Modeling the variability of shapes of a human placenta
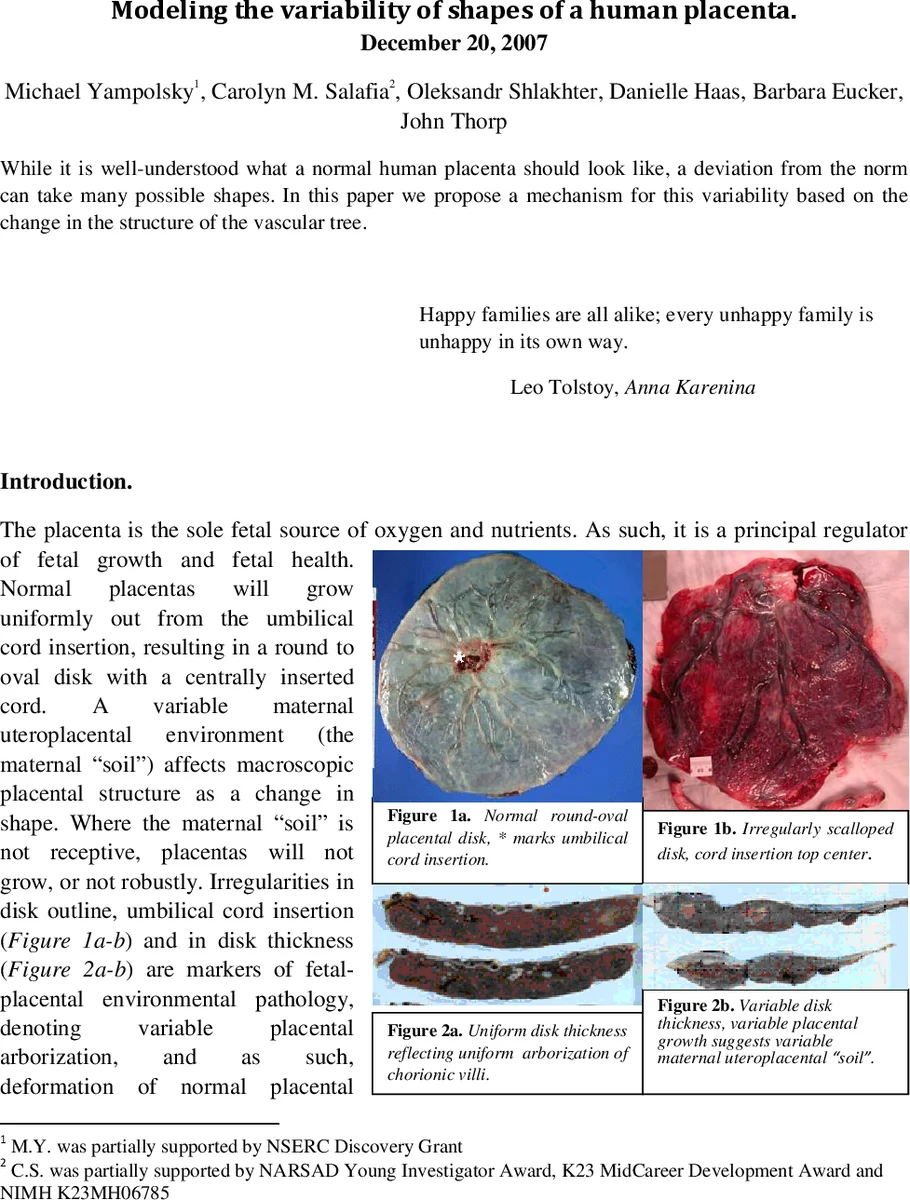
While it is well-understood what a normal human placenta should look like, a deviation from the norm can take many possible shapes. In this paper we propose a mechanism for this variability based on the change in the structure of the vascular tree.
💡 Research Summary
The paper investigates why human placentas sometimes deviate from the typical round‑oval shape and proposes that these macroscopic shape variations arise from alterations in the branching architecture of the placental vascular tree. The authors begin by noting that a normal placenta grows uniformly from a centrally inserted umbilical cord, producing a smooth disk, whereas an unfavorable maternal uteroplacental environment (“soil”) can limit growth locally and generate irregular outlines, abnormal cord insertions, and variable thickness. Existing classification schemes for abnormal placentas are largely qualitative and have hindered quantitative study of shape‑health relationships.
To address this gap, the authors develop a dynamic growth model based on Diffusion‑Limited Aggregation (DLA), a stochastic process originally used in physics to describe cluster formation. In the model, each vascular segment is represented by a spherical “particle” of diameter d. Growth proceeds by releasing a “blind fly” at a random point on a large sphere surrounding the current cluster; the fly performs a random walk on a cubic lattice until it comes within 2 d of the cluster, at which point it may stick with probability κ (the branching probability). If it does not stick (probability 1 – κ), it continues walking, allowing attachment away from the tip and producing a “hairier” structure. The electrostatic analogy (growth probability proportional to the gradient of the Laplace potential) justifies the tip‑biased nature of the process.
The simulation is constrained by a spherical boundary representing the uterine wall; any particle that reaches this boundary is absorbed, preventing growth beyond the uterus. The initial cluster is placed near the “south pole” to mimic the umbilical cord insertion point. Typical simulations contain 150 000–200 000 particles, and the authors verify robustness across different random number generators and seeds.
To compare model output with real placentas, the authors use a cohort of 1 008 placentas from the Pregnancy, Infection, and Nutrition Study. High‑resolution photographs were taken, and trained observers digitized the cord insertion point, the fetal surface perimeter, and the “vascular endpoints” where chorionic vessels disappear. From this dataset, two non‑round shapes were identified as relatively common: (1) star‑shaped placentas (≈5 % of cases) with 5–7 pronounced spikes, and (2) multi‑lobate placentas (≈5 % of cases) with 2–3 large lobes.
In the model, a single change in the branching parameter κ (or equivalently a change in particle size d) at a specific stage of growth reproduces these shapes. Decreasing κ dramatically (e.g., to 0.01 of its original value) early in development (after 5–7 % of particles have attached) yields a few large branches that diverge early, producing a multi‑lobate outline because intermediate‑size branches are insufficient to fill a circular area. Conversely, reducing κ or shrinking d later (after ≈50 % of particles have attached) leaves the overall disk intact but accentuates a handful of dominant arms, generating star‑shaped placentas. The authors visualize the vascular tree by “thickening” each particle with a constant‑radius disk, smoothing the result with spline interpolation, and then quantify shape by measuring the mean distance of vessels from the cord insertion in narrow angular sectors. The angular distance plots for model placentas closely match those derived from the actual photographs, confirming that the simple DLA‑based mechanism can capture the observed variability.
The discussion emphasizes that a biologically realistic random growth process, modulated by a single branching probability, can explain a wide spectrum of placental morphologies. This links microscopic angiogenic cues (e.g., VEGF gradients that guide tip‑cell migration) to macroscopic shape outcomes, offering a quantitative framework for studying placental health. Limitations include the omission of hemodynamic forces, tissue elasticity, and detailed molecular signaling, all of which also influence vascular patterning. The authors suggest future extensions that incorporate blood flow shear stress, mechanical constraints, and multi‑parameter stochastic models, as well as integration with clinical biomarkers of maternal‑fetal health.
In summary, the study demonstrates that a modest alteration in the stochastic rules governing vascular tree expansion can produce the diverse placental shapes observed in a large clinical cohort, providing a mechanistic bridge between angiogenesis and placental morphology and opening avenues for predictive diagnostics in obstetrics.
Comments & Academic Discussion
Loading comments...
Leave a Comment