd or 3 rd stag e varia ble appeals relatively insignificant. For example, the III y 2 values var y from on e another b y the l east extent ( 0.038% ), even though the desired y 2 -values are most varied ( 4.5% ), among all. 19 Now, it ma y also b e n oted, the example no. 6 (with : α = 0.53) in Table 3 is different from th e example no. 0 (with : α = 0.50) only for α . However, the variation is significant ( α = 6%). Yet, it may be pointed out, the corresponding estimates (i.e. III y d ’s ) are at relatively insi gnificant errors. This verifies th e pr ediction in Table 2 (cf. the III -values) and/ or the previous report 14 that III Y d ’s are much less sensitive towards the va riation in than that in the measured data. 4. CONCLUSIONS It is above shown ho w, in the case of an indirect measurement system represented specificall y by a COCP (viz. : X J I Y d II Y d … Y d ), the uncertainty ( d ) in a ny stage-specific or COCP estimate ( y d ), and hence the requirements for y d to be as representative as desired, can a priori be predicted. Basicall y, the study onl y helps generalize a previous finding 7 that the transformation of the measure d estimate (s) x i (s) into the desired estimate(s) y d (s) ( using the given (sin gle set of, or cascade of ) s ystem-specific relationship(s) of the measured X i (s) with the desired Y d (s) ) can cause the y d (s) to worse or even better represent the evaluation -system than the x i (s) . It is thus exemplified above that, while the 1 st stage result ( I y d ) could depending u pon the nature of the corresponding SSR be more inaccurate than the measured data ( I ε d > u i ), the 2 nd stage result might turn out more accurate ( II ε d < u i ). In other words, it is for alone the characteristic behavior(s) of the SSR(s) and/ or C OCP representing the evaluation-s ystem shown to be possible that the measured data and the corresponding derived (scale-converted) output(s) lead to quite diverging conclusions about the s ystem/ sample studied. The a priori uncertaint y ( d ) evaluation shown above can however help to avoid such a possible confusion, and even properl y preplan the required experiments, thereby arriving at the truth sought. 20 Further, the theoretical tool as an SSR is clarifie d to behave like a ph y si cal mea suring devi ce (MD), and hence is also referred to above as a da ta transformation scale (DTS). However, while a given evaluation refers a given (set of) DTS(s), there could always be more than one MD enabling the r equired X i -measurement(s). However the one, ensurin g the achievable accuracy to be not alone hi gh (i.e. of fering u i to be close to zero ) but unvarying as a function of X i , should generally be the preferre d technique. That is to sa y that the uniformit y in behavior should be a desired characteristic for an MD to be chosen in practice. However, even for given measurement- accuracy(s) u i (s) , it clari fied above that accuracy d can for one or the other given case of DTS considerably var y with alone the X i -value( s) . Non-uniformity in behavior is thus a ttributed to be the characteristic for the DTS in general, thereb y emphasizing the need fo r a priori est ablishment of not simply the me thod-specific u i but the nature of the DTS (i.e. -values, and hence d ). REFERENCES 1. ISO, Guide to the Expression of Uncertainty in Measurem ent (1995). 2. Nakashima, T.; Fujiwara T.; Kumamaru, T. Bull. Chem. Soc. Jpn. , 2002 , 75, 749. 3. Furusawa, H.; Takano, H.; Okahato, Y. Anal. Chem. , 2008 , 80, 1005. 4. Koskela, H; Rapinoja, M-L; Kuitunen, M-L; Vanninen, P. Anal. Chem. , 2007 , 79, 9098. 5. Yip, Y-C; Chu, H -S; Chan, K-C; Chan, K-K; Cheung, P-Y; Sham, W -C. Anal. Bioanal. Chem. 2006 , 386, 1475. 6. Da tta, B. P.; Kho dad e, P. S.; Par ab, A. R .; G o y al, A. H .; C hita mbar, S. A .; Ja in, H.C . In t. J . Ma ss Spec tro m. Io n Procs ses 1992 , 116, 87 ; 1995, 142, 69 ; Rapid Commun. Mass Spectrom. , 1993 , 7, 581. 7. Datta, B. P. arXiv: 0712 .1732v1 [phy sics.data-an]. 21 8. Coplen, T. P. IAEA-TECDOC-825, IAEA-TECDOC-825, IAEA, Vienna, 1995 , p. 31. 9. Coplen, T. P.; Brand, W. A.; Gehre, M.; Gr oning, M. Meijer, H. A; Toman, B.; Verkouteren, R. M. Anal. Chem. 2006 , 78, 2439. 10. Allison, C. E.; Franc y, R. J .; Meijer, H. A. J . IAEA -TECDOC-825, IAEA, Vienna, 1995 , p. 155. 11. Verkouteren, R. M.; Lee J. N. Fresenius J. Anal. Chem. 2001 , 370, 803. 12. Craig, H. Geochim. Cosmochim. Acta 1957, 12, 133. 13. Assonov, S. S.; Brenninkmeijer, C . A. M. Rapid Commun. Mass Spectrom. , 2003 , 17, 1007. 14. B. P. Datta, Int. J. Mass Spectrom. , 2004, 237, 135. 22 APPENDIX 1: III ’s and III [UF] d ’s corresponding to the set of DTS as Eqs. 12a-12c Eqs. 12a-12c can in differential form be expressed a s Eqs. A.1-A.3, respectivel y : 14 C J 1 III Y 1 + C J 2 III Y 2 = C J II Y J (A.1) C K 1 III Y 1 + C K 2 III Y 2 + C K 3 III Y 3 = C K II Y K (A.2) C α 2 III Y 2 + C α 3 III Y 3 = C α (A.3) where : C J1 = 1, C J2 = 2, and C J = 1 (cf. Eq.12a ); C K1 = 2 III Y 2 , C K2 = 2( III Y 1 + III Y 2 ), C K3 = 2, and C K = 1 (cf. Eq. 12b ); and : C α 2 = (1 / III Y 2 ), C α 3 = ( – / III Y 3 ), and C α = ln( III Y 3 / D E 3 ), (cf. Eq.12c ). Now let : Q = [ C J 1 ( C K 2 C α 3 – C K 3 C α 2 ) – C J 2 C K 1 C α 3 ]; N JJ = ( II Y J C J / Q ); N KK = ( II Y K C K / Q ); and : N αα = ( C α / Q ), then it could be shown that: III = [( C K 2 C α 3 – C K 3 C α 2 ) N JJ / III Y 1 ] (A.4) III = ( – C J 2 C α 3 N KK / III Y 1 ) (A.4a) III = ( C J 2 C K 3 N αα / III Y 1 ) (A.4b) III = ( – C K 1 C α 3 N JJ / III Y 2 ) (A.5) III = ( C J1 C α 3 N KK / III Y 2 ) (A.5a) III = ( – C J 1 C K3 N αα / III Y 2 ) (A.5b) III = ( C K 1 C α 2 N JJ / III Y 3 ) (A.6) III = ( – C J 1 C α 2 N KK / III Y 3 ) (A.6a) III = [( C J 1 C K 2 – C J 2 C K1 ) N αα / III Y 3 ] (A.6b) Further : III [UF ] 1 = ( III II [UF ] J + III II [UF ] K + III F α ), with : F α = ( u α / G u ); III [UF ] 2 = ( III II [UF ] J + III II [UF ] K + III F α ); and III [UF ] 3 = ( III II [UF ] J + III II [UF ] K + III F α ). 23 Table 1: Stage (DTS) specific parameters for thre e different COCP systems: Eqs. (5-5b), Eqs. (8-8a), and Eqs (9-9a) Block No. Stage No. (DTS) Y d (cf. Eq. 2) [UF ] d (cf. Eq. 7/ 7a/ 7b) d (cf. Eq. 6/ 6a/ 6b) 1 I (Eq. 5) I Y d = 2.0 I = X J / ( X J – X K ) = 2.5, and : I = – X K / ( X J – X K ) = – 1.5 I [UF ] d = K i = J I F i = K i = J I = 4 I d = I [UF ] d G u = 4 G u = 0.2 % II (Eq. 5a) II Y d = 0.75 II = I Y d / ( I Y d + 1) = 0.6667 II [UF ] d = II I [UF ] d = 0.6667 × 4 = 2.667 II d = II [UF] d G u = 2.667 G u = 0.133 % III (Eq. 5b) III Y d = 1.25 III = II Y A / ( II Y d + X L ) = 0.6, and : III = X L / ( II Y d + X L ) = 0.4 III [UF ] d = III II [UF ] d + III F L = (0.6 × 2.667) + (0.4 x 8) = 4.8 III d = III [UF ] d G u = 4.8 G u = 0.24 % 2 I (Eq. 8) Y C = 28.2 mmol dm -3 = 1.0, = - X K / ( X K – X L ) = -2.36, and : = X L / ( X K – X L ) = 1.36 [UF ] C = L i = J F i = 54.4 C = [UF] C G u = 54.44 G u = 27.2 %. II (Eq. 8a) Y G = -15.1 KJ mol -1 = ( A ln ( B / A ) ) -1 = -0.1637 (with : A = [1 + V 0 Y C ], and : B = V 0 Y C ) [UF ] G = [UF] C = 8.9 G = [UF] G G u = 8.9 G u = 4.45 % 3 I (Eq. 9) Y F = 0.01 = -1 , = 1 , and : = 1 [UF ] F = L i = J F i = L i = J = 3 F = [UF] F G u = 3 G u II (Eq. 9a / ) Y D = 0.2 = ( X K X L - X J 2 ) / ( X J Y D ) = -5.5, = X K ( X J – X L ) / ( X J Y D ) = 6.5, and : = X L ( X J – X K ) / ( X J Y D ) = -2.5x10 -8 [UF ] D = L i = J F i = L i = J = 12 D = [UF] D G u = 12 G u 24 Table 2: Stage (DTS) specific parameters for the (COCP as Eqs. 10-13 :) d etermination of constituent carbon and oxygen isotopic abundance ratios in the sample CO 2 gas (S) Stage No. (DTS) Y d (cf. Eq. 2) [UF ] d (cf. Eq. 6/ 6a/ 6b) d (cf. Eq. 5/ 5a/ 5b) I (Eq. 10) I Y J I Y K S = S X J ( A X J + 1) ( D K J + 1) / I Y J = 0.791 A = A X J ( S X J + 1) ( D K J + 1) / I Y J = 0.273 S = S X K ( A X K + 1) ( D K K + 1) / I Y K = 1.19 A = A X K ( S X K + 1) ( D K K + 1) / I Y K = -0.0608 I [UF ] J = S S F J + A A F J = S + A = 1.06 I [UF ] K = S S F K + A A F J = S + A = 1.25 I J = I [UF ] J G u = 1.06 G u I K = I [UF ] K G u =1.25 G u II (Eq. 11) II Y J II Y K II = I Y J / ( I Y J + 1) = -0.0258 II = I Y K / ( I Y K + 1) = 0.0165 II [UF ] J = II I [UF ] J = 0.027 II [UF ] K = II I [UF ] K = 0.021 II J = II [UF ] J G u = 0.027 G u II K = II [UF ] K G u = 0.021 G u III *1 (Eqs. 12a-12c) III Y 1 III Y 2 III Y 3 III = 1.07, III = -0.0352, and III = -5.85x10 -4 III = -0.0011, III = 0.501, and III = 0.0083 III = -0.0021, III = 1.001, and III = -1.6x10 -5 III [UF ] 1 = 0.031 III [UF ] 2 = 0.019 III [UF ] 3 = 0.021 III 1 = 0.031 G u III 2 = 0.019 G u III 3 = 0.021 G u IV (Eq. 13) Y 1 Y 2 Y 3 = III Y 1 / ( III Y 1 – D E 1 ) = -35.5 = III Y 2 / ( III Y 2 – D E 2 ) = 120.4 = III Y 3 / ( III Y 3 – D E 3 ) = 60.5 [UF ] 1 = III [UF ] 1 =1.09 [UF ] 2 = III [UF ] 2 =2.25 [UF ] 3 = III [UF ] 3 = 1.25 1 = [UF] 1 G u = 1.09 G u 2 = [UF] 2 G u = 2.25 G u 3 = [UF] 3 G u = 1.25 G u *1 : See APPENDI X 1 25 Table 3: Examples of variations in the stage specific outputs (for simultaneously determining carbon and oxygen isotopic a bundance ratios as CO 2 + ) as a function of variations in the inputs ( S X J , A X J , S X K , A X K and, ) Example No. I y J x 10 3 (% I J ) I y K x 10 3 (% I K ) II y J x 10 3 (% II J ) II y K x 10 4 (% II K ) III y 1 x 10 3 (% III 1 ) III y 2 x 10 5 (% III 2 ) III y 3 x 10 4 (% III 3 ) y 1 x 10 3 (% 1 ) y 2 x 10 4 (% 2 ) y 3 x 10 3 (% 3 ) 0 *0 -25.1508 (0.0) 16.7446 (0.0) -27.4230 (0.0) 16.8184 (0.0) 00 *00 -25.1508 (0.0) 16.7446 (0.0) 11.6970 (0.0) 42.5548 (0.0) 10.929043 (0.0) 38.399224 (0.0) 21.234718 (0.0) -27.422978 (0.0) 83.741391 (0.0) 16.818404 (0.0) 1 *1 -25.4185 (1.06) 16.9541 (1.25) 11.6938 (-0.027) 42.5636 (0.021) 10.925687 (-0.031) 38.406435 (0.019) 21.239108 (0.021) -27.721608 (1.09) 85.635087 (2.26) 17.028609 (1.25) 2 *2 -24.8831 (-1.06) 16.5351 (-1.25) 11.7002 (0.027) 42.5461 (-0.021) 10.932397 (0.031) 38.392093 (-0.019) 21.230328 (-0.021) -27.124460 (-1.09) 81.868677 (-2.24) 16.608200 (-1.25) 3 *3 -25.4185 (1.06) 16.9541 (1.25) 11.6938 (-0.027) 42.5636 (0.021) 10.925816 (-0.030) 38.399957 (0.0019) 21.239114 (0.021) -27.710078 (1.05) 83.933852 (0.23) 17.028936 (1.25) 4 *4 -25.2811 (0.52) 16.5351 (-1.25) 11.6955 ( - 0.013) 42.5461 (-0.021) 10.927622 ( - 0.013) 38.392109 (-0.019) 21.230346 (-0.021) -27.549384 (0.46) 81.872982 (-2.23) 16.609077 (-1.24) 5 *5 -25.4185 (1.06) 16.9338 (1.13) 11.6938 (-0.027) 42.5628 (0.019) 10.925695 (-0.031) 38.406047 (0.018) 21.238682 (0.019) -27.720916 (1.09) 85.533014 (2.14) 17.008227 (1.13) 6 *6 -25.1508 (0.0) 16.7446 (0.0) 11.6970 (0.0) 42.5548 (0.0) 10.928659 (-0.0035) 38.418423 ( 0.05 ) 21.234698 (-0.000095) -27.457148 (0.125) 88.783019 ( 6.02 ) 16.817435 (-0.0058) *0 : Results as reported 10 for : S X J = -0.020, A X J = -0.0070, S X K = 0.02 0, A X K = -0.0010, and = 0.5 (cf. the te xt). *00 : A verification that o ur evaluated results (for : x i (s) = X i (s) , i.e. for : i (s) = 0.0) are the same as given in [1 0]. *1 : S x J = -0.0202 , A x J = -0.00707, S x K = 0.0202 , A x K = -0.00099, and = 0.5050 (i.e. : S J = A J = S K = = 1% and : A K = -1%). *2 : Results for “ S J = A J = S K = = -1% and : A K = 1%”. *3 : S J = A J = S K = 1%, and : A K = = -1%. *4 : S J = A K = 1%, and : S K = A J = = -1%. *5 : S J = A J = S K = A K = = 1%. *6 : S J = A J = S K = A K = 0.0 , b ut = 6% .
Original Paper
Loading high-quality paper...
Comments & Academic Discussion
Loading comments...
Leave a Comment